150 Appendix 4 TABLE 73 Meta-analyses: etanercept s.c. all doses (including sublicence doses) versus placebo (with or without ongoing conventional DMARDs), end <strong>of</strong> trial Comparison or outcome Studies N included Statistical Effect size (95% CI) in analysis method ACR20 responder 7103,121,122,125,126,129,130 1672 RR (fixed) 3.48 (2.78 to 4.35)* ACR50 responder 7103,121,122,125,126,129,130 1672 RR (fixed) 4.97 (3.40 to 7.27)* ACR70 responder 6103,122,125,126,129,130 1492 RR (fixed) 8.55 (3.59 to 20.37)* RD ACR20 responder 7103,121,122,125,126,129,130 1672 RD (fixed) 0.43 (0.38 to 0.47)* RD ACR50 responder 7103,121,122,125,126,129,130 1672 RD (fixed) 0.26 (0.22 to 0.30)* RD ACR70 responder 6103,122,125,126,129,130 1492 RD (fixed) 0.11 (0.08 to 0.14)* SJC, end <strong>of</strong> study result 7103,121,122,125,126,129,130 1689 WMD (random) –5.78 (–8.12 to –3.43)* Patient’s global assessment, end <strong>of</strong> study 7 103,121,122,125,126,129,130 1689 WMD –2.33 (–2.56 to –2.10)* result (fixed) HAQ, end <strong>of</strong> study result 6 103,122,125,126,129,130 1440 WMD (fixed) –0.49 (–0.57 to –0.40)* DAS, end <strong>of</strong> study result 1 103 150 WMD (fixed) –1.50 (–1.89 to –1.11)* Modified van de Heijde–Sharp score, 0 0 Not No data available mean change from baseline estimable Withdrawal for any reasons 7 103,104,121,122,125,126,129 2168 RR (fixed) 0.43 (0.36 to 0.51)* Withdrawal due to lack <strong>of</strong> efficacy 6103,104,121,122,125,126 1748 RR (fixed) 0.28 (0.21 to 0.36)* Withdrawal due to adverse events 7103,104,121,122,125,126,129 2168 RR (fixed) 0.87 (0.54 to 1.38) Death 7103,104,121,122,125,126,129 2168 RR (fixed) 1.44 (0.44 to 4.69) SAEs 5103,104,122,125,129 1429 RR (fixed) 1.25 (0.76 to 2.06) Malignancy: all 6103,104,122,125,126,129 1988 RR (fixed) 0.47 (0.13 to 1.67) Malignancy: skin cancer excluding 6103,104,122,125,126,129 melanoma 1988 RR (fixed) 0.64 (0.15 to 2.77) Malignancy: all cancer excluding 6103,104,122,125,126,129 non-melanoma skin cancer 1988 RR (fixed) 0.34 (0.07 to 1.74) Serious infection 7103,104,122,125,126,129,130 2046 RR (fixed) 0.75 (0.37 to 1.48) Any infection 6103,104,122,125,126,129 1988 RR (random) 1.01 (0.83 to 1.24) * Statistically significant result (p < 0.05).
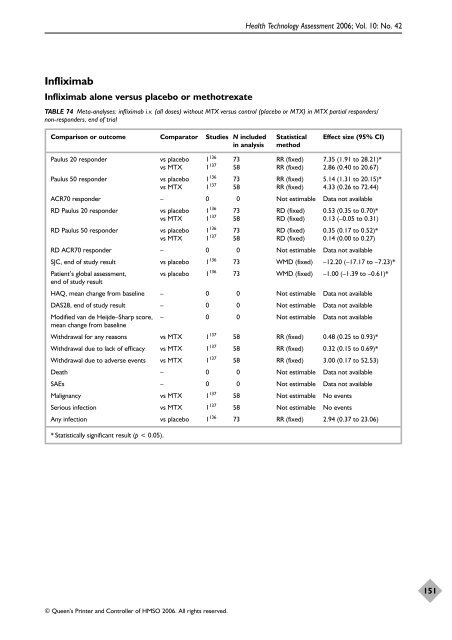
Infliximab Infliximab alone versus placebo or methotrexate © Queen’s Printer and Controller <strong>of</strong> HMSO 2006. All rights reserved. Health Technology Assessment 2006; Vol. 10: No. 42 TABLE 74 Meta-analyses: infliximab i.v. (all doses) without MTX versus control (placebo or MTX) in MTX partial responders/ non-responders, end <strong>of</strong> trial Comparison or outcome Comparator Studies N included Statistical Effect size (95% CI) in analysis method Paulus 20 responder vs placebo 1136 73 RR (fixed) 7.35 (1.91 to 28.21)* vs MTX 1 137 58 RR (fixed) 2.86 (0.40 to 20.67) Paulus 50 responder vs placebo 1136 73 RR (fixed) 5.14 (1.31 to 20.15)* vs MTX 1 137 58 RR (fixed) 4.33 (0.26 to 72.44) ACR70 responder – 0 0 Not estimable Data not available RD Paulus 20 responder vs placebo 1136 73 RD (fixed) 0.53 (0.35 to 0.70)* vs MTX 1 137 58 RD (fixed) 0.13 (–0.05 to 0.31) RD Paulus 50 responder vs placebo 1136 73 RD (fixed) 0.35 (0.17 to 0.52)* vs MTX 1 137 58 RD (fixed) 0.14 (0.00 to 0.27) RD ACR70 responder – 0 0 Not estimable Data not available SJC, end <strong>of</strong> study result vs placebo 1136 73 WMD (fixed) –12.20 (–17.17 to –7.23)* Patient’s global assessment, vs placebo 1136 end <strong>of</strong> study result 73 WMD (fixed) –1.00 (–1.39 to –0.61)* HAQ, mean change from baseline – 0 0 Not estimable Data not available DAS28, end <strong>of</strong> study result – 0 0 Not estimable Data not available Modified van de Heijde–Sharp score, mean change from baseline – 0 0 Not estimable Data not available Withdrawal for any reasons vs MTX 1137 58 RR (fixed) 0.48 (0.25 to 0.93)* Withdrawal due to lack <strong>of</strong> efficacy vs MTX 1137 58 RR (fixed) 0.32 (0.15 to 0.69)* Withdrawal due to adverse events vs MTX 1137 58 RR (fixed) 3.00 (0.17 to 52.53) Death – 0 0 Not estimable Data not available SAEs – 0 0 Not estimable Data not available Malignancy vs MTX 1137 58 Not estimable No events Serious infection vs MTX 1137 58 Not estimable No events Any infection vs placebo 1136 73 RR (fixed) 2.94 (0.37 to 23.06) * Statistically significant result (p < 0.05). 151
- Page 1 and 2:
A systematic review of the effectiv
- Page 3 and 4:
A systematic review of the effectiv
- Page 5 and 6:
Objectives: This report reviews the
- Page 7:
Glossary and list of abbreviations
- Page 10 and 11:
viii Glossary and list of abbreviat
- Page 13 and 14:
Background Rheumatoid arthritis (RA
- Page 15:
increased risk of serious infection
- Page 19 and 20:
Summary RA is a common, chronic, in
- Page 21 and 22:
(IL-2) and interleukin-6 (IL-6), pr
- Page 23 and 24:
Assessment of response to DMARDs Re
- Page 25 and 26:
combination with methotrexate or al
- Page 27:
disease between clinic appointments
- Page 30 and 31:
14 Effectiveness in juvenile arthri
- Page 32 and 33:
16 Effectiveness adalimumab plus me
- Page 34 and 35:
18 Effectiveness Monoclonal Antibod
- Page 36 and 37:
20 TABLE 1 Description of included
- Page 38 and 39:
22 TABLE 2 Quality of included RCTs
- Page 40 and 41:
24 Effectiveness change in modified
- Page 42 and 43:
26 Effectiveness TABLE 4 Meta-analy
- Page 44 and 45:
28 Effectiveness Review: Adalimumab
- Page 46 and 47:
30 Effectiveness Review: Adalimumab
- Page 48 and 49:
32 TABLE 6 Effectiveness Descriptio
- Page 50 and 51:
34 TABLE 6 Effectiveness Descriptio
- Page 52 and 53:
36 TABLE 7 Effectiveness Quality of
- Page 54 and 55:
38 Effectiveness etanercept trials.
- Page 56 and 57:
40 Effectiveness TABLE 9 Summary of
- Page 58 and 59:
42 Effectiveness Review: Etanercept
- Page 60 and 61:
44 Effectiveness Review: Etanercept
- Page 62 and 63:
46 Effectiveness Review: Etanercept
- Page 64 and 65:
48 Effectiveness Review: Etanercept
- Page 66 and 67:
50 Effectiveness Review: Etanercept
- Page 68 and 69:
52 Effectiveness Review: Etanercept
- Page 70 and 71:
54 Effectiveness TABLE 11 Summary o
- Page 72 and 73:
56 TABLE 12 Description of included
- Page 74 and 75:
58 TABLE 13 Quality of included RCT
- Page 76 and 77:
60 Effectiveness per week and escal
- Page 78 and 79:
62 Effectiveness significant advant
- Page 80 and 81:
64 Effectiveness Review: Infliximab
- Page 82 and 83:
66 Effectiveness Review: Infliximab
- Page 84 and 85:
68 Effectiveness FIGURE 44 Malignan
- Page 86 and 87:
70 TABLE 17 Effectiveness Summary o
- Page 89 and 90:
Summary of review of existing econo
- Page 91 and 92:
TABLE 20 Summary of published ICERs
- Page 93 and 94:
TABLE 21 Published etanercept econo
- Page 95 and 96:
TABLE 23 Published economic analyse
- Page 97 and 98:
TABLE 25 Treatment sequences: adali
- Page 99 and 100:
management of RA, i.e. 1st and 2nd
- Page 101 and 102:
To estimate the long-term consequen
- Page 103 and 104:
TABLE 32 TNF inhibitors as last act
- Page 105 and 106:
TABLE 34 Basic structure of the mod
- Page 107 and 108:
een quit on grounds of toxicity, ad
- Page 109 and 110:
TABLE 39 Strategy set: adalimumab a
- Page 111 and 112:
TABLE 43 Beta distributions for HAQ
- Page 113 and 114:
TABLE 44 Early cessation of DMARDs:
- Page 115 and 116: TABLE 46 Unit costs for tests and v
