View PDF Version - RePub - Erasmus Universiteit Rotterdam
View PDF Version - RePub - Erasmus Universiteit Rotterdam
View PDF Version - RePub - Erasmus Universiteit Rotterdam
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Chapter 4.2<br />
198<br />
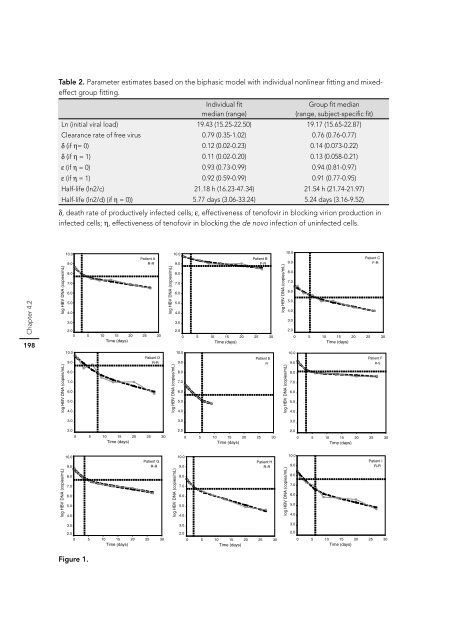
Table 2. Parameter estimates based on the biphasic model with individual nonlinear fi tting and mixedeffect<br />
group fi tting.<br />
Individual fi t<br />
median (range)<br />
Group fi t median<br />
(range, subject-specifi c fi t)<br />
Ln (initial viral load) 19.43 (15.25-22.50) 19.17 (15.65-22.87)<br />
Clearance rate of free virus 0.79 (0.35-1.02) 0.76 (0.76-0.77)<br />
δ (if η= 0) 0.12 (0.02-0.23) 0.14 (0.073-0.22)<br />
δ (if η = 1) 0.11 (0.02-0.20) 0.13 (0.058-0.21)<br />
ε (if η = 0) 0.93 (0.73-0.99) 0.94 (0.81-0.97)<br />
ε (if η = 1) 0.92 (0.59-0.99) 0.91 (0.77-0.95)<br />
Half-life (ln2/c) 21.18 h (16.23-47.34) 21.54 h (21.74-21.97)<br />
Half-life (ln2/d) (if η = 0)) 5.77 days (3.06-33.24) 5.24 days (3.16-9.52)<br />
δ, death rate of productively infected cells; ε, effectiveness of tenofovir in blocking virion production in<br />
infected cells; η, effectiveness of tenofovir in blocking the de novo infection of uninfected cells.<br />
log HBV DNA (copies/mL)<br />
log HBV DNA (copies/mL)<br />
log HBV DNA (copies/mL)<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
5<br />
5<br />
5<br />
Figure 1.<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
Patient A<br />
R-R<br />
25<br />
25<br />
30<br />
Patient D<br />
R-R<br />
Patient G<br />
R-R<br />
25<br />
30<br />
30<br />
log HBV DNA (copies/mL)<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
log HBV DNA (copies/mL)<br />
3.0<br />
2.0<br />
0<br />
log HBV DNA (copies/mL)<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
5<br />
5<br />
5<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
Patient B<br />
F-R<br />
25<br />
Patient E<br />
R<br />
25<br />
30<br />
Patient H<br />
R-R<br />
25<br />
30<br />
30<br />
log HBV DNA (copies/mL)<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
log HBV DNA (copies/mL)<br />
log HBV DNA (copies/mL)<br />
2.0<br />
0<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
10.0<br />
9.0<br />
8.0<br />
7.0<br />
6.0<br />
5.0<br />
4.0<br />
3.0<br />
2.0<br />
0<br />
5<br />
5<br />
5<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
10 15<br />
Time (days)<br />
20<br />
Patient C<br />
F-R<br />
25<br />
Patient F<br />
R-S<br />
25<br />
Patient I<br />
R-R<br />
25<br />
30<br />
30<br />
30