toxicological profile for malathion - Agency for Toxic Substances and ...
toxicological profile for malathion - Agency for Toxic Substances and ...
toxicological profile for malathion - Agency for Toxic Substances and ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
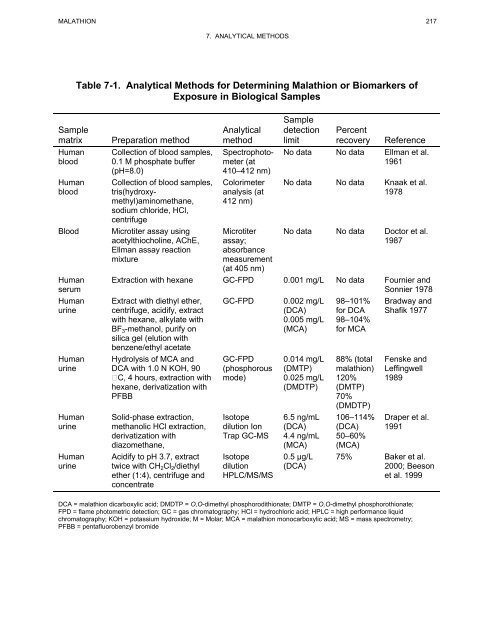
MALATHION 217<br />
Sample<br />
matrix<br />
Human<br />
blood<br />
Human<br />
blood<br />
7. ANALYTICAL METHODS<br />
Table 7-1. Analytical Methods <strong>for</strong> Determining Malathion or Biomarkers of<br />
Exposure in Biological Samples<br />
Preparation method<br />
Collection of blood samples,<br />
0.1 M phosphate buffer<br />
(pH=8.0)<br />
Collection of blood samples,<br />
tris(hydroxymethyl)aminomethane,<br />
sodium chloride, HCl,<br />
centrifuge<br />
Blood Microtiter assay using<br />
acetylthiocholine, AChE,<br />
Ellman assay reaction<br />
mixture<br />
Human<br />
serum<br />
Human<br />
urine<br />
Human<br />
urine<br />
Human<br />
urine<br />
Human<br />
urine<br />
Analytical<br />
method<br />
Spectrophotometer<br />
(at<br />
410–412 nm)<br />
Colorimeter<br />
analysis (at<br />
412 nm)<br />
Microtiter<br />
assay;<br />
absorbance<br />
measurement<br />
(at 405 nm)<br />
Sample<br />
detection Percent<br />
limit recovery Reference<br />
No data No data Ellman et al.<br />
1961<br />
No data No data Knaak et al.<br />
1978<br />
No data No data Doctor et al.<br />
1987<br />
Extraction with hexane GC-FPD 0.001 mg/L No data Fournier <strong>and</strong><br />
Sonnier 1978<br />
Extract with diethyl ether,<br />
centrifuge, acidify, extract<br />
with hexane, alkylate with<br />
BF3-methanol, purify on<br />
silica gel (elution with<br />
benzene/ethyl acetate<br />
Hydrolysis of MCA <strong>and</strong><br />
DCA with 1.0 N KOH, 90<br />
C, 4 hours, extraction with<br />
hexane, derivatization with<br />
PFBB<br />
Solid-phase extraction,<br />
methanolic HCl extraction,<br />
derivatization with<br />
diazomethane,<br />
Acidify to pH 3.7, extract<br />
twice with CH2Cl2/diethyl<br />
ether (1:4), centrifuge <strong>and</strong><br />
concentrate<br />
GC-FPD 0.002 mg/L<br />
(DCA)<br />
0.005 mg/L<br />
(MCA)<br />
GC-FPD<br />
(phosphorous<br />
mode)<br />
Isotope<br />
dilution Ion<br />
Trap GC-MS<br />
Isotope<br />
dilution<br />
HPLC/MS/MS<br />
0.014 mg/L<br />
(DMTP)<br />
0.025 mg/L<br />
(DMDTP)<br />
6.5 ng/mL<br />
(DCA)<br />
4.4 ng/mL<br />
(MCA)<br />
0.5 µg/L<br />
(DCA)<br />
98–101%<br />
<strong>for</strong> DCA<br />
98–104%<br />
<strong>for</strong> MCA<br />
88% (total<br />
<strong>malathion</strong>)<br />
120%<br />
(DMTP)<br />
70%<br />
(DMDTP)<br />
106–114%<br />
(DCA)<br />
50–60%<br />
(MCA)<br />
Bradway <strong>and</strong><br />
Shafik 1977<br />
Fenske <strong>and</strong><br />
Leffingwell<br />
1989<br />
Draper et al.<br />
1991<br />
75% Baker et al.<br />
2000; Beeson<br />
et al. 1999<br />
DCA = <strong>malathion</strong> dicarboxylic acid; DMDTP = O,O-dimethyl phosphorodithionate; DMTP = O,O-dimethyl phosphorothionate;<br />
FPD = flame photometric detection; GC = gas chromatography; HCl = hydrochloric acid; HPLC = high per<strong>for</strong>mance liquid<br />
chromatography; KOH = potassium hydroxide; M = Molar; MCA = <strong>malathion</strong> monocarboxylic acid; MS = mass spectrometry;<br />
PFBB = pentafluorobenzyl bromide