A systematic review of the effectiveness of adalimumab
A systematic review of the effectiveness of adalimumab
A systematic review of the effectiveness of adalimumab
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
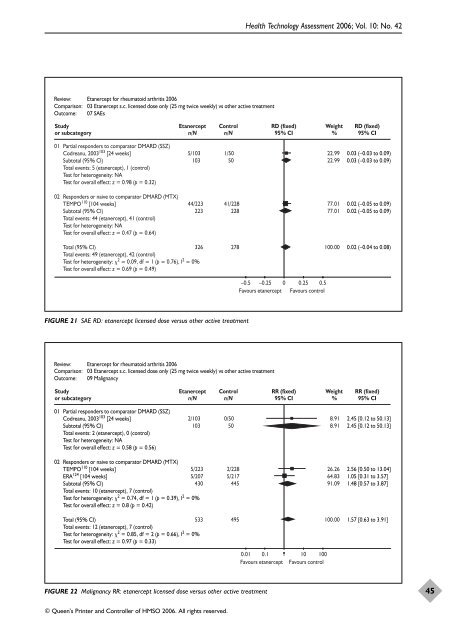
Review: Etanercept for rheumatoid arthritis 2006<br />
Comparison: 03 Etanercept s.c. licensed dose only (25 mg twice weekly) vs o<strong>the</strong>r active treatment<br />
Outcome: 07 SAEs<br />
Study<br />
or subcategory<br />
01 Partial responders to comparator DMARD (SSZ)<br />
Codreanu, 2003 103 [24 weeks]<br />
Subtotal (95% CI)<br />
Total events: 5 (etanercept), 1 (control)<br />
Test for heterogeneity: NA<br />
Test for overall effect: z = 0.98 (p = 0.32)<br />
02 Responders or naive to comparator DMARD (MTX)<br />
TEMPO 110 [104 weeks]<br />
Subtotal (95% CI)<br />
Total events: 44 (etanercept), 41 (control)<br />
Test for heterogeneity: NA<br />
Test for overall effect: z = 0.47 (p = 0.64)<br />
Etanercept<br />
n/N<br />
© Queen’s Printer and Controller <strong>of</strong> HMSO 2006. All rights reserved.<br />
Control<br />
n/N<br />
5/103 1/50<br />
103 50<br />
44/223 41/228<br />
223 228<br />
Total (95% CI)<br />
Total events: 49 (etanercept), 42 (control)<br />
Test for heterogeneity: 2 = 0.09, df = 1 (p = 0.76), I2 326 278<br />
= 0%<br />
Test for overall effect: z = 0.69 (p = 0.49)<br />
FIGURE 21 SAE RD: etanercept licensed dose versus o<strong>the</strong>r active treatment<br />
Review: Etanercept for rheumatoid arthritis 2006<br />
Comparison: 03 Etanercept s.c. licensed dose only (25 mg twice weekly) vs o<strong>the</strong>r active treatment<br />
Outcome: 09 Malignancy<br />
Study<br />
or subcategory<br />
01 Partial responders to comparator DMARD (SSZ)<br />
Codreanu, 2003 103 [24 weeks]<br />
Subtotal (95% CI)<br />
Total events: 2 (etanercept), 0 (control)<br />
Test for heterogeneity: NA<br />
Test for overall effect: z = 0.58 (p = 0.56)<br />
Etanercept<br />
n/N<br />
Control<br />
n/N<br />
2/103 0/50<br />
103 50<br />
02 Responders or naive to comparator DMARD (MTX)<br />
TEMPO110 [104 weeks]<br />
ERA124 [104 weeks]<br />
Subtotal (95% CI)<br />
Total events: 10 (etanercept), 7 (control)<br />
Test for heterogeneity: 2 = 0.74, df = 1 (p = 0.39), I2 5/223 2/228<br />
5/207 5/217<br />
430 445<br />
= 0%<br />
Test for overall effect: z = 0.8 (p = 0.42)<br />
Total (95% CI)<br />
Total events: 12 (etanercept), 7 (control)<br />
Test for heterogeneity: 2 = 0.85, df = 2 (p = 0.66), I2 533 495<br />
= 0%<br />
Test for overall effect: z = 0.97 (p = 0.33)<br />
FIGURE 22 Malignancy RR: etanercept licensed dose versus o<strong>the</strong>r active treatment<br />
Health Technology Assessment 2006; Vol. 10: No. 42<br />
RD (fixed)<br />
95% CI<br />
–0.5 –0.25 0 0.25 0.5<br />
Favours etanercept Favours control<br />
RR (fixed)<br />
95% CI<br />
0.01 0.1 1 10 100<br />
Favours etanercept Favours control<br />
Weight<br />
%<br />
22.99<br />
22.99<br />
77.01<br />
77.01<br />
100.00<br />
Weight<br />
%<br />
8.91<br />
8.91<br />
26.26<br />
64.83<br />
91.09<br />
100.00<br />
RD (fixed)<br />
95% CI<br />
0.03 (–0.03 to 0.09)<br />
0.03 (–0.03 to 0.09)<br />
0.02 (–0.05 to 0.09)<br />
0.02 (–0.05 to 0.09)<br />
0.02 (–0.04 to 0.08)<br />
RR (fixed)<br />
95% CI<br />
2.45 [0.12 to 50.13]<br />
2.45 [0.12 to 50.13]<br />
2.56 [0.50 to 13.04]<br />
1.05 [0.31 to 3.57]<br />
1.48 [0.57 to 3.87]<br />
1.57 [0.63 to 3.91]<br />
45