1.1 Porphyrins - Friedrich-Alexander-Universität Erlangen-Nürnberg
1.1 Porphyrins - Friedrich-Alexander-Universität Erlangen-Nürnberg
1.1 Porphyrins - Friedrich-Alexander-Universität Erlangen-Nürnberg
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
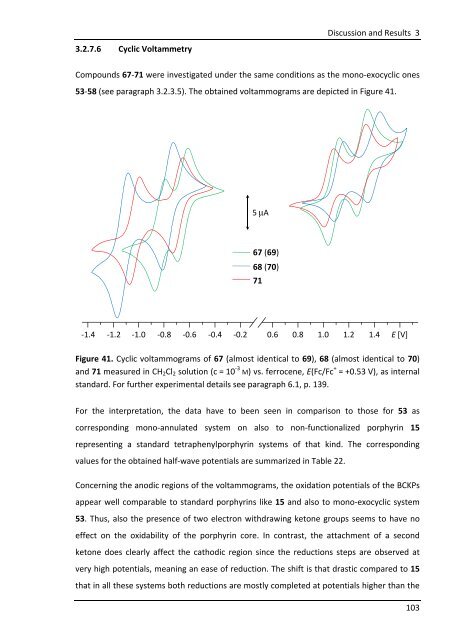
3.2.7.6 Cyclic Voltammetry<br />
Discussion and Results 3<br />
Compounds 67-71 were investigated under the same conditions as the mono-exocyclic ones<br />
53-58 (see paragraph 3.2.3.5). The obtained voltammograms are depicted in Figure 41.<br />
-1.4 -1.2 -1.0 -0.8 -0.6 -0.4 -0.2 0.6 0.8 1.0 1.2 1.4 E [V]<br />
Figure 41. Cyclic voltammograms of 67 (almost identical to 69), 68 (almost identical to 70)<br />
and 71 measured in CH2Cl2 solution (c = 10 -3 M) vs. ferrocene, E(Fc/Fc + = +0.53 V), as internal<br />
standard. For further experimental details see paragraph 6.1, p. 139.<br />
For the interpretation, the data have to been seen in comparison to those for 53 as<br />
corresponding mono-annulated system on also to non-functionalized porphyrin 15<br />
representing a standard tetraphenylporphyrin systems of that kind. The corresponding<br />
5 μA<br />
67 (69)<br />
68 (70)<br />
71<br />
values for the obtained half-wave potentials are summarized in Table 22.<br />
Concerning the anodic regions of the voltammograms, the oxidation potentials of the BCKPs<br />
appear well comparable to standard porphyrins like 15 and also to mono-exocyclic system<br />
53. Thus, also the presence of two electron withdrawing ketone groups seems to have no<br />
effect on the oxidability of the porphyrin core. In contrast, the attachment of a second<br />
ketone does clearly affect the cathodic region since the reductions steps are observed at<br />
very high potentials, meaning an ease of reduction. The shift is that drastic compared to 15<br />
that in all these systems both reductions are mostly completed at potentials higher than the<br />
103