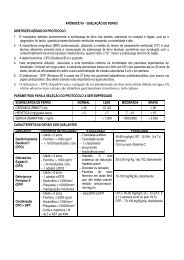
Protocols - Hemorio
Protocols - Hemorio
Protocols - Hemorio
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
PHARMACOKINETICS OF CLOTTING FACTORS: the pharmacokinetics study of concentrate factor to<br />
be used must be performed within an interval of one week to 3 days before surgery. ISTH recommends<br />
the study to be performed with 30 IU/kg only dose infusion and serial sample draws for dosage of infused<br />
factor activity level. The first sample is pre-infusion, followed by 8 more samples drawn from the arm not<br />
infused, to determine the maximum peak of factor level, considering at least 3 samples post-infusion. Reusage<br />
is given by the factor activity in these 3 samples and it is expressed in % by the formula (ml/kg/h) =<br />
infusion index (IU/kg/h) / plasma level (lU/ml).<br />
METHOD: One FVIII dose in “bolus” is infused on pre-operatory. This dose is calculated to reach plasma<br />
levels between 60-80%. The continuous infusion starts right after the end of surgery or even during it. The<br />
dose administered is based on clearance obtained by the pharmacokinetics study according to the<br />
formula: FVIII level post- and pre-infusion X weight (Kg) / Factor dose given in IU.<br />
NOTES: PTTa or FVIII dosage must be performed 4-6 hours after surgery to confirm if the dose is<br />
sufficient. Clearance is calculated every day, based on FVIII day activity to adjust the infusion and<br />
therefore, reach the desirable FVIII levels. For major surgery, the minimum desirable level is 50 and 30%<br />
during the 1 st week and 2 nd week, respectively.<br />
In case of emergency surgery or major bleeding, the “bolus” dose is the same for elective surgeries and<br />
the continuous infusion must be installed immediately after the “bolus” dose.<br />
When the pharmacokinetics study is not possible, it may be administered on the 1st infusion of 2 to<br />
4IU/kg/h depending on the surgery or bleeding. In the next day, a sample is drawn to dose the factor<br />
plasma level and establish continuity.<br />
Overall, it is noted a decreased amount of factor required to keep the same plasma level.<br />
DOMICILE DOSE PROGRAM<br />
Intervention in the hemorrhage episode in hemophilia carriers, reaches its maximum efficiency with<br />
immediate access to the clotting factor, limiting the bleeding and the extension of resulting tissue damage.<br />
Early replacement therapy decreased the amount of clotting factor required to control the bleeding<br />
situation. The Domicile Dose Program aims to offer eligible hemophilia patients, 3 doses of clotting factor<br />
concentrate which increases the plasma level to 40%, for self-infusion at home, allowing early drug<br />
therapy. Thus, it reduces the “stress” arising from the need to go get the specific assistance to the drug<br />
and also allows the patient to participate in an active manner in his/her treatment.<br />
68