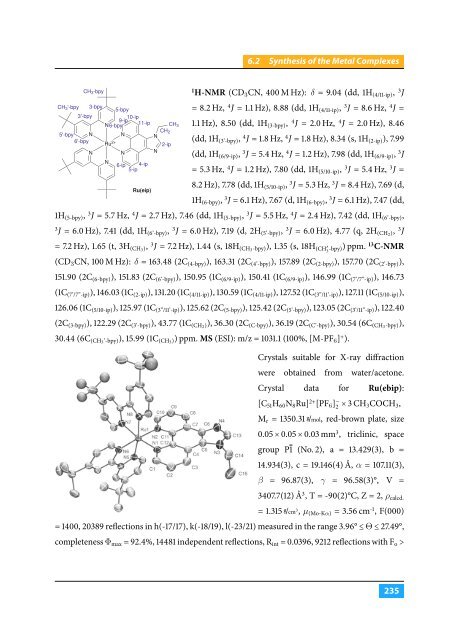
|6.2 Synthesis of the Metal Complexes| CH 3 -bpy 1 H-NMR (CD 3 CN, 400 M Hz): δ = 9.04 (dd, 1H (4/11-ip) , 3 J CH 3 '-bpy 3'-bpy 5'-bpy N 6'-bpy 3-bpy N 9-ip 10-ip N6-bpy 11-ip Ru 2+ N 5-bpy N N 6-ip 4-ip 5-ip Ru(eip) CH 2 N N 2-ip CH 3 = 8.2 Hz, 4 J = 1.1 Hz), 8.88 (dd, 1H (4/11-ip) , 3 J = 8.6 Hz, 4 J = 1.1 Hz), 8.50 (dd, 1H (3-bpy) , 4 J = 2.0 Hz, 4 J = 2.0 Hz), 8.46 (dd, 1H (3’-bpy) , 4 J = 1.8 Hz, 4 J = 1.8 Hz), 8.34 (s, 1H (2-ip) ), 7.99 (dd, 1H (6/9-ip) , 3 J = 5.4 Hz, 4 J = 1.2 Hz), 7.98 (dd, 1H (6/9-ip) , 3 J = 5.3 Hz, 4 J = 1.2 Hz), 7.80 (dd, 1H (5/10-ip) , 3 J = 5.4 Hz, 3 J = 8.2 Hz), 7.78 (dd, 1H (5/10-ip) , 3 J = 5.3 Hz, 3 J = 8.4 Hz), 7.69 (d, 1H (6-bpy) , 3 J = 6.1 Hz), 7.67 (d, 1H (6-bpy) , 3 J = 6.1 Hz), 7.47 (dd, 1H (5-bpy) , 3 J = 5.7 Hz, 4 J = 2.7 Hz), 7.46 (dd, 1H (5-bpy) , 3 J = 5.5 Hz, 4 J = 2.4 Hz), 7.42 (dd, 1H (6’-bpy) , 3 J = 6.0 Hz), 7.41 (dd, 1H (6’-bpy) , 3 J = 6.0 Hz), 7.19 (d, 2H (5’-bpy) , 3 J = 6.0 Hz), 4.77 (q, 2H (CH2 ), 3 J = 7.2 Hz), 1.65 (t, 3H (CH3 ), 3 J = 7.2 Hz), 1.44 (s, 18H (CH3 -bpy)), 1.35 (s, 18H (CH ′ 3 -bpy)) ppm. 13 C-NMR (CD 3 CN, 100 M Hz): δ = 163.48 (2C (4-bpy) ), 163.31 (2C (4’-bpy) ), 157.89 (2C (2-bpy) ), 157.70 (2C (2’-bpy) ), 151.90 (2C (6-bpy) ), 151.83 (2C (6’-bpy) ), 150.95 (1C (6/9-ip) ), 150.41 (1C (6/9-ip) ), 146.99 (1C (7’/7”-ip) ), 146.73 (1C (7’/7”-ip) ), 146.03 (1C (2-ip) ), 131.20 (1C (4/11-ip) ), 130.59 (1C (4/11-ip) ), 127.52 (1C (3”/11’-ip) ), 127.11 (1C (5/10-ip) ), 126.06 (1C (5/10-ip) ), 125.97 (1C (3”/11’-ip) ), 125.62 (2C (5-bpy) ), 125.42 (2C (5’-bpy) ), 123.05 (2C (3’/11”-ip) ), 122.40 (2C (3-bpy) ), 122.29 (2C (3’-bpy) ), 43.77 (1C (CH2 )), 36.30 (2C (C-bpy) ), 36.19 (2C (C’-bpy) ), 30.54 (6C (CH3 -bpy)), 30.44 (6C (CH3 ’-bpy)), 15.99 (1C (CH3 )) ppm. MS (ESI): m/z = 1031.1 (100%, [M-PF 6 ] + ). Crystals suitable for X-ray diffraction were obtained from water/acetone. Crystal data for Ru(ebip): [C 51 H 60 N 8 Ru] 2+ [PF 6 ] − 2 × 3 CH 3COCH 3 , M r = 1350.31 g /mol, red-brown plate, size 0.05 × 0.05 × 0.03 mm 3 , triclinic, space group P1 (No. 2), a = 13.429(3), b = 14.934(3), c = 19.146(4) Å, α = 107.11(3), β = 96.87(3), γ = 96.58(3)°, V = 3407.7(12) Å 3 , T = -90(2)°C, Z = 2, ρ calcd. = 1.315 g /cm 3 , µ (Mo-Kα) = 3.56 cm -1 , F(000) = 1400, 20389 reflections in h(-17/17), k(-18/19), l(-23/21) measured in the range 3.96° ≤ Θ ≤ 27.49°, completeness Φ max = 92.4%, 14481 independent reflections, R int = 0.0396, 9212 reflections with F o > |235|
