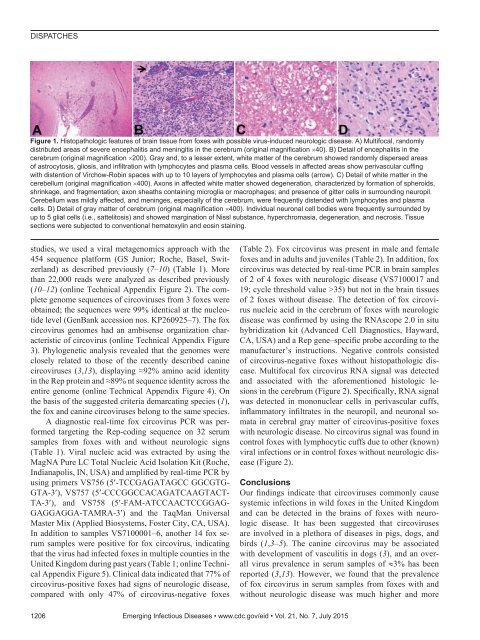
DISPATCHESFigure 1. Histopathologic features of brain tissue from foxes with possible virus-induced neurologic disease. A) Multifocal, randomlydistributed areas of severe encephalitis and meningitis in the cerebrum (original magnification ×40). B) Detail of encephalitis in thecerebrum (original magnification ×200). Gray and, to a lesser extent, white matter of the cerebrum showed randomly dispersed areasof astrocytosis, gliosis, and infiltration with lymphocytes and plasma cells. Blood vessels in affected areas show perivascular cuffingwith distention of Virchow-Robin spaces with up to 10 layers of lymphocytes and plasma cells (arrow). C) Detail of white matter in thecerebellum (original magnification ×400). Axons in affected white matter showed degeneration, characterized by formation of spheroids,shrinkage, and fragmentation; axon sheaths containing microglia or macrophages; and presence of gitter cells in surrounding neuropil.Cerebellum was mildly affected, and meninges, especially of the cerebrum, were frequently distended with lymphocytes and plasmacells. D) Detail of gray matter of cerebrum (original magnification ×400). Individual neuronal cell bodies were frequently surrounded byup to 5 glial cells (i.e., sattelitosis) and showed margination of Nissl substance, hyperchromasia, degeneration, and necrosis. Tissuesections were subjected to conventional hematoxylin and eosin staining.studies, we used a viral metagenomics approach with the454 sequence platform (GS Junior; Roche, Basel, Switzerland)as described previously (7–10) (Table 1). Morethan 22,000 reads were analyzed as described previously(10–12) (online Technical Appendix Figure 2). The completegenome sequences of circoviruses from 3 foxes wereobtained; the sequences were 99% identical at the nucleotidelevel (GenBank accession nos. KP260925–7). The foxcircovirus genomes had an ambisense organization characteristicof circovirus (online Technical Appendix Figure3). Phylogenetic analysis revealed that the genomes wereclosely related to those of the recently described caninecircoviruses (3,13), displaying ≈92% amino acid identityin the Rep protein and ≈89% nt sequence identity across theentire genome (online Technical Appendix Figure 4). Onthe basis of the suggested criteria demarcating species (1),the fox and canine circoviruses belong to the same species.A diagnostic real-time fox circovirus PCR was performedtargeting the Rep-coding sequence on 32 serumsamples from foxes with and without neurologic signs(Table 1). Viral nucleic acid was extracted by using theMagNA Pure LC Total Nucleic Acid Isolation Kit (Roche,Indianapolis, IN, USA) and amplified by real-time PCR byusing primers VS756 (5′-TCCGAGATAGCC GGCGTG-GTA-3′), VS757 (5′-CCCGGCCACAGATCAAGTACT-TA-3′), and VS758 (5′-FAM-ATCCAACTCCGGAG-GAGGAGGA-TAMRA-3′) and the TaqMan UniversalMaster Mix (Applied Biosystems, Foster City, CA, USA).In addition to samples VS7100001–6, another 14 fox serumsamples were positive for fox circovirus, indicatingthat the virus had infected foxes in multiple counties in theUnited Kingdom during past years (Table 1; online TechnicalAppendix Figure 5). Clinical data indicated that 77% ofcircovirus-positive foxes had signs of neurologic disease,compared with only 47% of circovirus-negative foxes(Table 2). Fox circovirus was present in male and femalefoxes and in adults and juveniles (Table 2). In addition, foxcircovirus was detected by real-time PCR in brain samplesof 2 of 4 foxes with neurologic disease (VS7100017 and19; cycle threshold value >35) but not in the brain tissuesof 2 foxes without disease. The detection of fox circovirusnucleic acid in the cerebrum of foxes with neurologicdisease was confirmed by using the RNAscope 2.0 in situhybridization kit (Advanced Cell Diagnostics, Hayward,CA, USA) and a Rep gene–specific probe according to themanufacturer’s instructions. Negative controls consistedof circovirus-negative foxes without histopathologic disease.Multifocal fox circovirus RNA signal was