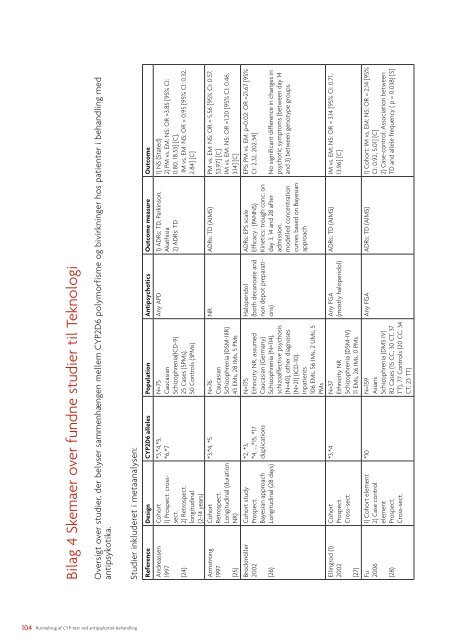
Bilag 4 Skemaer over fundne studier til TeknologiOversigt over studier, der belyser sammenhængen mellem <strong>CYP</strong>2D6 polymorfisme og bivirkninger hos patienter i <strong>behandling</strong> medantipsykotika.Studier inkluderet i metaanalysen:Reference Design <strong>CYP</strong>2D6 alleles Population Antipsychotics Outcome measure OutcomeAndreassen1997(24)Armstrong1997(25)Brockmöller2002(26)Ellingrod (1)2002(27)Fu2006(28)Cohort1) Prospect. crosssect.2) Retrospect.longitudinal(2-14 years)CohortRetrospect.Longitudinal (durationNR)Cohort studyProspect.Bayesian approachLongitudinal (28 days)CohortProspect.Cross-sect.1) Cohort element2) Case controlelementProspect.Cross-sect.*3,*4,*5,*6,*7N=75CaucasianSchizophrenia(ICD-9)25 Cases (5PMs),50 Controls (3PMs)*3,*4, *5 N=76CaucasianSchizophrenia (DSM-IIIR)43 EMs, 28 IMs, 5 PMs*2, *3,*4, …,*15, *17duplicationsN=175Ethnicity NR, assumedCaucasian (Germany)Schizophrenia (N=114),schizo<strong>af</strong>fective psychosis(N=40), other diagnoses(N=21) (ICD-10).Inpatients106 EMs, 56 IMs, 2 UMs, 5PMs*3,*4 N=37Ethnicity NRSchizophrenia (DSM-IV)11 EMs, 26 IMs, 0 PMs*10 N=159AsiansSchizophrenia (DMS IV)82 Cases (15 CC, 30 CT, 37TT), 77 Controls (20 CC, 34CT, 23 TT)Any APD 1) ADRs: TD, Parkinson,Akathisia2) ADRs: TD1) NS (Stated)2) PM vs. EM: NS: OR =3.85 [95% CI:0.80, 18.53] [C],IM vs. EM: NS: OR = 0.95 [95% CI: 0.32,2.84] [C]NR ADRs: TD (AIMS) PM vs. EM: NS: OR = 5.56 [95% CI: 0.57,53.97] [C]IM vs. EM: NS: OR =1.20 [95% CI: 0.46,3.14] [C]Haloperidol(both decanoate andnon depot preparations)Any FGA(mostly haloperidol)ADRs: EPS scaleEfficacy : (PANNS)Kinetics: trough conc. onday 3, 14 and 28 <strong>af</strong>teradmission,modelled concentrationcurves based on BayesianapproachEPS: PM vs. EM: p=0.02: OR =21.67 [95%CI: 2.32, 202.34]No significant difference in changes inpsychotic symptoms (between day 14and 3) between genotype groups.ADRs: TD (AIMS) IM vs. EM: NS: OR = 3.14 [95% CI: 0.71,13.96] [C]Any FGA ADRs: TD (AIMS) 1) Cohort: IM vs. EM: NS: OR = 2.14 [95%CI: 0.92, 5.01] [C]2) Case-control: Association betweenTD and allele frequency ( p = 0.038) [S]104 <strong>Rutinebrug</strong> <strong>af</strong> <strong>CYP</strong>-<strong>test</strong> <strong>ved</strong> <strong>antipsykotisk</strong> <strong>behandling</strong>