|6.2 Synthesis of the Metal Complexes| 4σ(F o ), 760 parameters, 0 restraints, R obs. = 0.0894, wR 2 obs. = 0.2298, R2 all = 0.1440, wR 2 all = 0.2703, GOOF = 1.029, largest difference peak and hole: 1.311 / -0.740 e /Å 3 . The data file TT3757 includes the full crystallographic data and can be obtained from Dr. Helmar Görls (IAAC, FSU-Jena). 6.2.24 [{Ru(tbbpy) 2 } 2 (µ-edip)][PF 6 ] 6 - Ru 2 (edip) The title compound was obtained, following method C1. 104 mg (141 µmol) of [Ru(tbbpy) 2 Cl 2 ] and 58 mg (70 µmol) of edip were reacted in the microwave for 300 minutes in 75 ml of ethanol/water. After cooling and removal of ethanol and a dark precipitate 12 equivalents of NH 4 PF 6 were added. The formed precipitate was collected and washed with water and dissolved in methylene chloride. After drying with Na 2 SO 4 and removal of the solvent a crude product was obtained. Purification was achieved ion exchange. The crude was dissolved in 10 ml of acetone/ethyl acetate (1:1) and was slowly added to a stirred solution of Bu 4 NCl in 40 ml of acetone/ethyl acetate (1:1). The formed precipitate was collected and washed with small amounts of acetone/ethyl acetate to yield pure [{Ru(tbbpy) 2 } 2 (µ-edip)]Cl 6 . After ion-back-exchange with NH 4 PF 6 and removal of water pure Ru 2 (edip) was obtained as red powder. The yield was 243 mg (85 µmol, 60%). N N N Ru 2+ N 9-ip 10-ip CH 11-ip CH 3 2 N N 6-ip 4-ip 5-ip N N 2-ip Ar Ru 2 (edip) N N 5-bpy 6-bpy N N N Ru 2+ N CH 3 -bpy 3-bpy 3'-bpy N 5'-bpy 6'-bpy N CH 3 '-bpy 1 H-NMR (CD 3 CN, 400 MHz): δ = 9.404 (dd, 2H (4-ip) , 3 J = 8.2 Hz, 4 J = 0.9 Hz), 8.994 (dd, 2H (11-ip) , 3 J = 8.2Hz, 4 J = 0.9 Hz), 8.552 (s, 2H (2-ip) ), 8.525 (m, 4H (3/3’-bpy) ), 8.485 (m, 4H (3’/3-bpy) ), 7.976 (dd, 2H (5-ip) , 3 J = 8.2 Hz, 3 J = 5.5 Hz), 7.934 (dd, 2H (9-ip) , 3 J = 8.2 Hz, 3 J = 5.5 Hz), 7,668 (d, 2H (6/6’-bpy) , 3 J = 7.7 Hz ), 7,660 (d, 2H (6/6’-bpy) , 3 J = 7.7 Hz ), 7.467 (m, 4H (5/5’-bpy) ), 7.415 (d, 2H (6’/6-bpy) , 3 J = 6.0 Hz), 7.408 (d, 2H (6’/6-bpy) , 3 J = 6.0 Hz), 7.219 (m, 4H (5’/5-bpy) ), 6.245 (d, 2H (CH2 -Ar), 2 J = 14.4 Hz), 6.210 (d, 2H (CH2 -Ar), 2 J = 14.4 Hz), 5.00 (q, 4H (CH2 -Al), 3 J = 7.0 Hz), 2.353 (s, 12H (CH3 -Ar)), 1.652 (t, 6H (CH3 -Al), 3 J = 7.3 Hz), 1.436 (s, 18H ( t Bu)), 1.431 (s, 18H ( t Bu)), 1.344 (s, 18H ( t Bu)), 1.342 (s, 18H ( t Bu)) ppm; 13 C-NMR (CD 3 CN, 100 MHz): δ = 163.05 (4C (4-bpy) ), 162.91 (4C (4’-bpy) ), 156.90 (4C (2-bpy) ), 156.82 (4C (2’-bpy) ), 152.98 (2C (6/9-ip) ), 151.28 (4C (3-bpy) ), 151.05 (4C (3’-bpy) ), 147.79 (2C (6’-ip) ), 147.70 (2C (6”-ip) ), 140.80 (2C (2-ip) ), |236|
- Page 1 and 2:
Development of Novel Catalysts for
- Page 3 and 4:
Diese Arbeit entstand auf Anregung
- Page 5 and 6:
Den Arbeisgruppen von Prof. Dr. U.
