SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
M3 <br />
AMPHETAMINE BINDING TO SYNTHETIC MELANIN AND SCATCHARD ANALYSIS OF BINDING<br />
DATA<br />
Lata Gautam*, Karen S Scott, and Michael Cole: Department <strong>of</strong> <strong>Forensic</strong> Science and Chemistry, APU, Cambridge,<br />
United Kingdom, CBI IPT<br />
The determination <strong>of</strong> drug use and drug history has historically been carried out through the analysis <strong>of</strong> biological<br />
fluids (blood and urine). In the past decade, hair analysis has been exceedingly used in order to determine recent past<br />
drug use as well as long-term drug histories through the use <strong>of</strong> segmental analysis. In order to fully understand the<br />
implications <strong>of</strong> a positive hair test result, it is important to understand how the drugs are incorporated into hair and how<br />
they bind. Keratin, melanin and lipids are possible drug binding sites in hair. Keratin and melanin have many polar<br />
groups which serve as attachment points for the drugs. Previous research into drug-hair binding has shown that<br />
different amounts <strong>of</strong> drug bind in hair <strong>of</strong> different colours. There are no structural differences in hair <strong>of</strong> different<br />
colours other than in the type and content <strong>of</strong> melanin present. For this reason, this investigation focuses on<br />
amphetamine binding to synthetic melanin in order to help to explore the possible binding mechanisms.<br />
The binding study was carried out at room temperature in the presence <strong>of</strong> light and oxygen using 0.1 M phosphate<br />
buffer at pH 7.4. Both compounds were found to be stable under these conditions. Amphetamine-d-sulphate and<br />
melanin, synthesized by the oxidation <strong>of</strong> tyrosine, were purchased from Sigma Chemical Company. As melanin is only<br />
sparingly soluble in buffer, it was solubilised in dimethyl sulphoxide (DMSO) prior to dilution with buffer. An indirect<br />
spectroscopic method was used by measuring the decrease in absorbance <strong>of</strong> amphetamine as the concentration <strong>of</strong><br />
melanin was increased. Aliquots <strong>of</strong> amphetamine were titrated against the melanin solution. Samples were incubated<br />
for 45 minutes with constant shaking. Background correction was done at each step using the same amount <strong>of</strong> melanin<br />
as present in the sample for matrix matching. The interaction was monitored using a Genesys 6v, UVNis<br />
spectrophotometer in scanning mode from 250-264 nm focussing on changes in the amphetamine spectral fingerprint at<br />
257.2 nm. Positive controls were prepared using the same amount <strong>of</strong> amphetamine as in the sample with the addition <strong>of</strong><br />
buffer instead <strong>of</strong> melanin at each step. Buffer was used for the background correction to run positive controls.<br />
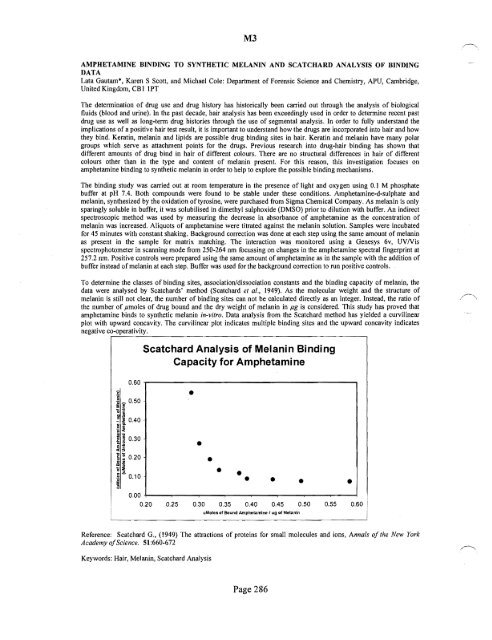
To determine the classes <strong>of</strong> binding sites, association/dissociation constants and the binding capacity <strong>of</strong> melanin, the<br />
data were analysed by Scatchards' method (Scatchard et al., 1949). As the molecular weight and the structure <strong>of</strong><br />
melanin is still not clear, the number <strong>of</strong> binding sites can not be calculated directly as an integer. Instead, the ratio <strong>of</strong><br />
the number <strong>of</strong> pmoles <strong>of</strong> drug bound and the dry weight <strong>of</strong> melanin in pg is considered. This study has proved that<br />
amphetamine binds to synthetic melanin in-vitro. Data analysis from the Scatchard method has yielded a curvilinear<br />
plot with upward concavity. The curvilinear plot indicates multiple binding sites and the upward concavity indicates<br />
negative co-operativity.<br />
Scatchard Analysis <strong>of</strong> Melanin Binding <br />
Capacity for Amphetamine <br />
0.60,.......-----------------------, <br />
•<br />
0.10<br />
•<br />
•<br />
• •• • • •<br />
0.00<br />
0.20 0.25 0.30 0.35 0.40 0.45 0.50 0.55 0.60 !<br />
Reference: Scatchard G., (1949) The attractions <strong>of</strong> proteins for small molecules and ions, Annals 0/ the New York<br />
Academy o/Science. 51:660-672<br />
Keywords: Hair, Melanin, Scatchard Analysis<br />
Page 286