SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
SOFT 2004 Meeting Abstracts - Society of Forensic Toxicologists
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
A48 <br />
MULTICENTER EV ALUA TION OF THE ROCHE ONLINE® TDM CARBAZEPINE ASSA Y FOR<br />
ROCHEIHITACHI ANALYZER SYSTEMS<br />
Mary Jane C<strong>of</strong>fing* I, Roger L. Bertholf, Martin Kroll 3 , Hilmar H. Luthe 4 , Alain Verstraete 5, Heike<br />
Sauter 6 , RaifRoeddiger 6<br />
IClinical Trials, Roche Diagnostics Cooperation, Indianapolis, IN, USA; 2University <strong>of</strong> Florida Health<br />
Science Center, Jacksonville, FL, USA; 3Dallas VA Medical Center, Dallas, TX, USA; 4Department<br />
Clinical Chemistry, Universitiit G6ttingen, Germany; 5Ghent University Hospital, Ghent, Belgium;<br />
6Clinical Trials, Roche Diagnostics GmbH, Mannheim, Germany<br />
Carbamazepine is an anticonvulsant and anti-manic drug, used in the treatment <strong>of</strong> trigeminal neuralgia,<br />
bipolar disorders, epilepsy, and a wide variety <strong>of</strong> seizure disorders. Monitoring carbamazepine<br />
concentrations is essential during therapy in order to ensure achievement <strong>of</strong> optimal therapeutic effect,<br />
while avoiding the impact <strong>of</strong> both subtherapeutic and toxic drug levels.<br />
The analytical performance <strong>of</strong> a new, Roche homogeneous microparticle immunoassay for Carbamazepine<br />
was evaluated at four sites two in Europe and two in the Unites States. Intra- and interassay imprecision,<br />
lower detection limit, control recovery, and linearity were assessed. Further, the assay method was compared<br />
to the CEDIA Carbamazepine assay on RochelHitachi 917 and MODULAR P analyzers, Abbott FPIA<br />
Carbamazepine assay on the Abbott AxSYM and TDx analyzers, and the Roche FP assay on INTEGRA 800.<br />
Serum vs. sodium heparin plasma comparison was also performed.<br />
OnLine Carbamazepine intra-assay imprecision showed 8Ds sO.14 J..lg/mL for concentrations up to 4<br />
J..lg/mL and CVs s3.4 for concentrations >4 J..lg/mL. Interassay imprecision showed SDs s0.15 J..lglmL for<br />
concentrations up to 4 J..lglmL and CVs s4.1 for concentrations >4 J..lglmL. Analytical sensitivity (lower<br />
detection limit) to 0.22 J.lglmL and linearity to the 20.0 J..lglmL were observed. Roche COBAS FP control<br />
materials recovered within range at all sites and competitor control materials recovered within<br />
manufacturers' ranges with one exception.<br />
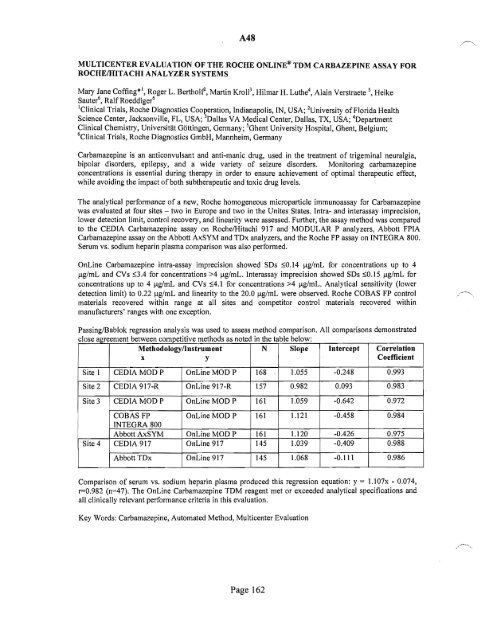
PassinglBablok regression analysis was used to assess method comparison. All comparisons demonstrated<br />
c I ose agreement between competitive met h 0 d s as note d'mthetable b eow: I<br />
Methodoiogyllnstrument<br />
N Slope Intercept Correlation<br />
x<br />
y<br />
Coefficient<br />
Site 1 CEDIA MODP OnLine MOD P 168 1.055 -0.248 0.993<br />
8ite2 CEDIA 917-R OnLine 917-R 157 0.982 0.093 0.983<br />
Site 3 CEDIA MODP OnLine MODP 161 1.059 -0.642 0.972<br />
COBASFP OnLineMODP 161 1.121 -0.458 0.984<br />
INTEGRA 800<br />
Abbott AxSYM I OnLine MOD P 161 1.120 -0.426 0.975<br />
Site 4 CEDIA 917 OnLine 917 145 1.039 -0.409 0.988<br />
Abbott TDx OnLine 917 145 1.068 -0.111 0.986<br />
Comparison <strong>of</strong> serum vs. sodium heparin plasma produced this regression equation: y 1.l07x - 0.074,<br />
r=0.982 (n=47). The OnLine Carbamazepine TDM reagent met or exceeded analytical specifications and<br />
all clinically relevant performance criteria in this evaluation.<br />
Key Words: Carbamazepine, Automated Method, Multicenter Evaluation<br />
Page 162