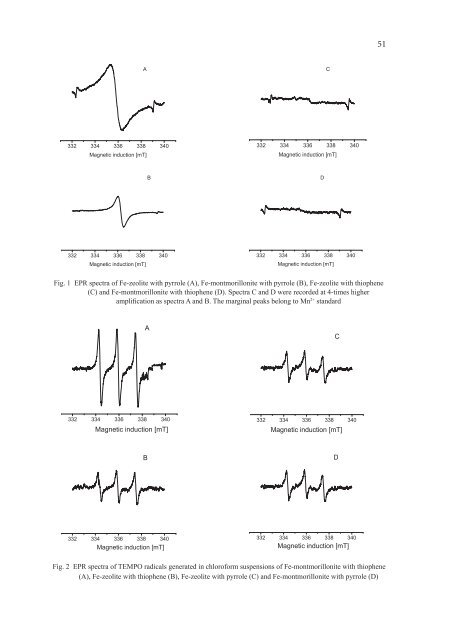
51AC332 334 336 338 340Magnetic induction [mT]332 334 336 338 340Magnetic induction [mT]BD332 334 336 338 340Magnetic induction [mT]332 334 336 338 340Magnetic induction [mT]Fig. 1 EPR spectra of Fe-zeolite with pyrrole (A), Fe-montmorillonite with pyrrole (B), Fe-zeolite with thiophene(C) and Fe-montmorillonite with thiophene (D). Spectra C and D were recorded at 4-times higheramplification as spectra A and B. The marginal peaks belong to Mn 2+ standardAC332 334 336 338 340Magnetic induction [mT]332 334 336 338 340Magnetic induction [mT]BD332 334 336 338 340Magnetic induction [mT]332 334 336 338 340Magnetic induction [mT]Fig. 2 EPR spectra of TEMPO radicals generated in chloroform suspensions of Fe-montmorillonite with thiophene(A), Fe-zeolite with thiophene (B), Fe-zeolite with pyrrole (C) and Fe-montmorillonite with pyrrole (D)
52absorbance [a.u.]1,000,750,500,250,00200 250 300 350Fig 3 Absorption spectra of 4-chlorophenol solutions(10 –4 mol dm –3 ) after 48 hours of irradiation byvisible light (black line); in the presence of Fe--zeolite with thiophene (blue line); Fe-zeolitewith pyrrole (green line); Fe-montmorillonitewith thiophene (violet line) and Fe-montmorillonitewith pyrrole (red line)We suppose that the first step of 4-chlorophenoldecomposition is the formation of the singletoxygen in 4-chlorophenol water solutions with themodified zeolites and montmorillonites. 1 O 2thencan enter reactions with molecular oxygen whichis present in water and therefore other reactive oxygenspecies (hydroxyl and superoxide anion radicals)are produced. These radicals can react with4-chlorophenol and cause its decomposition. Thiseffect of modified natural zeolites and montmorillonitescan be exploited to decontamination ofthe environment polluted by chlorophenol compounds.AcknowledgementsThis work was financially supported by theScientific Grant Agency VEGA of the Ministryof Education of the Slovak Republic, grant No.1/3411/06, No. 2/6180/6 and 1/4453/07.REFERENCESwave length [nm]1. HUI, R. E., NETA, P.: Reactive oxygen species. In: Gilbert,D. L. Reactive oxygen species in biological system.Kluwer Academic Publishers, 1999, p. 33–63.2. HALLIWELL, B., GUTTERIDGE, J. M. C.: Freeradicals in biology and medicine. 2nd ed. Oxford ,Claredon Press, 1989, p. 335.3. SCAIANO, J. C., REDMOND, R. W., MEHTA, B.,ARNASON, J. T.: Efficiency of the photoprogressleading to singlet oxygen ( 1 ∆ g) generation by α-terthienyl:Optical absorption, optoacustic calorimetryand infrared luminiscence studies. In: Photochem.Photobiol. 1990, <strong>vo</strong>l. 52, p. 655–659.4. BOCH, R., MEHTA, B., CONNOLLY T., DURST,T., ARNASON, J. T., REDMOND, R. W., SCAIA-NO, J. C.: Singlet oxygen photosensitizing propertiesof bithiophene and terthiophene derivatives. In:J. Photochem. Photobiol. A Chem. 1996, <strong>vo</strong>l. 93,p. 39–47.5. JESENÁK, K.: Environmentálna anorganická chémia.Bratislava: Univerzita Komenského v Bratislave, p. 68.ISBN 80-223-2072-2, 2005. (in Slovak).6. CASPAR, J. V., RAMAMURTHY, V., CORBIN, D.R.: Preparation and spectroscopic characterizationof polarons and bipolarons of thiophene oligomerswithin the channels of pentasil zeolites: The e<strong>vo</strong>lutionof organic radical ions into conducting polymers.In: J. Am. Chem. Soc., 1991, <strong>vo</strong>l: 113, p. 600–610.7. ČÍK, G., HUBINOVÁ, M., ŠERŠEŇ, F. KRIŠTÍN,J., ANTOŠOVÁ, M.: Photocatalytic degradation of4-chlorophenol by ·OH radicals generated by thiopheneoligomers incorporated in ZSM-5 zeoliteschannels. In: Coll. Czech. Chem. Comm., 2003, <strong>vo</strong>l:68, p. 2219–2230.8. BALLAV, S., BISWAS, M.: A conducting nanocompositevia intercalative polymerisation of thiophenein montmorillonite clay. In: Synt. Met., 2004, <strong>vo</strong>l:142, p. 309–315.9. KOZÁČ, J., OČENÁŠ, D., RUSŇÁK, D., HOP-PAN, J.: Mineralogy, characcteristic properties anutilization possibilities of zeolitic tuffite from NižnýHrabovec (East Slovakia). In: Mineralia Slov., 1982,<strong>vo</strong>l: 14, p. 263–275.10. JESENÁK, K.: Laboratory Device for SedimentationSeparation of Powders. In: Ceramics, 1994, <strong>vo</strong>l:38, p. 35–36.11. JESENÁK, K., HLAVATÝ, V.: Laboratory device forsedimentation of fine bentonite fractions. In: ScriptaFac. Sci. Nat. Univ. Masaryk Brun., 2000, <strong>vo</strong>l: 28–29,p. 33–36.12. ŠERŠEŇ, F., SIGMUNDOVÁ, I., CIGÁŇ, M.: Styrylbenzothiazolderivatives and their ammonium saltsas singlet oxygen generators. Chem. Listy, 2006, <strong>vo</strong>l:100, p. 995–996. (in Slovak).
