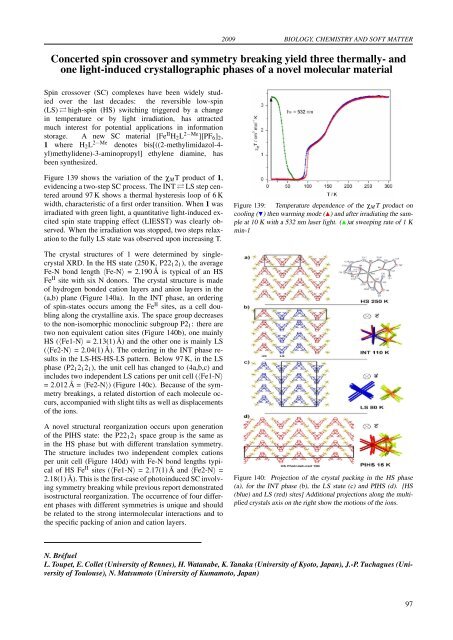
2009 BIOLOGY, CHEMISTRY AND SOFT MATTERConcerted spin crossover and symmetry breaking yield three thermally- andone light-induced crystallographic phases of a novel molecular materialSpin crossover (SC) complexes have be<strong>en</strong> widely studiedover the last deca<strong>des</strong>: the reversible low-spin(LS) ⇄ high-spin (HS) switching triggered by a changein temperature or by light irradiation, has attractedmuch interest for pot<strong>en</strong>tial applications in informationstorage. A new SC material [Fe II H 2 L 2−Me ][PF 6 ] 2 ,1 where H 2 L 2−Me d<strong>en</strong>otes bis[((2-methylimidazol-4-yl)methylid<strong>en</strong>e)-3-aminopropyl] ethyl<strong>en</strong>e diamine, hasbe<strong>en</strong> synthesized.Figure 139 shows the variation of the χ M T product of 1,evid<strong>en</strong>cing a two-step SC process. The INT ⇄ LS step c<strong>en</strong>teredaround 97 K shows a thermal hysteresis loop of 6 Kwidth, characteristic of a first order transition. Wh<strong>en</strong> 1 wasirradiated with gre<strong>en</strong> light, a quantitative light-induced excitedspin state trapping effect (LIESST) was clearly observed.Wh<strong>en</strong> the irradiation was stopped, two steps relaxationto the fully LS state was observed upon increasing T.Figure 139: Temperature dep<strong>en</strong>d<strong>en</strong>ce of the χ M T product oncooling () th<strong>en</strong> warming mode () and after irradiating the sampleat 10 K with a 532 nm laser light. ()at sweeping rate of 1 Kmin-1The crystal structures of 1 were determined by singlecrystalXRD. In the HS state (250 K, P22 1 2 1 ), the averageFe-N bond l<strong>en</strong>gth 〈Fe-N〉 = 2.190 Å is typical of an HSFe II site with six N donors. The crystal structure is madeof hydrog<strong>en</strong> bonded cation layers and anion layers in the(a,b) plane (Figure 140a). In the INT phase, an orderingof spin-states occurs among the Fe II sites, as a cell doublingalong the crystalline axis. The space group decreasesto the non-isomorphic monoclinic subgroup P2 1 : there aretwo non equival<strong>en</strong>t cation sites (Figure 140b), one mainlyHS (〈Fe1-N〉 = 2.13(1) Å) and the other one is mainly LS(〈Fe2-N〉 = 2.04(1) Å). The ordering in the INT phase resultsin the LS-HS-HS-LS pattern. Below 97 K, in the LSphase (P2 1 2 1 2 1 ), the unit cell has changed to (4a,b,c) andinclu<strong>des</strong> two indep<strong>en</strong>d<strong>en</strong>t LS cations per unit cell (〈Fe1-N〉= 2.012 Å = 〈Fe2-N〉) (Figure 140c). Because of the symmetrybreakings, a related distortion of each molecule occurs,accompanied with slight tilts as well as displacem<strong>en</strong>tsof the ions.A novel structural reorganization occurs upon g<strong>en</strong>erationof the PIHS state: the P22 1 2 1 space group is the same asin the HS phase but with differ<strong>en</strong>t translation symmetry.The structure inclu<strong>des</strong> two indep<strong>en</strong>d<strong>en</strong>t complex cationsper unit cell (Figure 140d) with Fe-N bond l<strong>en</strong>gths typicalof HS