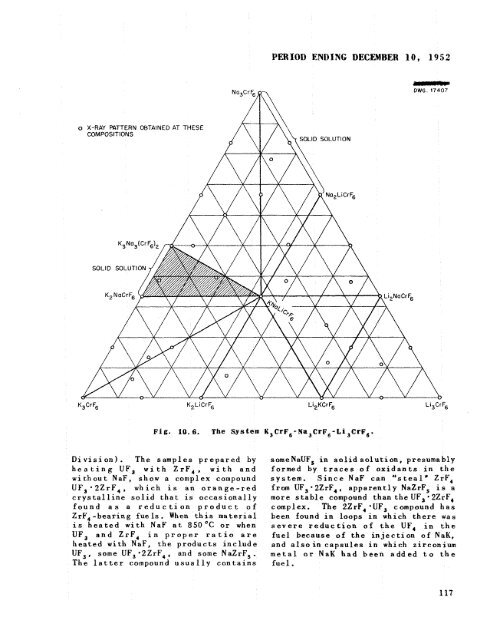
o X-RAY PATTERN OBTAINED AT THESE COMPOSITIONS SOLID SOLUTIO PIERIOD ENDING DECEMBER 10, 1952 SOLID SOLUTION - DWG. 17407 K3CrF6 K2 L i Cr F6 Li2KCrF6 Li,CrF, Fig. 10.6. The System K3CrF,-Na,CrF,-Li3CrFd. Division). The samples prepared by heating UF, with ZrF4, with and without NaF, show a complex compound UF3*2ZrF4, which is an orange-red crystalline solid that is occasionally found as a reduction product of ZrF4 -bearing fue is. When this material is heated with NaF at 850 OC or when UF, and ZrF, in proper ratio are heated with NaF, <strong>the</strong> products include UF,, some UF, *2ZrF,, and some NaZrFS. The latter compound usually contains someNaUFS in solid solution, presumably formed by traces of oxidants in <strong>the</strong> system. Since NaF can "steal' ZrF, from UF;2ZrF4, apparently NaZrFS is a more stable compound than<strong>the</strong>UFJ-2ZrF4 complex. The 2ZrF, *UF, compound has been found in loops in which <strong>the</strong>re was severe reduction of <strong>the</strong> UF, in <strong>the</strong> fuel because of <strong>the</strong> injection of NaK, and alsoin capsules in which zirconium metal or NaK had been added to <strong>the</strong> fuel. 117
ANP PR0,jlECT QUARTERLY PROGRESS REPORT NaF-ZrF, (P. A. Agron, Materials Chemistry Division) ‘The x-ray studies of <strong>the</strong> NaF-ZrF, system(g) have been extended to <strong>the</strong> region of 75 mole % of ZrF,. The crystal structures of <strong>the</strong> solid phases Na,ZrF, and NaZrF5 have been reported previously.( lo) Studies of <strong>the</strong> behavior of <strong>the</strong> phases that appear in cornpositions lying between <strong>the</strong>se two compounds and of those that lie beyond <strong>the</strong> NaZrF5 compositions are being made; considerable additional study w i l l be required, since <strong>the</strong> system is an exceedingly complex one. O<strong>the</strong>r Fluoride ComnpSexes (8. J. Sturm and I,. G. Overholser, Mat.erials Chemistry Division). It was previously noted that a preparation corresponding to K2NaFeFs was thonght to be iso- morphous with K2NaCrF6. Fur<strong>the</strong>r study has shown that Na3FeF, and K,FeF, apparently form solid solutions over a wide range, probably in all proportions, X-ray patterns for materials corre- sponding to K,FeF,, K3NaFeF6, K,Na( FeF,),, and KNa2FeF, differ only by slight shifts in lines, Fusion of equal molar proportions of KMF,, NaHF,, and NiF;4H,O at 850°C resulted in a yellow compound, probably KNaNiF,. A fusion in <strong>the</strong> proportions corresponding to K3Na(NiF2), gave a mixture of K,NiF, and KNaNiF,. Heating a mixture of KNiF,*lXH,O (prepared from aqueous solution) and NH,F, at 700’C yielded a product that agrees with <strong>the</strong> ASTM x-ray diffraction data for anhydrous KNiF,. Interaction of chromium metal. with K,NiF, at 850°C for 5 hr resulted in <strong>the</strong> formation of K3CrF, by <strong>the</strong> f ol 1 o wi 11 g rea c t ion : 3K,NiF, f 2Cr ---+ 2K3CrF, + 3Ni . This behavior is comparable to that previously noted for displacement of Fe from K,NaFeF, by Cr. (9)L. M. Bretcber and C. J. Barton, wCoolaat Development, ” this chapter. (lo)@. Agron, ANP Quar. Prog. Rep. Sept. 20, 19.52, ORML-1375, p. 85. 118 REACTIONS OF FEIJBBIXDE MIXTURES WITH REDIlCXWG AGENTS W. 13. Grimes Materials Chemistry Division L. A. Mann ANP Division The reactions of possible ARE fuel mixtures with reducing agents were first examined in an effort to evaluate <strong>the</strong> damage that would result if NaK were inadvertently admitted to <strong>the</strong> fuel circuit during operation. Identi- fication of <strong>the</strong> prodiicts in <strong>the</strong>se complex mixtures was shown to bea very di f ficu 1 t pro blem. Ne cen tly , however, it has been demonstrated that addition of reducing agents to <strong>the</strong> fluoride melts decreases <strong>the</strong> corrosion by <strong>the</strong>se materials. Since production of UF, and <strong>the</strong> consequent precipitation of uranium at high temperature can result from excessive addition of reductant, a careful studyof <strong>the</strong> fluoride systems under reducing conditions has been undertaken. Reducing Power of Varioiis Additives (J. C. White, Analytical Chemistry Division). The total reducing power of reaction products resulting from <strong>the</strong> addition of NaK to such materials as ZrF,, NaF, and UF, and such mixtures as NaF-ZrF, -1JF, and NaF-ZrF,, has been measured by two methods: (I) oxi- dation with standard ceric sulfate solution and (2) hydrogen evolution from dissolution in mineral acid. The validity of <strong>the</strong>se procedures has been verified by using control samples, such as UF, and Zr. In practically none of <strong>the</strong> NaK addition tests, however, has a reduci-rig action equiva- lent to <strong>the</strong> amount of NaK added been observed . This reduction phenomenon has also been observed when UF, has been added to NaF and ZrF, and treated as in fuel preparation. One compound, Na3U,F,, gave nearly <strong>the</strong> <strong>the</strong>oretical amount of UF, added. Several samples of fuels that had undergone corrosion testing were


![Review of Molten Salt Reactor Physics Calculations [Disc 2]](https://img.yumpu.com/21979492/1/190x247/review-of-molten-salt-reactor-physics-calculations-disc-2.jpg?quality=85)