- Page 2 and 3: Acta FacultatisEcologiaeJournal of
- Page 4 and 5: OBSAH / CONTENTSISOL M., MICHALÍKO
- Page 6: 5ACTA FACULTATIS ECOLOGIAE, 16: Sup
- Page 12: 11ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 18 and 19: 17ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 20 and 21: 19are lower in ill patients compare
- Page 22: 21are considered as the most accura
- Page 25 and 26: 24- multimode cavities are usually
- Page 27 and 28: 26the load during its exposure to f
- Page 29 and 30: 28Tradescantia paludosa 02 test and
- Page 31 and 32: 30Tab. 5: Results of positive contr
- Page 34 and 35: 33ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 36 and 37: 35DISCUSSIONThe ionising radiation
- Page 38 and 39: 37ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 40 and 41: 39222Rn is produced by radioactive
- Page 42 and 43: 41180160140this reason we also pick
- Page 44: 435001450400350hKz0,8h [m]300250200
- Page 47 and 48: 46deposit is that stripped in off-l
- Page 49 and 50: 48TruenessTrueness was determined i
- Page 51: 50MATERIAL AND METHODSChloroform (p
- Page 55 and 56: 54Tab. 1: Rrequirements determinati
- Page 57 and 58: 56Methods of VOC testing were set a
- Page 60 and 61: 59Tab. 6: ContinuedSamples withsurf
- Page 62 and 63: 61ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 64 and 65: 63One of the possible explanations
- Page 66 and 67: 65Ai - Ai-1 [Bq.m -3 ]86420-2-4-6-8
- Page 68 and 69: 67ACTA FACULTATIS ECOLOGIAE, 15: Su
- Page 70 and 71: 69BiodegradabilityThe great variety
- Page 72 and 73: 71degradation starts of late days,
- Page 74 and 75: 73Fig. 4 Treated (after 28 days of
- Page 76: 75parameters of the cutting process
- Page 80 and 81: 79Fraction: D (residual rest) prese
- Page 82: 81was not confirmed. Maximum of mer
- Page 85 and 86: 84Fig. 1 Schematic diagram of atomi
- Page 87 and 88: 86Alpha spectrometryAlpha spectrome
- Page 89 and 90: 8880007000y = 6622xR 2 = 0.939SIMS
- Page 92 and 93: 91ACTA FACULTATIS ECOLOGIAE, 16: Su
- Page 94 and 95: 93Gemer according to the German mod
- Page 96 and 97: 95Tab. 1 Results of the chemical an
- Page 98 and 99: 97Continuation of Tab. 2 Results of
- Page 100 and 101: 99Vlčia Dolina and from the reserv
- Page 102 and 103:
101ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 104 and 105:
103mg.dm -3mg.dm -35,004,003,002,00
- Page 106 and 107:
105year and the average value repre
- Page 108 and 109:
107ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 110 and 111:
109Sample site 1 Sample site 2 Samp
- Page 112 and 113:
111As for the sampling time (Fig. 5
- Page 114 and 115:
113ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 116 and 117:
115Typha latifolia, Carex sp., Scir
- Page 118 and 119:
117conditions for decomposition of
- Page 120 and 121:
119ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 122 and 123:
121from the background (derived fro
- Page 124 and 125:
12311. PETROVSKÝ, E., ELWOOD, B.:
- Page 126 and 127:
125ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 128 and 129:
1272.52.0Correlation coefficient 0,
- Page 130 and 131:
129ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 132 and 133:
131RESULTS AND DISCUSSIONTable 2 gi
- Page 134 and 135:
133ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 136 and 137:
135V-1 BOREHOLEThe courses of 222 R
- Page 138 and 139:
137AV-2 (40m) 2006A ( 222 Rn) [kBq/
- Page 140 and 141:
139soaks into the soil, another par
- Page 142 and 143:
141ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 144 and 145:
143Fig. 2 The continuous monitoring
- Page 146 and 147:
145Indoor radon activity concentrat
- Page 148 and 149:
147ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 150 and 151:
149Fig. 1 Podlipa dump-fieldCanada)
- Page 152 and 153:
151concentrations of Fe. Cu. Cd. Ni
- Page 154 and 155:
153DUMP-FIELDREFERENCE SITEppm15001
- Page 156 and 157:
155Fig. 5 Compression of wood forma
- Page 158 and 159:
157decrease in the following order:
- Page 160 and 161:
159ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 162 and 163:
161SPECIFIC EXAMPLES OFFACTORS THAT
- Page 164 and 165:
163ACTA FACULTATIS ECOLOGIAE, 16: S
- Page 166 and 167:
165The methods developed to incorpo
- Page 168 and 169:
167The effects of wind on ozone con
- Page 170 and 171:
169Fig. 6 Mean total and stomatal f
- Page 172 and 173:
171transport modelling in North Ame
- Page 175:
Acta Facultatis Ecologiae, Volume 1