LRP1, Adipogenesis, ObesityFigure 3. Inhibition of LRP1 expression by silencing <strong>du</strong>ring adipocyte differentiation. (A) Silencing of LRP1 by siRNAs. 3T3F442Apreadipocytes were transiently transfected with Luc siRNA or LRP1 siRNAs. LRP1b protein expression was monitored by immunoblotting 2 days posttransfection.a tubulin was used as a loading control. A representative experiment out of 3 is shown. (B) Time-course of LRP1b proteinexpression after Luc siRNA or LRP1 siRNAs transfection <strong>du</strong>ring adipogenesis. 3T3F442A preadipocytes were transiently transfected withLuc siRNA or LRP1 siRNAs. Two-days post-transfection, LRP1b protein expression was monitored by immunoblotting 0, 3, 5 or 7 days afterdifferentiation in<strong>du</strong>ction. ERK2 was used as a loading control.doi:10.1371/journal.pone.0007422.g003expression in adipocytes iso<strong>la</strong>ted from C57BI6/J mice either fed aHFD or ND (Fig. 7B). As attempted HFD-fed C57BI6/J miceexhibited a significant increase in body mass (47.661.4 g) whencompared to their control littermates (31.161.2 g) (data not shown).LRP1 expression was significantly enhanced in adipocytes of HFDobese mice when compared to ND control mice (Fig. 7B).Altog<strong>et</strong>her, our results indicate that LRP1 expression is upregu<strong>la</strong>tedin human and mouse obese adipose tissues.Silencing of LRP1 expression inhibits the cellu<strong>la</strong>r lipidcontent of fully-differentiated adipocytesSince we observed that LRP1 expression was up-regu<strong>la</strong>ted inobese adipose tissues, we finally investigated wh<strong>et</strong>her extinction ofLRP1 expression in fully-differentiated adipocytes could diminishtheir lipid content (Fig. 8), as recently suggested in adipose-specificLRP12/2 mouse mo<strong>de</strong>l [12]. 3T3F442A preadipocytes werecultured for 10 days in adipogenic differentiation medium (Fig. 8A,panel a) and fully-differentiated adipocytes were transientlytransfected with Luc siRNA or LRP1 siRNA1 (Fig. 8A, panel b).Oil red O staining revealed that, after 7 days of culture, the lipiddropl<strong>et</strong> size and number were <strong>de</strong>creased in LRP1 siRNA1transfected adipocytes (Fig. 8B–C). Quantification of lipids atdays 2 and 7 of culture revealed that, in Luc siRNA transfectedadipocytes, the level of cellu<strong>la</strong>r lipid remained unchanged whereas,in LRP1 siRNA1 transfected cells, the amount of lipid wassignificantly diminished by 27.8% (Fig. 8D). Interestingly, basallipolysis was significantly stimu<strong>la</strong>ted in LRP1 siRNA1 transfectedadipocytes (Fig. 9, panel a). We postu<strong>la</strong>te that, since cells could notinternalized triglyceri<strong>de</strong>s in the absence of LRP1 as previouslyshown [12], they were m<strong>et</strong>abolizing their intracellu<strong>la</strong>r lipid stock.However, in<strong>du</strong>ced lipolysis was not different in LRP1 and LucsiRNA transfected cells (Fig. 9, panel b). Altog<strong>et</strong>her, these findingshighlight, for the first time, the crucial role of LRP1 in controllingadipogenesis and maintaining the lipid content in fully-differen-PLoS ONE | www.plosone.org 4 October 2009 | Volume 4 | Issue 10 | e7422
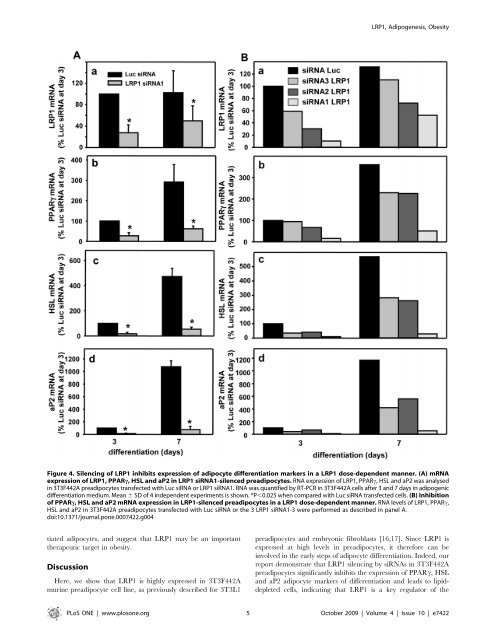
