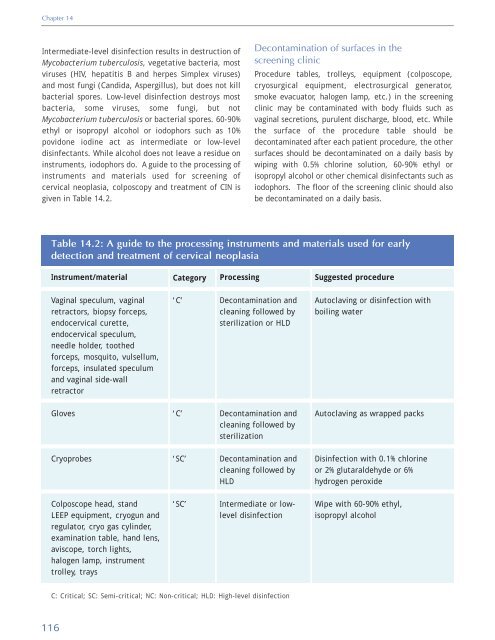
Chapter 14 Intermediate-level disinfection results in destruction <strong>of</strong> Mycobacterium tuberculosis, vegetative bacteria, most viruses (HIV, hepatitis B <strong>and</strong> herpes Simplex viruses) <strong>and</strong> most fungi (C<strong>and</strong>ida, Aspergillus), but does not kill bacterial spores. Low-level disinfection destroys most bacteria, some viruses, some fungi, but not Mycobacterium tuberculosis or bacterial spores. 60-90% ethyl or isopropyl alcohol or iodophors such as 10% povidone iodine act as intermediate or low-level disinfectants. While alcohol does not leave a residue on instruments, iodophors do. A guide to the processing <strong>of</strong> instruments <strong>and</strong> materials used for screening <strong>of</strong> cervical neoplasia, colposcopy <strong>and</strong> treatment <strong>of</strong> CIN is given in Table 14.2. Decontamination <strong>of</strong> surfaces in the screening clinic Procedure tables, trolleys, equipment (colposcope, cryosurgical equipment, electrosurgical generator, smoke evacuator, halogen lamp, etc.) in the screening clinic may be contaminated with body fluids such as vaginal secretions, purulent discharge, blood, etc. While the surface <strong>of</strong> the procedure table should be decontaminated after each patient procedure, the other surfaces should be decontaminated on a daily basis by wiping with 0.5% chlorine solution, 60-90% ethyl or isopropyl alcohol or other chemical disinfectants such as iodophors. The floor <strong>of</strong> the screening clinic should also be decontaminated on a daily basis. Table 14.2: A guide to the processing instruments <strong>and</strong> materials used for early detection <strong>and</strong> treatment <strong>of</strong> cervical neoplasia Instrument/material Category Processing Suggested procedure Vaginal speculum, vaginal retractors, biopsy forceps, endocervical curette, endocervical speculum, needle holder, toothed forceps, mosquito, vulsellum, forceps, insulated speculum <strong>and</strong> vaginal side-wall retractor ‘C’ Decontamination <strong>and</strong> cleaning followed by sterilization or HLD Autoclaving or disinfection with boiling water Gloves ‘C’ Decontamination <strong>and</strong> cleaning followed by sterilization Autoclaving as wrapped packs Cryoprobes ‘SC’ Decontamination <strong>and</strong> cleaning followed by HLD Disinfection with 0.1% chlorine or 2% glutaraldehyde or 6% hydrogen peroxide Colposcope head, st<strong>and</strong> LEEP equipment, cryogun <strong>and</strong> regulator, cryo gas cylinder, examination table, h<strong>and</strong> lens, aviscope, torch lights, halogen lamp, instrument trolley, trays ‘SC’ Intermediate or lowlevel disinfection Wipe with 60-90% ethyl, isopropyl alcohol C: Critical; SC: Semi-critical; NC: Non-critical; HLD: High-level disinfection 116
References Herrero, R. (1997) Prevalence surveys <strong>of</strong> HPV infection in high- <strong>and</strong> low-incidence areas for cervical cancer. In: International Agency for Research on Cancer-Biennial report 1996/1997. Lyon, France: IARC press, 68-69. Hinselmann, H. (1925) Verbesserung der Inspektionsmoglichkeiten von Vulva, Vagina <strong>and</strong> Portio. Munchner Med Wochenschr, 72, 1733-42. Ho, G.Y, Bierman, R., Beardsley, L., Chang, C.J., & Burk, R.D. (1998) Natural history <strong>of</strong> cervicovaginal papillomavirus infection in young women. N. Engl. J. Med., 338, 423-428. Ho, G.Y., Burk, R.D., Klein, S., Kadish, A.S., Chang, C.J., Palan, P., Basu, J., Tachezy, R., Lewis, R., & Romney, S. (1995) Persistent genital human papillomavirus infection as a risk factor for persistent cervical dysplasia. J. Natl. Cancer Inst., 87, 1365-1371. Holowaty, P., Miller, A.B., & Rohan, T. (1999) Natural history <strong>of</strong> dysplasia <strong>of</strong> the uterine cervix. J. Natl. Cancer Inst., 91, 252-258. Howard, M., Sellors, J., & Lytwyn, A. (2002) <strong>Cervical</strong> intraepithelial neoplasia in women presenting with external genital warts. CMAJ, 166, 598-599. IARC Working Group. (1995) Human papillomaviruses. IARC Monograph on the evaluation <strong>of</strong> carcinogenic risks to humans. Vol. 65. Lyon, France: International Agency for Research on Cancer. Koutsky, L.A., Holmes, K.K., Critchlow, C.W., Stevens, C.E., Paavonen, J., Beckmann, A.M., DeRouen, T.A., Galloway, D.A., Vernon, D., & Kiviat, N.B. (1992) A cohort study <strong>of</strong> the risk <strong>of</strong> cervical intraepithelial