3. FOOD ChEMISTRy & bIOTEChNOLOGy 3.1. Lectures
3. FOOD ChEMISTRy & bIOTEChNOLOGy 3.1. Lectures
3. FOOD ChEMISTRy & bIOTEChNOLOGy 3.1. Lectures
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Chem. Listy, 102, s265–s1311 (2008) Food Chemistry & Biotechnology<br />
P70 MONITORING OF bARLEy STARCh<br />
AMyLOLySIS by GRAVITATIONAL FIELD<br />
FLOw FRACTIONATION AND MALDI-TOF/<br />
TOF MS<br />
KAREL MAZAnEC and JAnETTE BOBáľOVá<br />
Institute of Analytical Chemistry of the ASCR, v. v. i.Veveří<br />
97, 602 00 Brno, Czech Republic,<br />
mazanec@iach.cz<br />
Introduction<br />
In barley grain, starch occurs in form of granules with<br />
bimodal size distribution. The oval large starch granules (A)<br />
have diameters in the range from 10 to 40 µm and they prevail<br />
in weight. On the other hand, the spherical small starch<br />
granules (B) have diameters from 1 to 10 µm and they prevail<br />
in number 1 . Enzymatic starch granule hydrolysis is one of<br />
the most important reactions occurring during malting and<br />
mashing.<br />
In this study, the capacity of gravitational field flow<br />
fractionation (GFFF) to monitor the amylolysis of starch granules<br />
was investigated. In previous works 2-6 GFFF was successfully<br />
used for study of size distribution of starch granules<br />
in different barley varieties. Degradation of starch particles<br />
from wheat was monitored by sedimentation FFF 7 . Lower<br />
saccharides released during amylolysis can be studied by<br />
MALDI-MS.<br />
Experimental<br />
Kernels of barley variety Jersey were used for isolation<br />
of starch granules. Kernels were graded and the fractions<br />
over 2.5 mm were used. The isolation procedure is described<br />
in detail elsewhere 8 . It combines classical approaches<br />
(incl. crushing of barley kernels by a roll crusher, steeping<br />
in 0.02M HCl, repeated rubbing and filtering through sieve<br />
0.08 mm) and present knowledge (treatment with ß-glucanase<br />
and cellulase).<br />
Starch granules were hydrolyzed by 2 units of<br />
α-amylase mg –1 starch (Sigma-Aldrich) added to 2 ml of 3%<br />
(w/v) starch suspension at 35 °C. Aliquots were removed in<br />
time interval during hydrolysis and centrifuged. Supernatant<br />
was mixed with 2,5-dihydroxybenzoic acid (DHB) and measured<br />
by 4700 Proteomics Analyzer (Applied Biosystems,<br />
USA) MALDI-TOF/TOF mass spectrometer (equipped with<br />
nd/YAG laser; 355 nm).<br />
Starch granules were resuspended in 10 -3 % sodium<br />
dodecyl sulphate (SDS) (Fluka, Germany), sonicated and<br />
analyzed by GFFF. Apparatus for GFFF is described in 6 .<br />
The channel dimensions were 360 × 20 × 0.150 mm. The highpressure<br />
pump HPP 4001 (Laboratory Instruments, Prague,<br />
Czech Republic) was used to introduce the carrier liquid<br />
into the channel via an inlet capillary situated at the channel<br />
head. UV/VIS Spectra 100 (Spectra Physics, San Jose, USA)<br />
operated at 470 nm was used as a detector. The samples were<br />
injected at the stopped flow by using a Hamilton microsyringe.<br />
Just after injection, a loading flow rate of 0.2 ml min –1<br />
s728<br />
was applied for 10 s. Then a relaxation time (stopped-flow<br />
period) was 1.5 min. After this time period, a linear flow rate<br />
of 0.8 ml min –1 was applied.<br />
Results<br />
Experimental conditions of GFFF separation were chosen<br />
in order to elute starch particles in focusing or lift-hyperlayer<br />
elution mode 6 . In this elution mode, starch granules do<br />
not interact with the channel bottom; therefore particle-wall<br />
interactions are negligible.<br />
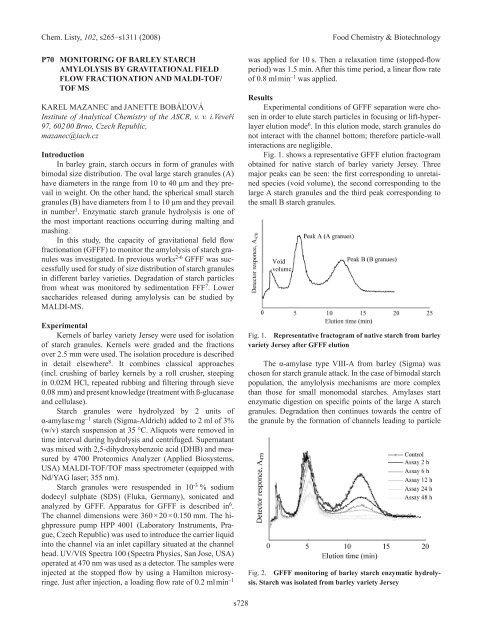
Fig. 1. shows a representative GFFF elution fractogram<br />
obtained for native starch of barley variety Jersey. Three<br />
major peaks can be seen: the first corresponding to unretained<br />
species (void volume), the second corresponding to the<br />
large A starch granules and the third peak corresponding to<br />
the small B starch granules.<br />
Fig. 1. Representative fractogram of native starch from barley<br />
variety jersey after GFFF elution<br />
The α-amylase type VIII-A from barley (Sigma) was<br />
chosen for starch granule attack. In the case of bimodal starch<br />
population, the amylolysis mechanisms are more complex<br />
than those for small monomodal starches. Amylases start<br />
enzymatic digestion on specific points of the large A starch<br />
granules. Degradation then continues towards the centre of<br />
the granule by the formation of channels leading to particle<br />
Fig. 2. GFFF monitoring of barley starch enzymatic hydrolysis.<br />
Starch was isolated from barley variety jersey