LRP1, Adipogenesis, ObesityFigure 4. Silencing of LRP1 inhibits expression of adipocyte differentiation markers in a LRP1 dose-<strong>de</strong>pen<strong>de</strong>nt manner. (A) mRNAexpression of LRP1, PPARc, HSL and aP2 in LRP1 siRNA1-silenced preadipocytes. RNA expression of LRP1, PPARc, HSL and aP2 was analysedin 3T3F442A preadipocytes transfected with Luc siRNA or LRP1 siRNA1. RNA was quantified by RT-PCR in 3T3F442A cells after 3 and 7 days in adipogenicdifferentiation medium. Mean 6 SD of 4 in<strong>de</strong>pen<strong>de</strong>nt experiments is shown. *P,0.025 when compared with Luc siRNA transfected cells. (B) Inhibitionof PPARc, HSL and aP2 mRNA expression in LRP1-silenced preadipocytes in a LRP1 dose-<strong>de</strong>pen<strong>de</strong>nt manner. RNA levels of LRP1, PPARc,HSL and aP2 in 3T3F442A preadipocytes transfected with Luc siRNA or the 3 LRP1 siRNA1-3 were performed as <strong>de</strong>scribed in panel A.doi:10.1371/journal.pone.0007422.g004tiated adipocytes, and suggest that LRP1 may be an importanttherapeutic targ<strong>et</strong> in obesity.DiscussionHere, we show that LRP1 is highly expressed in 3T3F442Amurine preadipocyte cell line, as previously <strong>de</strong>scribed for 3T3L1preadipocytes and embryonic fibrob<strong>la</strong>sts [16,17]. Since LRP1 isexpressed at high levels in preadipocytes, it therefore can beinvolved in the early steps of adipocyte differentiation. In<strong>de</strong>ed, ourreport <strong>de</strong>monstrate that LRP1 silencing by siRNAs in 3T3F442Apreadipocytes significantly inhibits the expression of PPARc, HSLand aP2 adipocyte markers of differentiation and leads to lipid<strong>de</strong>pl<strong>et</strong>edcells, indicating that LRP1 is a key regu<strong>la</strong>tor of thePLoS ONE | www.plosone.org 5 October 2009 | Volume 4 | Issue 10 | e7422
- Page 1 and 2:
Université Montpellier I UFR Méd
- Page 3 and 4:
Un grand merci à tous mes collabor
- Page 5 and 6:
TitleBreast cancer and tumoral micr
- Page 7 and 8:
Cancer du sein et micro-environneme
- Page 9 and 10:
C. PRESENTATION DU TRAVAIL DE THESE
- Page 11 and 12:
But de la thèseLa cathepsine-D (ca
- Page 13 and 14:
I. Le tissu adipeux1) Généralité
- Page 15 and 16:
(Adenosine triphosphate). Cette pro
- Page 18 and 19:
Cette synthèse de novo a lieu dans
- Page 20 and 21:
ABFigure 6 : Schéma de la oxydati
- Page 22 and 23:
d. Ladipocyte : une cellule sécré
- Page 24 and 25:
3) Ladipogenèsea. Les différentes
- Page 26 and 27:
. Les facteurs de transcriptionLa d
- Page 28 and 29:
Lexpression de C/EBP est quant à e
- Page 30 and 31:
adipeux. Les analyses histologiques
- Page 32 and 33:
adipocyteFigure 10 : Schéma repré
- Page 34 and 35:
II. La cathepsine-D1) Synthèse, ma
- Page 36 and 37:
les cath-B et L, en une forme matur
- Page 38 and 39:
Il existe deux RM6P : le RM6P/IGFII
- Page 40 and 41:
2) Fonctions de la cath-Da. Dans la
- Page 42 and 43:
. Dans les pathologiesEn plus de se
- Page 44 and 45:
c. Rôles et mécanismes daction da
- Page 46 and 47:
carcinome de prostate serait respon
- Page 48 and 49:
La cath-D joue donc un rôle import
- Page 50 and 51: Une fois le marquage des peptides e
- Page 52 and 53: Afin de réaliser ce projet, nous a
- Page 54 and 55: III. Le récepteur LRP11) Organisat
- Page 56 and 57: 2) Trafic intra-cellulaireComme le
- Page 58 and 59: LRP1αLRP1Membrane associatedprotea
- Page 60 and 61: Tableau 3 : Ligands connus du LRP1(
- Page 62 and 63: I. Etude du rôle de la cathepsine
- Page 64 and 65: Cathepsin-D, a key protease in brea
- Page 66 and 67: IntroductionConsumption of meals ri
- Page 68 and 69: (Fig. 1A, panel a). This differenti
- Page 70 and 71: differentiated adipocyte (Fig. 4B).