Reference Design <strong>CYP</strong>2D6 alleles Population Antipsychotics Outcome measure OutcomeInada2003(29)Jaanson2002(30)Kapitany1998(31)Lam2001(32)Liou2004(33)Lohmann2003(34)CohortProspectCross sect.Cohort studyProspect.Longitudinal(8 weeks)CohortProspect.Cross sect.1) Cohort2) Case controlMatched gender andageProspect.Cross-sect.CohortProspectCross-sect.1)Cohort2)Case controlRetrospect.Longitudinal*2,*3,*4,*10, *121) N= 309 genotyped for<strong>CYP</strong>2D6*2, *3, *4234 (*1/*1), 68 (*1/*2), 7(*2/*2)2) N=214 genotyped for<strong>CYP</strong>2D6*10, *1278 (*1/*1), 97 (*1/*10), 39(*10/*10)AsianSchizophrenia (DSM-IIIR)*3,*4 N=52Ethnicity NR, assumedCaucasian (Estonia)Schizophrenia or Schizo<strong>af</strong>fecti<strong>ved</strong>isorder (ICD-10)Outpatients(35 EMs, 13, IMs, 4 PMs)*3,*4,*5 N= 44 Caucasian (28 EMs,16 IMs)Chronic course ofSchizophrenia (DSM IV)Outpatients*10 N=76Asian (Chinese)Schizophrenia DSM-IVInpatients38 cases, 38 controls188 C/T polymorphism(*10)*3,*4,*5,*6duplicationsN = 216Asian (Taiwan)Schizophrenia (DSM-IV)(48 CC, 81 CT, 87 TT)N=109Ethnicity NR, assumedCaucasian (Germany)Schizophrenia DSM-IVInpatients50 cases, 59 controlsAny APD> 6 month beforeinclusionZuclopenthixol(only decanotate)ADRs: TD (AIMS), EPS(DIESS)ADRs: PS (ESRS), TD (AIMS)Kinetics: SS-conc.Any APD (FGA?) ADRs: Persistent TD(TARDS)NR ADRs: TD (AIMS, Schoolerand Kane criteria),parkinsonism (SAS)Any APD ADRs: TD (RDC/Schoolerand Kane criteria)Any APD(long time treatment)1) IM(4) vs. EM: NS: OR = 6.70 [95% CI:1.26, 35.65] [C]2) IM (10) vs. EM: NS: OR= 1.33 [95% CI:0.54, 3.31] [C]ADRs: PM vs. EM: NS: OR = 6.80 [95%CI: 0.34, 136.02] [C]IM vs. EM: NS: OR = 1.20 [95% CI: 0.33,4.41] [C]Kinetics: Higher median C/D-ratio inPM and IM vs. EM (p
- Page 1 and 2:
Rutinebrug af CYP-test vedantipsyko
- Page 3 and 4:
IndholdHvad er Medicinsk Teknologiv
- Page 5:
Hvad er Medicinsk Teknologivurderin
- Page 8 and 9:
TeknologiCYP-testen er en gentest.
