ORNL-1816 - the Molten Salt Energy Technologies Web Site
ORNL-1816 - the Molten Salt Energy Technologies Web Site
ORNL-1816 - the Molten Salt Energy Technologies Web Site
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
It should be noted that of <strong>the</strong> types of reactions<br />
discussed above only a few examples have been<br />
studied experimentally in a detailed or quantitative<br />
way; most have not been studied at all or else<br />
are represented experimentally by some of <strong>the</strong><br />
more obvious and less interesting examples.<br />
kali Metal Solutions at<br />
High Temperatures<br />
G. P. Smith M. E. Steidlitz<br />
rgy Division<br />
In <strong>the</strong> prece port12 data were given for<br />
<strong>the</strong> flammability of alloys of <strong>the</strong> sodium-bismuth<br />
system. Additional data have now been obtained<br />
for bismuth-rich alloys that provide a much more<br />
complete picture of <strong>the</strong> reactivity of this system.<br />
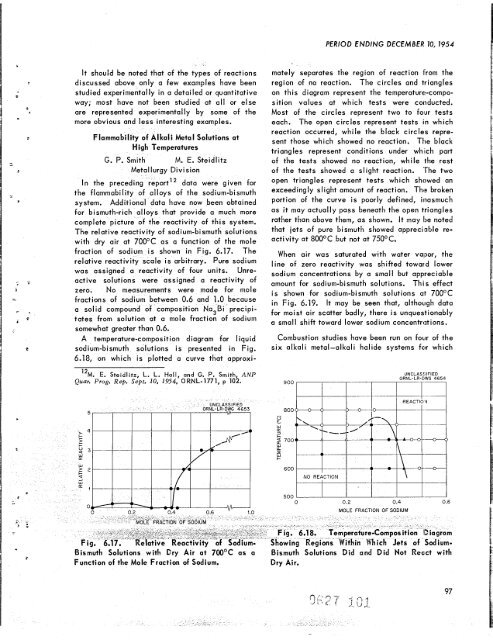
The relative reactivity of sodium-bismuth solutions<br />
with dry air at 700°C as a function of <strong>the</strong> mole<br />
fraction of sodium is shown in Fig. 6.17. The<br />
relative reactivity scale is arbitrary. Pure sodium<br />
was assigned a reactivity of four units. Unreactive<br />
solutions were assigned a reactivity of<br />
zero. No measurements were made for mole<br />
fractions of sodium between 0.6 and 1.0 because<br />
a solid compound of composition Na,Bi precipitates<br />
from solution at a male fraction of sodium<br />
somewhat greater than 0.6.<br />
A temperature-composition diagram for liquid<br />
sodium-bismuth solutions is presented in Fig.<br />
6.18, on which is plotted a curve that approxi-<br />
12M. E. Steidlitz, L. L. Hall, and G. P. Smith ANP<br />
Quar. Prog. Rep. Sept. 10, 1954, <strong>ORNL</strong>-1771, p 105.<br />
5<br />
I<br />
PERIOD ENDlNG DECEMBER 70,1954<br />
mately separates <strong>the</strong> region of reaction from <strong>the</strong><br />
region of no reaction. The circles and triangles<br />
on this diagram represent <strong>the</strong> temperature-compo-<br />
sition values at which tests were conducted.<br />
Most of <strong>the</strong> circles represent two to folur tests<br />
each. The open circles represent tests in which<br />
reaction occurred, while <strong>the</strong> black circles repre-<br />
sent those which showed no reaction. The black<br />
triangles represent conditions under whiich part<br />
of <strong>the</strong> tests showed no reaction, while <strong>the</strong> rest<br />
of <strong>the</strong> tests showed a slight reaction. ‘The two<br />
open triangles represent tests which showed an<br />
exceedingly slight amount of reaction. The broken<br />
portion of <strong>the</strong> curve is poorly defined, inasmuch<br />
as it may actually pass beneath <strong>the</strong> open triangles<br />
ra<strong>the</strong>r than above <strong>the</strong>m, as shown. It may be noted<br />
that jets of pure bismuth showed appreciuble re-<br />
activity at 800°C but not at 75OOC.<br />
When air was saturated with water vapor, <strong>the</strong><br />
line of zero reactivity was shifted toward lower<br />
sodium concentrations by a smal I but appreciable<br />
amount for sodium-bismuth solutions. This effect<br />
is shown for sodium-bismuth solutions at 700°C<br />
in Fig. 6.19. It may be seen that, although data<br />
for moist air scatter badly, <strong>the</strong>re is unquestionably<br />
a small shift toward lower sodium concentrations.<br />
Combustion studies have been run on four of <strong>the</strong><br />
six alkali metal-alkali halide systems far which<br />
, , , I <strong>ORNL</strong>-LR-rWG<br />
9oo<br />
800<br />
700<br />
600<br />
UNCLASSIFIED<br />
4654,<br />
500 I<br />
0 0.2 0.4 0.6<br />
MOLE FRACTION OF SODIUM<br />
Bismuth Solutions with Cry Air at 700°C as a Bismuth Solutions Did and Did Not React with<br />
Function of <strong>the</strong> Mole Fraction of Sodium. Dry Air.<br />
97


![Review of Molten Salt Reactor Physics Calculations [Disc 2]](https://img.yumpu.com/21979492/1/190x247/review-of-molten-salt-reactor-physics-calculations-disc-2.jpg?quality=85)