- Page 72 and 73: DiscussionOur results demonstrate t
- Page 74 and 75: from normal and peri-al breast adip
- Page 76 and 77: mouse RS9 (sens 5CGGCCCGGGAGCTGTTGA
- Page 78 and 79: agreement of local ethic committee.
- Page 80 and 81: Statistical analysis. Results are e
- Page 82 and 83: Loncarek J, Freiss G, Vignon F and
- Page 84 and 85: Aa3000 *b4000*B8000**cath-D mRNA(ar
- Page 86 and 87: ABcath-D mRNA(ratio RS9)PPARg mRNA(
- Page 88 and 89: A D3 D7 D14D0BaD0 D3 D7 D14D0 D3 D7
- Page 90 and 91: AshLucshcath-DF442A C34 C37 A4 D10c
- Page 92 and 93: AF442-AC34C37A4D10BF442-AC34C37A4D1
- Page 94 and 95: II. Etude du rôle du LRP1 dans les
- Page 96 and 97: 2) Article 2:LRP1 receptor controls
- Page 98 and 99: LRP1, Adipogenesis, Obesityrelative
- Page 102 and 103: LRP1, Adipogenesis, ObesityFigure 5
- Page 104 and 105: LRP1, Adipogenesis, ObesitySome ins
- Page 106 and 107: LRP1, Adipogenesis, ObesityLipolysi
- Page 108 and 109: CONCLUSIONLa cathepsine D (cath-D)
- Page 110 and 111: la souris obèses. Enfin, linhibiti
- Page 112 and 113: carcinogénique sont encore mal con
- Page 114 and 115: Ce travail de thèse a étudié, po
- Page 116 and 117: REFERENCESAhima, R. S. (2006). Adip
- Page 118 and 119: implicates them as antigen presenti
- Page 120 and 121: Cataldo, A. M., Barnett, J. L., Ber
- Page 122 and 123: Folkman, J. (2003). Fundamental con
- Page 124 and 125: Hofmann, S. M., Zhou, L., Perez-Til
- Page 126 and 127: Langin, D. (2006a). Adipose tissue
- Page 128 and 129: Ludwig, T., Ovitt, C. E., Bauer, U.
- Page 130 and 131: Nirde, P., Derocq, D., Maynadier, M
- Page 132 and 133: Rozanov, D. V., Hahn-Dantona, E., S
- Page 134 and 135: Taleb, S., Cancello, R., Clement, K
- Page 136 and 137: Xiao, Y., Junfeng, H., Tianhong, L.
- Page 138 and 139: F. ANNEXE98
- Page 140 and 141: Cathepsin D is a new ligand for ext
- Page 142 and 143: INTRODUCTIONLysosomal aspartic prot
- Page 144 and 145: pcDNA3.1(+)Myc-tagged LRP1b into pc
- Page 146 and 147: (Protein refolding kit, Novagen) fo
- Page 148 and 149: anti-mouse-gold (Aurion). Sections
- Page 150 and 151:
not secrete detectable levels of pr
- Page 152 and 153:
using cath-D-/- MEF transfected wit
- Page 154 and 155:
co-culture outgrowth assays with ca
- Page 156 and 157:
REFERENCES1. Vignon, F., Capony, F.
- Page 158 and 159:
steps in vivo: proliferation, angio
- Page 160 and 161:
28. Laurent-Matha, V., Lucas, A., H
- Page 162 and 163:
42. Zurhove, K., Nakajima, C., Herz
- Page 164 and 165:
Figure 1. Cath-D interacts with the
- Page 166 and 167:
Figure 2. Cath-D binds to residues
- Page 168 and 169:
Figure 3. Cath-D interacts with LRP
- Page 170 and 171:
Figure 5. Silencing LRP1 in cath-D
- Page 172 and 173:
Figure 1. Cath-D interacts with the
- Page 174 and 175:
Figure 6. LRP1 is the receptor medi
- Page 176 and 177:
Beaujouin, Figure Sup. 2cath-D-/-ME
- Page 178:
RésuméLaspartyl protéase catheps