- Page 7 and 8:
Contents 1 Introduction 1 1.1 Fossi
- Page 9 and 10:
|Contents| 6 Experimental Section 1
- Page 12 and 13:
|1 Introduction| 1 Introduction The
- Page 14 and 15:
|1.1 Fossil Fuels and Nuclear Power
- Page 16 and 17:
|1.2 Renewable Fuels| of natural ga
- Page 18 and 19:
|1.3 Energy Transformation and Stor
- Page 20 and 21:
|1.4 Solar Energy Conversion| A fra
- Page 22 and 23:
|1.5 Photosynthesis| Figure 9: Mole
- Page 24 and 25:
|1.6 Photocatalyzed Reactions| 2 NA
- Page 26 and 27:
|1.6 Photocatalyzed Reactions| (1)
- Page 28 and 29:
|1.7 Mimicking Photosynthesis| So t
- Page 30 and 31:
|1.8 Formalisms of Photocatalytic S
- Page 32 and 33:
|1.9 Multicomponent Systems from Fu
- Page 34 and 35:
|1.9 Multicomponent Systems from Fu
- Page 36 and 37:
|1.9 Multicomponent Systems from Fu
- Page 38 and 39:
|1.9 Multicomponent Systems from Fu
- Page 40 and 41:
|1.9 Multicomponent Systems from Fu
- Page 42 and 43:
|1.9 Multicomponent Systems from Fu
- Page 44 and 45:
|1.9 Multicomponent Systems from Fu
- Page 46 and 47:
|1.9 Multicomponent Systems from Fu
- Page 48 and 49:
|1.9 Multicomponent Systems from Fu
- Page 50 and 51:
|1.9 Multicomponent Systems from Fu
- Page 52 and 53:
|1.10 Intramolecular Photoredoxcata
- Page 54 and 55:
|1.10 Intramolecular Photoredoxcata
- Page 56 and 57:
|1.10 Intramolecular Photoredoxcata
- Page 58 and 59:
|1.10 Intramolecular Photoredoxcata
- Page 60 and 61:
|1.10 Intramolecular Photoredoxcata
- Page 62 and 63:
|1.10 Intramolecular Photoredoxcata
- Page 64 and 65:
|2 Scope of the Thesis| motifs with
- Page 66 and 67:
|3.1 Brominated Phenanthrolines - A
- Page 68 and 69:
|3.1 Brominated Phenanthrolines - A
- Page 70 and 71:
|3.1 Brominated Phenanthrolines - A
- Page 72 and 73:
|3.1 Brominated Phenanthrolines - A
- Page 74 and 75:
|3.1 Brominated Phenanthrolines - A
- Page 76 and 77:
|3.1 Brominated Phenanthrolines - A
- Page 78 and 79:
|3.1 Brominated Phenanthrolines - A
- Page 80 and 81:
|3.1 Brominated Phenanthrolines - A
- Page 82 and 83:
|3.1 Brominated Phenanthrolines - A
- Page 84 and 85:
|3.1 Brominated Phenanthrolines - A
- Page 86 and 87:
|3.1 Brominated Phenanthrolines - A
- Page 88 and 89:
|3.1 Brominated Phenanthrolines - A
- Page 90 and 91:
|3.1 Brominated Phenanthrolines - A
- Page 92 and 93:
|3.1 Brominated Phenanthrolines - A
- Page 94 and 95:
|3.2 Bisphenanthroline: A Suitable
- Page 96 and 97:
|3.2 Bisphenanthroline: A Suitable
- Page 98 and 99:
|3.2 Bisphenanthroline: A Suitable
- Page 100 and 101:
|3.2 Bisphenanthroline: A Suitable
- Page 102 and 103:
|3.2 Bisphenanthroline: A Suitable
- Page 104 and 105:
f f f f f f |3.2 Bisphenanthroline:
- Page 106 and 107:
|3.2 Bisphenanthroline: A Suitable
- Page 108 and 109:
|3.2 Bisphenanthroline: A Suitable
- Page 110 and 111:
|3.2 Bisphenanthroline: A Suitable
- Page 112 and 113:
|3.2 Bisphenanthroline: A Suitable
- Page 114 and 115:
|3.2 Bisphenanthroline: A Suitable
- Page 116 and 117:
|3.2 Bisphenanthroline: A Suitable
- Page 118 and 119:
|3.2 Bisphenanthroline: A Suitable
- Page 120 and 121:
|3.2 Bisphenanthroline: A Suitable
