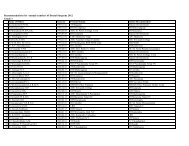
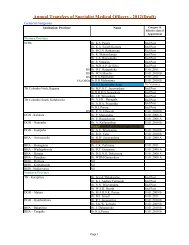
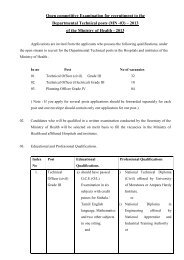
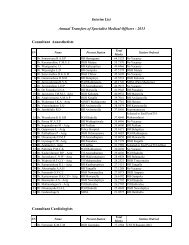
- Page 2 and 3:
MANUAL ON MANAGEMENT OF DRUGS SECON
- Page 4 and 5:
MESSAGE BY THE SECRETARY/ MINISTRY
- Page 6 and 7:
ACKNOWLEDGEMENT It is a great pleas
- Page 8 and 9:
Dr. J. M. J. Munasinghe Dr. K. C. L
- Page 10 and 11:
ABBREVIATIONS AD CP CDD CDDA D(F) D
- Page 12 and 13:
CONTENTS Message by the Hon. Minist
- Page 14 and 15:
X Form for Drug sample for quality
- Page 16 and 17:
CHAPTER 1 INTRODUCTION 1.Ensuring a
- Page 18 and 19:
3.The expenditure on drugs has been
- Page 20 and 21:
of poisons, opium, and dangerous dr
- Page 22 and 23:
(Medical Technology & Supplies) No
- Page 24 and 25:
c) NDQAL for quality assurance of d
- Page 26 and 27:
Deputy Director General (Finance) D
- Page 28 and 29:
(d) The Institutional Drug Therapeu
- Page 30 and 31:
CHAPTER 4 ESTIMATION OF ANNUAL REQU
- Page 32 and 33:
Step 2 Identify the stock available
- Page 34 and 35:
In the case of institutions under t
- Page 36 and 37:
oard appointed by him and a report
- Page 38 and 39:
Head of the Institution may add any
- Page 40 and 41:
ii. Injections costing Rs. 25.00 or
- Page 42 and 43:
42. Delivery schedules are planned
- Page 44 and 45:
CHAPTER 6 CARE IN PRESCRIBING DRUGS
- Page 46 and 47:
CHAPTER 7 DISTRIBUTION AND LOGISTIC
- Page 48 and 49:
the institution. Records of the inv
- Page 50 and 51:
RMSD or institutional stores, the o
- Page 52 and 53:
CHAPTER 8 QUALITY ASSURANCE OF DRUG
- Page 54 and 55:
ioavailability. Further, in apprais
- Page 56 and 57:
x. Details of any preservative adde
- Page 58 and 59:
To request institutions to inform t
- Page 60 and 61:
CHAPTER 9 STORAGE OF DRUGS. 1. Prop
- Page 62 and 63:
xi. Storage of vaccine in the refri
- Page 64 and 65: CHAPTER 10 MONITORING OF CONSUMPTIO
- Page 66 and 67: ii. VEN analysis ( See annexure XV
- Page 68 and 69: 9. Monitoring of quality of drugs;
- Page 70 and 71: alance as per ledger, actual balanc
- Page 72 and 73: 1.3 In case of institutions coming
- Page 74 and 75: Ampoules should be crushed and buri
- Page 76 and 77: ANNEXURE I1: Main functions of Medi
- Page 78 and 79: I - Annual stock holding cost as a
- Page 80 and 81: Anti Rabies cell culture vaccine. (
- Page 82 and 83: unobservable (animal dead, killed,
- Page 84 and 85: on D0, D3, D7, D14 & D30. 6.1.2 For
- Page 86 and 87: All patients who receive rabies PET
- Page 88 and 89: Monthly return for vaccine consumpt
- Page 90 and 91: Death in the house / outside a Medi
- Page 92 and 93: 2.2 In case of major exposures: 2.2
- Page 94 and 95: ANNEXURE - V MyNo……/…../….
- Page 96 and 97: Advertisement Screening Unit Unit o
- Page 98 and 99: ANNEXURE VIII (To be amended To: My
- Page 100 and 101: ANNEXURE X ……………………
- Page 102 and 103: 6. Quality failure of registration
- Page 105 and 106: ANNEXURE XIII Lot Card SR No……
- Page 107 and 108: ANNEXURE XV VEN Analysis This analy
- Page 109: Sample Guidelines for VEN Category
- Page 112 and 113: Unsatisfactory clinical response, a