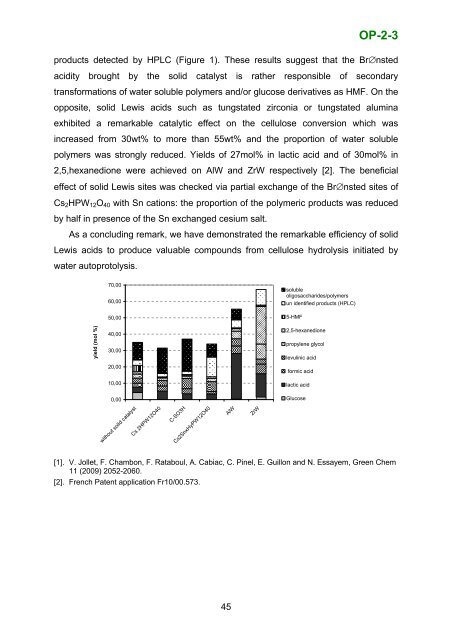
OP-2-3CELLULOSE HYDROTHERMAL CONVERSION PROMOTED BYHETEROGENEOUS BRØNSTED AND LEWIS ACIDS: REMARKABLEEFFICIENCY OF SOLID LEWIS ACIDS TO PRODUCE VALUABLEMOLECULESNadine Essayem 1 , Flora Chambon 1 , Franck Rataboul 1 , Amandine Cabiac 2 ,Catherine Pinel 1 , Emmanuelle Guillon 21 IRCELYON, Institut de recherche sur la catalyse et l’environnement de Lyon, CNRS,UMR 5256, 2 avenue Albert Einstein, F-69626 Villeurbanne, France2 IFP-Lyon, BP 3, F-69360 Solaize, FranceIntroductionOver the past few years, the transformation <strong>of</strong> cellulose into valuable organicchemicals has attracted a growing interest. Indeed, cellulose is a widely availableand non edible polysaccharide, which makes it a promising resource for theproduction <strong>of</strong> bio-products and bio-fuels. Liquid acids catalyzed hydrolysis <strong>of</strong>cellulose is a process known for more than 80 years. This process presentdrawbacks as a poor selectivity, problems <strong>of</strong> corrosion and production <strong>of</strong> largevolume <strong>of</strong> waste acidic water. Recent studies reported the feasibility <strong>of</strong> cellulosedepolymerization by mean <strong>of</strong> solid Br∅nsted acids. In particular, sulfonated carbonbased catalysts were reported to be active in the depolymerization <strong>of</strong> grindedcellulose. The present study reports investigations <strong>of</strong> hydrothermal conversion <strong>of</strong>crystalline cellulose, aimed to compare the efficiency <strong>of</strong> solid Br∅nsted acids to solidLewis acids.Results and DiscussionIn this study, the results <strong>of</strong> cellulose hydrolysis in hydrothermal conditions, thatmeans in the absence <strong>of</strong> solid catalysts were used as a reference experiment.Indeed, in a previous study, we have shown that crystalline cellulose may besignificantly depolymerised in hydrothermal conditions, depending on temperatureand duration conditions; at T=190°C, in the absence <strong>of</strong> solid catalyst, a conversion <strong>of</strong>35wt% was achieved after 24 h <strong>of</strong> reaction and glucose and HMF were detected onlyin poor yield [1]. It was shown by combining HPLC, TOC and MALDI TOFF analysisthat mainly water soluble oligosaccharides/polymers were formed (Figure 1). In thepresence <strong>of</strong> pure Br∅nsted catalysts such as an acidic cesium salt <strong>of</strong>tungstophosphoric acid or sulfonated carbon, the extent <strong>of</strong> cellulose depolymerizationwas not changed. However, the product distribution was deeply modified; theproportion <strong>of</strong> water soluble polymers was improved at the expense <strong>of</strong> the pool <strong>of</strong>44
OP-2-3products detected by HPLC (Figure 1). These results suggest that the Br∅nstedacidity brought by the solid catalyst is rather responsible <strong>of</strong> secondarytransformations <strong>of</strong> water soluble polymers and/or glucose derivatives as HMF. On theopposite, solid Lewis acids such as tungstated zirconia or tungstated aluminaexhibited a remarkable catalytic effect on the cellulose conversion which wasincreased from 30wt% to more than 55wt% and the proportion <strong>of</strong> water solublepolymers was strongly reduced. Yields <strong>of</strong> 27mol% in lactic acid and <strong>of</strong> 30mol% in2,5,hexanedione were achieved on AlW and ZrW respectively [2]. The beneficialeffect <strong>of</strong> solid Lewis sites was checked via partial exchange <strong>of</strong> the Br∅nsted sites <strong>of</strong>Cs 2 HPW 12 O 40 with Sn cations: the proportion <strong>of</strong> the polymeric products was reducedby half in presence <strong>of</strong> the Sn exchanged cesium salt.As a concluding remark, we have demonstrated the remarkable efficiency <strong>of</strong> solidLewis acids to produce valuable compounds from cellulose hydrolysis initiated bywater autoprotolysis.yield (mol %)70,0060,0050,0040,0030,0020,0010,000,00solubleoligosaccharides/polymersun identified products (HPLC)5-HMF2,5-hexanedionepropylene glycollevulinic acidformic acidlactic acidGlucosewithout solid catalystCs 2HPW12O40C-SO3HCs2SnxHyPW12O40AlWZrW[1]. V. Jollet, F. Chambon, F. Rataboul, A. Cabiac, C. Pinel, E. Guillon and N. Essayem, Green Chem11 (2009) 2052-2060.[2]. French Patent application Fr10/00.573.45