detectedand associated with the aforementioned histologic lesionsin the cerebrum (Figure 2). Specifically, RNA signalwas detected in mononuclear cells in perivascular cuffs,inflammatory infiltrates in the neuropil, and neuronal somatain cerebral gray matter of circovirus-positive foxeswith neurologic disease. No circovirus signal was found incontrol foxes with lymphocytic cuffs due to other (known)viral infections or in control foxes without neurologic disease(Figure 2).ConclusionsOur findings indicate that circoviruses commonly causesystemic infections in wild foxes in the United Kingdomand can be detected in the brains of foxes with neurologicdisease. It has been suggested that circovirusesare involved in a plethora of diseases in pigs, dogs, andbirds (1,3–5). The canine circovirus may be associatedwith development of vasculitis in dogs (3), and an overallvirus prevalence in serum samples of ≈3% has beenreported (3,13). However, we found that the prevalenceof fox circovirus in serum samples from foxes with andwithout neurologic disease was much higher and more1206 Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015
Circovirus in Foxes with MeningoencephalitisTable 1. Overview of testing results for fox serum samples used in a study of the detection of circovirus in foxes withmeningoencephalitis, United Kingdom, 2009–2013*Year serumAnimal sample obtained Age Sex County Signs† Outcome 454‡ PCR§ C t FFPEVS7100001 2013 Adult F Nor Yes Euthanized Yes Pos 16.5 YesVS7100002 2013 Adult F Ess Yes Euthanized Yes Pos 39.3 NoVS7100003 2013 Adult F Nor Yes Euthanized Yes Pos 16.4 YesVS7100004 2013 Adult M Suf Yes Released Yes Pos 35.0 NoVS7100005 2013 Adult F Nor Yes Euthanized Yes Pos 16.4 YesVS7100006 2013 Adult M Bed Yes Euthanized Yes Pos 36.9 NoVS7100010 2009 Adult F Lei Yes Euthanized No Pos 23.4 NoVS7100014 2010 Adult M Suf Yes Euthanized No Pos 14.1 YesVS7100015 2010 Adult M Lin Yes Euthanized No Pos 14.3 NoVS7100017 2011 Adult M Nor Yes Euthanized No Neg ND NoVS7100018 2011 Adult F Nor Yes Euthanized No Neg ND NoVS7100019 2011 Adult F Cam Yes Euthanized No Pos 36.8 NoVS7100021 2011 Adult M Cam Yes Euthanized No Pos 25.3 NoVS7100025 2012 Adult M Suf Yes Euthanized No Neg ND NoVS7100030 2012 Adult F Nor Yes Euthanized No Neg ND NoVS7100032 2012 Adult M Ess Yes Euthanized No Pos 38.0 NoVS7100038 2013 Adult F Suf Yes Euthanized No Pos 17.9 YesVS7100008 2007 Adult M Cam No Euthanized No Neg ND NoVS7100011 2009 Adult M Lin No Euthanized No Pos 29.7 NoVS7100012 2010 Adult M Nor No Died No Neg ND YesVS7100020 2011 Adult M Cam No Euthanized No Neg ND NoVS7100022 2012 Adult M Cam No Euthanized No Pos 39.5 NoVS7100023 2012 Adult M Cam No Died No Pos 37.3 NoVS7100024 2012 Adult F Lin No Euthanized No Neg ND NoVS7100026 2012 Juvenile F Lei No Released No Pos 14.4 NoVS7100027 2012 Juvenile F Nor No Released No Pos 14.6 NoVS7100028 2012 Juvenile F Suf No Released No Neg ND NoVS7100029 2012 Juvenile F Lin No Released No Pos 14.2 NoVS7100031 2012 Juvenile M Suf No Released No Neg ND NoVS7100033 2012 Juvenile M Nor No Euthanized No Neg ND NoVS7100035 2013 Juvenile F Cam No Released No Neg ND NoVS7100036 2013 Adult M Cam No Released No Pos 39.2 No*Bed, Bedfordshire; Cam, Cambridgeshire; Ct, cycle threshold values of real-time PCR; Ess, Essex; FFPE, formalin-fixed paraffin-embedded tissue; Lei,Leicester; Lin, Lincolnshire; ND, not determined; Neg, negative; Nor, Norfolk; Pos, positive; Suf, Suffolk.†Neurologic signs were abnormal behavior, lack of fear, reduced alertness, aimless wandering, circling, facial muscle twitching, progressive weakness ofhind legs, and visual abnormalities.‡Samples were analyzed by using a viral metagenomics approach with the 454 sequence platform (GS Junior; Roche, Basel, Switzerland).