of the Max - MDC
of the Max - MDC
of the Max - MDC
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Molecular Mechanisms<br />
in Embryonic Forebrain<br />
Development<br />
Annette Hammes<br />
(Helmholtz Fellow)<br />
Defects in early forebrain development can lead to a fatal disorder, defined as holoprosencephaly<br />
(HPE), in which <strong>the</strong> cerebral hemispheres fail to separate along <strong>the</strong> midline. HPE is <strong>the</strong> most<br />
common brain malformation in human embryos. Megalin, a member <strong>of</strong> <strong>the</strong> low density lipoprotein<br />
receptor gene family, is a candidate gene for holoprosencephaly and recent studies have indeed<br />
identified megalin as a novel genetic factor, that affects dorsal midline separation and ventral neuronal<br />
cell fate specification in <strong>the</strong> developing forebrain. Our group is studying <strong>the</strong> mechanisms whereby<br />
megalin regulates molecular pathways important for early embryonic forebrain development.<br />
The role <strong>of</strong> megalin/LRP2 in forebrain development:<br />
Identification <strong>of</strong> defects in <strong>the</strong> molecular<br />
pathways underlying holoprosencephaly<br />
(collaboration with Thomas Willnow)<br />
Megalin/Lrp2 is expressed in <strong>the</strong> neuroepi<strong>the</strong>lium and <strong>the</strong><br />
yolk sac <strong>of</strong> <strong>the</strong> early embryo. Loss <strong>of</strong> megalin expression in<br />
mutant mice results in forebrain defects and in holoprosencephaly<br />
(HPE), indicating an essential function in brain<br />
development.<br />
In detailed studies we could show that megalin-deficiency in<br />
<strong>the</strong> neuroepi<strong>the</strong>lium leads to changes in <strong>the</strong> expression and<br />
activity <strong>of</strong> <strong>the</strong> key morphogens bone morphogenic protein 4<br />
(BMP4), sonic hedgehog (SHH), and fibroblast growth factor<br />
8 (FGF8) that regulate dorso-ventral patterning <strong>of</strong> <strong>the</strong><br />
neural tube. An intricate balance <strong>of</strong> dorsal (BMPs) and ventral<br />
(SHH) morphogen gradients is crucial for specifying<br />
dorso-ventral forebrain patterning. We could show that<br />
Bmp4 expression and signalling in <strong>the</strong> rostral and dorsal<br />
neuroepi<strong>the</strong>lium is increased in megalin -/- embryos, Shh<br />
expression is lost in <strong>the</strong> ventral telencephalon, and Fgf8 is<br />
aberrantly expressed in <strong>the</strong> rostral midline <strong>of</strong> <strong>the</strong> forebrain.<br />
As a consequence <strong>of</strong> absent SHH activity, ventrally derived<br />
oligodendroglial and interneuronal cell populations are lost<br />
in <strong>the</strong> forebrain <strong>of</strong> megalin -/- embryos.<br />
These findings identified megalin as a novel genetic factor<br />
that affects ventral neuronal cell fate specification in <strong>the</strong><br />
forebrain and show that lack <strong>of</strong> megalin is central to <strong>the</strong><br />
axial patterning defects and to <strong>the</strong> pathophysiology <strong>of</strong><br />
holoprosencephaly.<br />
Identification <strong>of</strong> <strong>the</strong> mechanisms underlying megalindependent<br />
holoprosencephaly and identification <strong>of</strong> novel<br />
candidate genes for HPE will substantially extend our understanding<br />
<strong>of</strong> neurodevelopmental disorders such as holoprosencephaly<br />
and <strong>the</strong> signals that are crucial for normal<br />
forebrain development.<br />
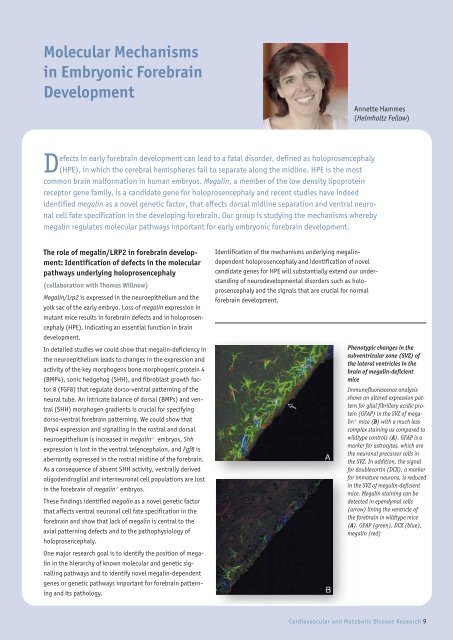
Phenotypic changes in <strong>the</strong><br />
subventricular zone (SVZ) <strong>of</strong><br />
<strong>the</strong> lateral ventricles in <strong>the</strong><br />
brain <strong>of</strong> megalin-deficient<br />
mice<br />
Immun<strong>of</strong>luorescence analysis<br />
shows an altered expression pattern<br />
for glial fibrillary acidic protein<br />
(GFAP) in <strong>the</strong> SVZ <strong>of</strong> megalin<br />
-/- mice (B) with a much less<br />
complex staining as compared to<br />
wildtype controls (A). GFAP is a<br />
marker for astrocytes, which are<br />
<strong>the</strong> neuronal precursor cells in<br />
<strong>the</strong> SVZ. In addition, <strong>the</strong> signal<br />
for doublecortin (DCX), a marker<br />
for immature neurons, is reduced<br />
in <strong>the</strong> SVZ <strong>of</strong> megalin-deficient<br />
mice. Megalin staining can be<br />
detected in ependymal cells<br />
(arrow) lining <strong>the</strong> ventricle <strong>of</strong><br />
<strong>the</strong> forebrain in wildtype mice<br />
(A). GFAP (green), DCX (blue),<br />
megalin (red)<br />
One major research goal is to identify <strong>the</strong> position <strong>of</strong> megalin<br />
in <strong>the</strong> hierarchy <strong>of</strong> known molecular and genetic signalling<br />
pathways and to identify novel megalin-dependent<br />
genes or genetic pathways important for forebrain patterning<br />
and its pathology.<br />
Cardiovascular and Metabolic Disease Research 9