neoplasia grade 2 or 3 in relation to papillomavirus infection. N. Engl. J. Med., 327, 1272-1278 Kosary, C.L. (1994) FIGO stage, histology,histologic grade, age <strong>and</strong> race as prognostic factors in determining survival for cancers <strong>of</strong> the female gynecological system: an analysis <strong>of</strong> 1973-87 SEER cases <strong>of</strong> cancers <strong>of</strong> the endometrium, cervix, ovary, vulva, <strong>and</strong> vagina. Semin. Surg. Oncol., 10, 31-46. Kurman, R.J., Malkasian, G.D. Jr, Sedlis, A., & Solomon, D. (1991) From Papanicolaou to Bethesda: the rationale for a new cervical cytologic classification. Obstet. Gynecol., 77, 779-782. Liaw, K.L., Glass, A.G., Manos, M.M., Greer, C.E., Scott, D.R., Sherman, M., Burk, R.D., Kurman, R.J., Wacholder, S., Rush, B.B., Cadell, D.M., Lawlerm, P., Tabor, D., & Schiffman, M. (1999) Detection <strong>of</strong> human papillomavirus DNA in cytologically normal women <strong>and</strong> subsequent cervical squamous intraepithelial lesions. J. Nat.l Cancer Inst., 91, 954-960. Liaw, K.L., Hildesheim, A., Burk, R.D., Gravitt, P., Wacholder, S., Manos, M.M., Scott, D.R., Sherman, M.E., Kurman, R.J., Glass, A.G., Anderson, S.M., & Schiffman, M. (2001) A prospective study <strong>of</strong> human papillomavirus (HPV) type 16 DNA detection by polymerase chain reaction <strong>and</strong> its association with acquisition <strong>and</strong> persistence <strong>of</strong> other HPV types. J. Infect. Dis., 183, 8-15. Martin-Hirsch, P.L., Paraskevaidis, E., & Kitchener, H. (2000) Surgery for cervical intraepithelial neoplasia. Cochrane Database Syst. Rev., 2, CD001318. McIndoe, W.A., McLean, M.R., Jones, R.W., & Mullins, P.R. (1984) The invasive potential <strong>of</strong> carcinoma in situ <strong>of</strong> the cervix. Obstet. Gynecol., 64, 451-458. Melnikow, J., Nuovo, J., Willan, A.R., Chan, B.K., & Howell, L.P. (1998) Natural history <strong>of</strong> cervical squamous intraepithelial lesions: a meta-analysis. Obstet Gynecol., 92(4 Pt 2), 727-735. Mitchell, M.F., Hittelman, W.N., Hong, W.K., Lotan, R., & Schottenfeld, D. (1994) The natural history <strong>of</strong> cervical intraepithelial neoplasia: an argument for intermediate endpoint biomarkers. Cancer Epidemiol. Biomarkers Prev., 3, 619-626. Mitchell, M.F., Schottenfeld, D., Tortolero-Luna, G., Cantor, S.B., & Richards-Kortum, R. (1998) <strong>Colposcopy</strong> for the diagnosis <strong>of</strong> squamous intraepithelial lesions: Metaanalysis. Obstet. Gynaecol., 91, 626-31. Moscicki, A.B., Hills, N., Shiboski, S., Powell, K., Jay, N., Hanson, E., Miller, S., Clayton, L., Farhat, S., Broering, J., Darragh, T., & Palefsky, J. (2001) Risks for incident human papillomavirus infection <strong>and</strong> low grade squamous intraepithelial lesion development in young females. JAMA, 285, 2995-3002. Moscicki, A.B., Shiboski, S., Broering, J., Powell, K., Clayton, L., Jay, N., Darragh, T.M., Brescia, R., Kanowitz, S., Miller, S.B., Stone, J., Hanson, E., & Palefsky, J. (1998) The natural history <strong>of</strong> human papillomavirus infection as measured by repeated DNA testing in adolescent <strong>and</strong> young women. J. Pediatr., 132, 277-284. National Cancer Institute Workshop. (1989) The 1988 Bethesda System for reporting cervical/vaginal cytologic diagnoses. JAMA, 262, 931-934. National Cancer Institute Workshop. (1993) The Bethesda System for reporting cervical/vaginal cytologic diagnoses: revised after the second National Cancer Institute Workshop, April 29-30, 1991. Acta Cytol., 37, 115-124. Nuovo, J., Melnikov, J., Willan, A.R., & Chan, N.K. (2000) <strong>Treatment</strong> outcomes for squamous intraepithelial lesions. Int. J. Gynaecol. Obstet., 68, 25-33. Östor, A.G. (1993) Natural history <strong>of</strong> cervical intraepithelial neoplasia : a critical review. Int J Gynecol. Pathol., 12, 186-192. Popkin, D.R. (1995) Pitfalls in colposcopy. In: Cecil Wright V, Likrish GM, & Shier RM (Eds). Basic <strong>and</strong> advanced colposcopy. Part one: A practical h<strong>and</strong>book for diagnosis. Second edition. Houston. Biomedical Communications. Reagan, J.W., Seidermann, I.L., & Saracusa, Y. (1953) The cellular morphology <strong>of</strong> carcinoma in situ <strong>and</strong> dysplasia or atypical hyperplasia <strong>of</strong> the uterine cervix. Cancer, 6, 224- 235. Reid, R. (1987) A rapid method for improving colposcopic accuracy. Colposcopic <strong>and</strong> Gynaecologic Laser Surgery, 3, 139–146 . 118