Fe II sites (〈Fe1-N〉 = 2.17(1) Å and 〈Fe2-N〉 =2.18(1) Å). This is the first-case of photoinduced SC involvingsymmetry breaking while previous report demonstratedisostructural reorganization. The occurr<strong>en</strong>ce of four differ<strong>en</strong>tphases with differ<strong>en</strong>t symmetries is unique and shouldbe related to the strong intermolecular interactions and tothe specific packing of anion and cation layers.Figure 140: Projection of the crystal packing in the HS phase(a), for the INT phase (b), the LS state (c) and PIHS (d). [HS(blue) and LS (red) sites] Additional projections along the multipliedcrystals axis on the right show the motions of the ions.N. BréfuelL. Toupet, E. Collet (University of R<strong>en</strong>nes), H. Watanabe, K. Tanaka (University of Kyoto, Japan), J.-P. Tuchagues (Universityof Toulouse), N. Matsumoto (University of Kumamoto, Japan)97
BIOLOGY, CHEMISTRY AND SOFT MATTER 2009Magnetostructural correlations in Tetrairon(III) single-molecule magnetsSlow magnetic relaxation in molecular systems is a livelyresearch area which spans the interface betwe<strong>en</strong> chemistry,physics and material sci<strong>en</strong>ce. This ph<strong>en</strong>om<strong>en</strong>on is observedin two main families of compounds: single-molecule magnets(SMM) and single-chain magnets (SCM). The persist<strong>en</strong>ceof magnetization in such systems is limited to lowtemperature (below about 4.2 K), but can be in principleexploited for applications in the field of magnetic storageand information processing. Tetrairon(III) complexes withformula [Fe 4 (L) 2 (dpm) 6 ],are providing a growing class ofSingle Molecule Magnets displaying unpreced<strong>en</strong>ted syntheticflexibility and ease of functionalization ( Hdpm =2,2,6,6 − tetramethyl-heptane-3,5-dione).Here we report on three novel derivatives prepared byusing as bridging ligands p<strong>en</strong>taerythritol monoethers,H 3 L = R’-O-CH 2 C(CH 2 OH) 3 with R’=allyl (1), (R,S))-2-methyl-1-butyl (2), and S-2-methyl-1-butyl (3) along witha new polymorph the complex containing 11-(acetylthio)-2,2-bis(hydroxymethyl) undecan-1-ol ligands (4b) [Gregoliet al., Chem. Eur. J. 15, 6456 (2009)]. High-Frequ<strong>en</strong>cyEPR (HF-EPR) spectra have be<strong>en</strong> collected, at two frequ<strong>en</strong>cies(190 and 230 GHz) and several temperatures betwe<strong>en</strong> 5and 30 K, on polycrystalline samples of the four complexesin order to determine the zero-field splitting (zfs) parametersin the ground spin state. The spectra obtained show thetypical behavior of systems with an S = 5 ground spin statecharacterized by an easy-axis type anisotropy (D < 0):H = µ 0 B.g.S + DS 2 z + E(S 2 x − S 2 y) + B 0 4 O0 4 (17)where O 0 4 is a Stev<strong>en</strong>s operator, while D, E and B0 4 arethe crystal field parameters defining the anisotropy of thesystem. Setting an isotropic Landé factor g = 2.0, the bestsimulations of experim<strong>en</strong>tal spectra were obtained with thefollowing parameters (in cm −1 ): D = −0.417, E = 0.015,B 0 4 = +1.3 × 10−5 for (1a), D = −0.435, E = 0.009,B 0 4 = +0.9 × 10−5 for (1b), D = −0.449, E = 0.030,B 0 4 = +2.4 × 10−5 for (2), D = −0.442, E = 0.0312,B 0 4 = +1.65 × 10−5 for (3) and D = −0.412, E = 0.006,B 0 4 = +1.8 × 10−5 for (4b)(figure 1). (1a) and (1b) standfor the two structurally inequival<strong>en</strong>t molecules pres<strong>en</strong>t inthe cell of crystal (1). Experim<strong>en</strong>tal and calculated spectrafor complex (2) at 230 GHz are displayed in figure 141.In (1-3) and (4b), and in other six isostructural compoundspreviously reported, a remarkable correlation is found betwe<strong>en</strong>the axial zfs parameter D and the helical pitch γof the propeller-like structure (figure 142). γ is definedas the average dihedral angle betwe<strong>en</strong> the mean Fe4 planeand the three FeO 2 Fe ”bla<strong>des</strong>”.The relationship is directlydemonstrated by (1), which features both structurally andmagnetically inequival<strong>en</strong>t molecules in the crystal. Thetwo polymorphs of (4) ((4a,b)) span the whole range ofanisotropies for [Fe 4 (L) 2 (dpm) 6 ] complexes, suggestingthat crystal packing effects may be largely responsible forthe observed structural and magnetic differ<strong>en</strong>ces.The dynamics of the magnetization in the four complexeshas be<strong>en</strong> investigated by AC susceptometry, and the resultsanalyzed by master-matrix calculations. The large rhombicityof (2) and (3) is found responsible for the fast magneticrelaxation observed in the two compounds. However, complex(3) shows an additional faster relaxation mechanismwhich is unaccounted for by the set of spin-Hamiltonianparameters determined by HF-EPR.Figure 141: HF-EPR experim<strong>en</strong>tal (bold) and simulated powderspectra recorded at 230 GHz on derivative (2).Figure 142: Axial anisotropy D versus helical pitch γ for thetwelve Tetrairon(III) propellers so far characterized. The redpoints correspond to the four derivatives discussed here. The solidline correspond to the best fit for the derivatives featuring twotripodal ligands.A.L. Barra, P. NeugebauerA. Cornia, L. Gregoli, C. Dianeli (University of Mod<strong>en</strong>a, Italy), R. Sessoli (University of Flor<strong>en</strong>ce, Italy)98
- Page 1 and 2:
LABORATOIRE NATIONAL DES CHAMPS MAG
- Page 4 and 5:
TABLE OF CONTENTSPreface 1Carbon Al
- Page 6 and 7:
Coexistence of closed orbit and qua
- Page 8:
2009PrefaceDear Reader,You have bef
- Page 12 and 13:
2009 CARBON ALLOTROPESInvestigation
- Page 14 and 15:
2009 CARBON ALLOTROPESPropagative L
- Page 16 and 17:
2009 CARBON ALLOTROPESEdge fingerpr
- Page 18 and 19:
2009 CARBON ALLOTROPESObservation o
- Page 20 and 21:
2009 CARBON ALLOTROPESImproving gra
- Page 22 and 23:
2009 CARBON ALLOTROPESHow perfect c
- Page 24 and 25:
2009 CARBON ALLOTROPESTuning the el
- Page 26 and 27:
2009 CARBON ALLOTROPESElectric fiel
- Page 28 and 29:
2009 CARBON ALLOTROPESMagnetotransp
- Page 30 and 31:
2009 CARBON ALLOTROPESGraphite from
- Page 32:
2009Two-Dimensional Electron Gas25
- Page 35 and 36:
TWO-DIMENSIONAL ELECTRON GAS 2009Di
- Page 37 and 38:
TWO-DIMENSIONAL ELECTRON GAS 2009Sp
- Page 39 and 40:
TWO-DIMENSIONAL ELECTRON GAS 2009Cr
- Page 41 and 42:
TWO-DIMENSIONAL ELECTRON GAS 2009Re
- Page 43 and 44:
TWO-DIMENSIONAL ELECTRON GAS 2009In
- Page 45 and 46:
TWO-DIMENSIONAL ELECTRON GAS 2009Ho
- Page 47 and 48:
TWO-DIMENSIONAL ELECTRON GAS 2009Te
- Page 50 and 51:
2009 SEMICONDUCTORS AND NANOSTRUCTU
- Page 52 and 53: 2009 SEMICONDUCTORS AND NANOSTRUCTU
- Page 54 and 55: 2009 SEMICONDUCTORS AND NANOSTRUCTU
- Page 56 and 57: 2009 SEMICONDUCTORS AND NANOSTRUCTU
- Page 58 and 59: 2009 SEMICONDUCTORS AND NANOSTRUCTU
- Page 60: 2009Metals, Superconductors and Str
- Page 63 and 64: METALS, SUPERCONDUCTORS... 2009Anom
- Page 65 and 66: METALS, SUPERCONDUCTORS... 2009Magn
- Page 67 and 68: METALS, SUPERCONDUCTORS ... 2009Coe
- Page 69 and 70: METALS, SUPERCONDUCTORS ... 2009Fie
- Page 71 and 72: METALS, SUPERCONDUCTORS... 2009High
- Page 73 and 74: METALS, SUPERCONDUCTORS... 2009Angu
- Page 75 and 76: METALS, SUPERCONDUCTORS... 2009Magn
- Page 77 and 78: METALS, SUPERCONDUCTORS... 2009Meta
- Page 79 and 80: METALS, SUPERCONDUCTORS... 2009Temp
- Page 81 and 82: METALS, SUPERCONDUCTORS... 200974
- Page 84 and 85: 2009 MAGNETIC SYSTEMSY b 3+ → Er
- Page 86 and 87: 2009 MAGNETIC SYSTEMSMagnetotranspo
- Page 88 and 89: 2009 MAGNETIC SYSTEMSHigh field tor
- Page 90 and 91: 2009 MAGNETIC SYSTEMSNuclear magnet
- Page 92 and 93: 2009 MAGNETIC SYSTEMSStructural ana
- Page 94 and 95: 2009 MAGNETIC SYSTEMSEnhancement ma
- Page 96 and 97: 2009 MAGNETIC SYSTEMSInvestigation
- Page 98 and 99: 2009 MAGNETIC SYSTEMSField-induced
- Page 100 and 101: 2009 MAGNETIC SYSTEMSMagnetic prope
- Page 102: 2009Biology, Chemistry and Soft Mat
- Page 108 and 109: 2009 APPLIED SUPERCONDUCTIVITYMagne
- Page 110 and 111: 2009 APPLIED SUPERCONDUCTIVITYPhtha
- Page 112: 2009Magneto-Science105
- Page 115 and 116: MAGNETO-SCIENCE 2009Study of the in
- Page 117 and 118: MAGNETO-SCIENCE 2009Magnetohydrodyn
- Page 119 and 120: MAGNETO-SCIENCE 2009112
- Page 122 and 123: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 124 and 125: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 126 and 127: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 128 and 129: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 130 and 131: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 132 and 133: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 134 and 135: 2009 MAGNET DEVELOPMENT AND INSTRUM
- Page 136 and 137: 2009 PROPOSALSProposals for Magnet
- Page 138 and 139: 2009 PROPOSALSSpin-Jahn-Teller effe
- Page 140 and 141: 2009 PROPOSALSQuantum Oscillations
- Page 142 and 143: 2009 PROPOSALSThermoelectric tensor
- Page 144 and 145: 2009 PROPOSALSDr. EscoffierCyclotro
- Page 146 and 147: 2009 PROPOSALSHigh field magnetotra
- Page 148 and 149: 2009 THESESPhD Theses 20091. Nanot
- Page 150 and 151: 2009 PUBLICATIONS[21] O. Drachenko,
- Page 152 and 153: 2009 PUBLICATIONS[75] S. Nowak, T.
- Page 154 and 155:
Contributors of the LNCMI to the Pr
- Page 156 and 157:
Institut Jean Lamour, Nancy : 68Ins
- Page 158 and 159:
Lawrence Berkeley National Laborato