- Page 122 and 123:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 124 and 125:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 126 and 127:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 128 and 129:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 130 and 131:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 132 and 133:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 134 and 135:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 136 and 137:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 138 and 139:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 140 and 141:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 142 and 143:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 144 and 145:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 146 and 147:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 148 and 149:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 150 and 151:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 152 and 153:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 154 and 155:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 156 and 157:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 158 and 159:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 160 and 161:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 162 and 163:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 164 and 165:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 166 and 167:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 168 and 169:
|3.3 NN-NHC-Ligand bbip: Toward Sec
- Page 170 and 171:
|3.4 Outlook, Exploratory Investiga
- Page 173 and 174:
|3.4 Outlook, Exploratory Investiga
- Page 175 and 176:
|3.4 Outlook, Exploratory Investiga
- Page 177 and 178:
|3.4 Outlook, Exploratory Investiga
- Page 179 and 180:
|3.4 Outlook, Exploratory Investiga
- Page 181 and 182:
|3.4 Outlook, Exploratory Investiga
- Page 183 and 184:
|3.4 Outlook, Exploratory Investiga
- Page 185 and 186:
|4 Summary| 4 Summary Against the b
- Page 187 and 188:
|4 Summary| The resulting complexes
- Page 189 and 190:
|4 Summary| 10 [TEA] + TEA visible
- Page 191 and 192:
|4 Summary| absorption between 430
- Page 193 and 194:
|4 Summary| additional NN- and NHC-
- Page 195 and 196: |5 Zusammenfassung| Die Herstellung
- Page 197 and 198: |5 Zusammenfassung| phenphen und Ru
- Page 199 and 200: |5 Zusammenfassung| Fragment tragen
- Page 201 and 202: |5 Zusammenfassung| [TEA] + TEA vis
- Page 203 and 204: |5 Zusammenfassung| In einer abschl
- Page 205 and 206: |6 Experimental Section| Spectroele
- Page 207 and 208: |6 Experimental Section| 1.3648 was
- Page 209 and 210: |6.1 Synthesis of the Organic Ligan
- Page 211 and 212: |6.1 Synthesis of the Organic Ligan
- Page 213 and 214: |6.1 Synthesis of the Organic Ligan
- Page 215 and 216: |6.1 Synthesis of the Organic Ligan
- Page 217 and 218: |6.1 Synthesis of the Organic Ligan
- Page 219 and 220: |6.1 Synthesis of the Organic Ligan
- Page 221 and 222: |6.1 Synthesis of the Organic Ligan
- Page 223 and 224: |6.2 Synthesis of the Metal Complex
- Page 225 and 226: |6.2 Synthesis of the Metal Complex
- Page 227 and 228: |6.2 Synthesis of the Metal Complex
- Page 229 and 230: |6.2 Synthesis of the Metal Complex
- Page 231 and 232: |6.2 Synthesis of the Metal Complex
- Page 233 and 234: |6.2 Synthesis of the Metal Complex
- Page 235 and 236: |6.2 Synthesis of the Metal Complex
- Page 237 and 238: |6.2 Synthesis of the Metal Complex
- Page 239 and 240: |6.2 Synthesis of the Metal Complex
- Page 241 and 242: |6.2 Synthesis of the Metal Complex
- Page 243 and 244: |6.2 Synthesis of the Metal Complex
- Page 245: |6.2 Synthesis of the Metal Complex
- Page 249 and 250: |7 Appendix| 7 Appendix dd ddd DEI
- Page 251 and 252: |7 Appendix| Ru(tpphz)Os [Ru(bpy) 2
- Page 253 and 254: |References| [26] M. Chanon, M. Sch
- Page 255 and 256: |References| [81] S. Tschierlei, M.
- Page 257 and 258: |References| [138] L. Pazderski, J.
- Page 259 and 260: |References| [194] C. Liu, J. Li, B
- Page 261: Selbstständigkeitserklärung: Ich