- Page 10 and 11:
Samlet påviser dette studie, at en
- Page 12 and 13:
sættelse. Dette skyldes den store
- Page 14 and 15:
SummaryIntroductionA large part of
- Page 16:
• Are CYP2D6 and/or CYP2C19 geno
- Page 20 and 21:
The costs of pharmacy-dispensed dru
- Page 23 and 24:
Sundhedsstyrelsen vil gerne takke p
- Page 25 and 26:
1 Introduktion1.1 BaggrundDen medik
- Page 27 and 28:
faktor for testens anvendelighed. S
- Page 29 and 30:
lægens ordination af såvel antips
- Page 31 and 32:
NomenklaturCYP-enzymerne inddeles i
- Page 33 and 34:
I det følgende præsenteres egne s
- Page 35 and 36:
Figur 2 Studiepopulationerne i stud
- Page 37 and 38:
ner. Således er de hyppigste mutat
- Page 39 and 40:
lerens kendskab til CYP2D6-genotype
- Page 41 and 42:
3.3 Diskussion af resultaterFra CYP
- Page 43 and 44:
I RCT'et er valgt at vurdere effekt
- Page 45 and 46:
CYP2C19. Ud fra et teoretisk ration
- Page 47 and 48:
Den begrænsede eksisterende viden
- Page 49 and 50:
4.2 ResultaterDet centrale spørgsm
- Page 51 and 52:
med en læge fra det afsnit, han er
- Page 53 and 54: • Til tværfaglige behandlingsko
- Page 55 and 56: liver det tydeligt, at CYP-testen k
- Page 57 and 58: Lægerne i DPC udtrykker i spørges
- Page 59 and 60: På linje med hvad der er fundet i
- Page 61 and 62: Patienter er en central aktør i or
- Page 63 and 64: live udsat for den ’trial and err
- Page 65 and 66: Samtidig rapporterer blot halvdelen
- Page 67: Anna ved, at hun er blevet CYP-test
- Page 70 and 71: dering, er den et godt redskab til
- Page 72 and 73: CYP-testens potentiale ligger i at
- Page 74 and 75: De enkelte omkostningskomponenter,
- Page 76 and 77: medført at anvende CYP-testen. Kon
- Page 79 and 80: Gennemsnittene er dog påvirket af
- Page 82 and 83: Tabel 9 Resultaterne fra to-trins-m
- Page 84 and 85: dette forbrug. Resultaterne viser s
- Page 86 and 87: 7 Samlet vurderingDet overordnede f
- Page 88 and 89: 8 Referencer1. Lieberman JA, Stroup
- Page 90 and 91: 27. Ellingrod VL, Schultz SK, Arndt
- Page 92 and 93: 51. Fleeman N, Dundar Y, Dickson R,
- Page 94 and 95: 79. Cohen AP. Participant observati
- Page 96 and 97: 109. Psykiatrisk Center Sankt Hans.
- Page 98 and 99: Bilag 1 Søgestrategi for Teknologi
- Page 100 and 101: Bilag 3 Metodebeskrivelse af det ra
- Page 102 and 103: Statistiske analyserBehandlingspers
- Page 106 and 107: Reference Design CYP2D6 alleles Pop
- Page 108 and 109: Reference Design CYP2D6 alleles Pop
- Page 110 and 111: Reference Design CYP2D6 alleles Pop
- Page 112 and 113: Bilag 6 IM’ere versus EM’ere me
- Page 114 and 115: Til intensiveret fokus på en regel
- Page 116 and 117: Påvirkning Spørgsmål Rating afin
- Page 118 and 119: Symptom Spørgsmål Rating af svær
- Page 120 and 121: Vrangforestillinger Spørgsmål Rat
- Page 122 and 123: Bilag 8 Søgebeskrivelse: socioetis
- Page 124 and 125: Embase((Mental disease.mp. or menta
- Page 126 and 127: York CRD-databasernePharmacogenetic
- Page 128 and 129: 19841983198219811980197919781977197
- Page 130 and 131: I hvilket omfang føler du dig klæ
- Page 132 and 133: Indtryk af CYP-testenI hvilket omfa
- Page 134 and 135: Hvor hyppigt bruger du følgende re
- Page 136 and 137: Holdninger til CYP-testenAfslutning
- Page 138 and 139: Bilag 10 Søgestrategi for den Sund
- Page 140 and 141: Bilag 12 Resultaterne fra den blind
- Page 142 and 143: Bilag 13 Variable i den økonomiske
- Page 144 and 145: Bilag 15 Studiepopulation i den øk
- Page 146 and 147: IndholdsfortegnelseSide 2:Side 3:Si
- Page 148 and 149: Hvem anvender undersøgelsen:Rekvir
- Page 150 and 151: Samtlige genotyper:CYP2D6Genotype A
- Page 152 and 153: CYP 2D6 vejledninger:Tabel 1. Hurti
- Page 154:
Bilag 17 APPENDIKS til instruks ved
- Page 157 and 158:
(19-hydroxy-risperidon).Det må for
- Page 159 and 160:
CYP 2C19 analyseTabel 4. CYP 2C19-a
- Page 161 and 162:
Tabel 5. CYP 2C19-tilpasset doserin