- Page 117 and 118: following properties, according to
- Page 119 and 120: TABLE 52 Base case: TNF inhibitors
- Page 121 and 122: TABLE 54 Base case: TNF inhibitors
- Page 123 and 124: TABLE 59 Third TNF inhibitor follow
- Page 125 and 126: TABLE 65 Sensitivity analyses: TNF
- Page 127 and 128: TABLE 67 Sensitivity analyses: TNF
- Page 129: The substantial economic impact of
- Page 133 and 134: Summary Effectiveness: principal fi
- Page 135 and 136: inhibitors, although the incrementa
- Page 137 and 138: introduce bias which generally exag
- Page 139: Adalimumab, etanercept and inflixim
- Page 143 and 144: 1. Jobanputra P, Barton P, Bryan S,
- Page 145 and 146: 46. Young A, Dixey J, Cox N, Davies
- Page 147 and 148: tumor necrosis factor therapy in th
- Page 149 and 150: arthritis: a 12-week, double-blind,
- Page 151 and 152: 171. Geborek P, Crnkic M, Petersson
- Page 153 and 154: 216. Schotte H, Willeke P, Mickholz
- Page 155 and 156: The Health Assessment Questionnaire
- Page 157 and 158: Cochrane Library (CENTRAL) 2005 Iss
- Page 159 and 160: Appendix 3 © Queen’s Printer and
- Page 161: TABLE 69 Studies excluded from clin
- Page 164 and 165: 148 Appendix 4 TABLE 71 Meta-analys
- Page 168 and 169: 152 Appendix 4 Infliximab versus pl
- Page 170 and 171: 154 Appendix 4 Infliximab plus MTX
- Page 173: Ovid MEDLINE(R) 1966 to February we
- Page 177 and 178: Appendix 8 © Queen’s Printer and
- Page 179 and 180: TABLE 79 Wong et al., 2002 161 © Q
- Page 181 and 182: TABLE 80 Kobelt et al., 2003 162 (c
- Page 183 and 184: TABLE 82 Brennan et al., 2004 160
- Page 185 and 186: TABLE 83 Kobelt et al., 2004 163 (c
- Page 187 and 188: TABLE 85 Bansback et al., 2005 166
- Page 189: TABLE 86 Kobelt et al., 2005 167 (c
- Page 192 and 193: 176 Appendix 9 TABLE 89 Strategy se
- Page 195 and 196: Extensive sensitivity analysis was
- Page 197 and 198: TABLE 96 Variation 1: TNF inhibitor
- Page 199 and 200: TABLE 99 Variation 2: TNF inhibitor
- Page 201 and 202: TABLE 102 Variation 3: TNF inhibito
- Page 203 and 204: TABLE 105 Variation 3: TNF inhibito
- Page 205 and 206: TABLE 108 Variation 4: TNF inhibito
- Page 207 and 208: TABLE 111 Variation 5: TNF inhibito
- Page 209 and 210: TABLE 114 Variation 6: TNF inhibito
- Page 211 and 212: TABLE 117 Variation 6: TNF inhibito
- Page 213 and 214: TABLE 120 Variation 7: TNF inhibito
- Page 215 and 216: TABLE 123 Variation 8: TNF inhibito
- Page 217 and 218:
TABLE 126 Variation 9: TNF inhibito
- Page 219 and 220:
TABLE 129 Variation 9: TNF inhibito
- Page 221 and 222:
TABLE 132 Variation 10: TNF inhibit
- Page 223 and 224:
TABLE 135 Variation 11: TNF inhibit
- Page 225 and 226:
TABLE 138 Variation 12: TNF inhibit
- Page 227 and 228:
TABLE 141 Variation 12: TNF inhibit
- Page 229 and 230:
TABLE 144 Variation 13: TNF inhibit
- Page 231 and 232:
TABLE 147 Variation 14: TNF inhibit
- Page 233 and 234:
TABLE 150 Variation 15: TNF inhibit
- Page 235 and 236:
TABLE 153 Variation 15: TNF inhibit
- Page 237 and 238:
TABLE 156 Variation 16: TNF inhibit
- Page 239 and 240:
TABLE 159 Variation 17: TNF inhibit
- Page 241 and 242:
TABLE 162 Variation 18: TNF inhibit
- Page 243:
TABLE 165 Variation 18: TNF inhibit
- Page 247 and 248:
Volume 1, 1997 No. 1 Home parentera
- Page 249 and 250:
No. 3 Screening for sickle cell dis
- Page 251 and 252:
No. 25 A rapid and systematic revie
- Page 253 and 254:
No. 11 First and second trimester a
- Page 255 and 256:
No. 23 Clinical effectiveness and c
- Page 257 and 258:
No. 28 Outcomes of electrically sti
- Page 259:
No. 28 Adefovir dipivoxil and pegyl
- Page 262 and 263:
246 Health Technology Assessment Pr
- Page 264:
248 Health Technology Assessment Pr