§TaqMan real-time PCR.comparable to the prevalence of porcine circovirusesamong pigs (14). No association of virus infection withvasculitis was apparent. Instead, fox circoviruses may beassociated with development of neurologic disease directlyor as a contributory complicating cofactor.Cycloviruses, which belong to a proposed new genusin the family Circoviridae, were recently found in serumand cerebrospinal fluid of humans with paraplegia andacute infections of the central nervous system (11,15), suggestingthat viruses from the family Circoviridae may haveTable 2. Univariate statistical analysis of age, sex, disease signs, and circovirus real-time PCR results for foxes in a study of thedetection of circovirus in foxes with meningoencephalitis, United Kingdom, 2009–2013*PCR results for foxes, no. (%)† p value by χ 2ParameterWithout neurologic signs With neurologic signstest OR (95% CI)All foxes 15 (46.9) 17 (53.1)SexM 9 (60.0) 8 (47.1) 0.502 1.69 (0.41–6.88)F 6 (40.0) 9 (52.9)Age, yJuvenile 7 (46.7) 0 (0) 0.002 3.13 (1.77–5.53)Adult 8 (53.3) 17 (100)Circovirus positiveNo 8 (53.3) 4 (23.5) 0.144 3.71 (0.82–16.84)Yes 7 (46.7) 13 (76.5)*OR, odds ratio.†Neurologic signs were abnormal behavior, lack of fear, reduced alertness, aimless wandering, circling, facial muscle twitching, progressive weakness ofhind legs, and visual abnormalities.Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015 1207
- Page 3 and 4:
July 2015SynopsisOn the CoverMarian
- Page 5 and 6:
1240 Gastroenteritis OutbreaksCause
- Page 7 and 8:
SYNOPSISDisseminated Infections wit
- Page 9 and 10:
Disseminated Infections with Talaro
- Page 11 and 12:
Disseminated Infections with Talaro
- Page 13 and 14:
Macacine Herpesvirus 1 inLong-Taile
- Page 15 and 16:
Macacine Herpesvirus 1 in Macaques,
- Page 17 and 18:
Macacine Herpesvirus 1 in Macaques,
- Page 19:
Macacine Herpesvirus 1 in Macaques,
- Page 23:
Malaria among Young Infants, Africa
- Page 26 and 27:
RESEARCHFigure 3. Dynamics of 19-kD
- Page 28 and 29:
Transdermal Diagnosis of MalariaUsi
- Page 30 and 31:
RESEARCHFigure 2. A) Acoustic trace
- Page 32 and 33:
RESEARCHof malaria-infected mosquit
- Page 34 and 35:
Lack of Transmission amongClose Con
- Page 36 and 37:
RESEARCH(IFA) and microneutralizati
- Page 38 and 39:
RESEARCHoropharyngeal, and serum sa
- Page 40 and 41:
RESEARCH6. Assiri A, McGeer A, Perl
- Page 42 and 43:
RESEARCHadvanced genomic sequencing
- Page 44 and 45:
RESEARCHTable 2. Next-generation se
- Page 46 and 47:
RESEARCHTable 3. Mutation analysis
- Page 48 and 49:
RESEARCHReferences1. Baize S, Panne
- Page 50 and 51:
Parechovirus Genotype 3 Outbreakamo
- Page 52 and 53:
RESEARCHFigure 1. Venn diagramshowi
- Page 54 and 55:
RESEARCHTable 2. HPeV testing of sp
- Page 56 and 57:
RESEARCHFigure 5. Distribution of h
- Page 58 and 59:
RESEARCHReferences1. Selvarangan R,
- Page 60 and 61:
RESEARCHthe left lobe was sampled b
- Page 62 and 63: RESEARCHTable 2. Middle East respir
- Page 64 and 65: RESEARCHseroprevalence in domestic
- Page 66 and 67: RESEARCHmeasure their current surve
- Page 68 and 69: RESEARCHTable 2. States with labora
- Page 70 and 71: RESEARCHFigure 2. Comparison of sur
- Page 72 and 73: RESEARCH9. Centers for Disease Cont
- Page 74 and 75: RESEARCHthe analyses. Cases in pers
- Page 76 and 77: RESEARCHTable 3. Sampling results (
- Page 78 and 79: RESEARCHpresence of Legionella spp.
- Page 80 and 81: Seroprevalence for Hepatitis Eand O
- Page 82 and 83: RESEARCHTable 1. Description of stu
- Page 84 and 85: RESEARCHTable 3. Crude and adjusted
- Page 86 and 87: RESEARCHrates by gender or HIV stat
- Page 88 and 89: RESEARCH25. Taha TE, Kumwenda N, Ka
- Page 90 and 91: POLICY REVIEWDutch Consensus Guidel
- Page 92 and 93: POLICY REVIEWTable 3. Comparison of
- Page 94 and 95: POLICY REVIEW6. Botelho-Nevers E, F
- Page 96 and 97: DISPATCHESFigure 1. Phylogenetic tr
- Page 98 and 99: DISPATCHESSevere Pediatric Adenovir
- Page 100 and 101: DISPATCHESTable 1. Demographics and
- Page 102 and 103: DISPATCHES13. Kim YJ, Hong JY, Lee
- Page 104 and 105: DISPATCHESTable. Alignment of resid
- Page 106 and 107: DISPATCHESFigure 2. Interaction of
- Page 108 and 109: DISPATCHESSchmallenberg Virus Recur
- Page 110 and 111: DISPATCHESFigure 2. Detection of Sc
- Page 114: DISPATCHESFigure 2. Detection of fo
- Page 117 and 118: Influenza Virus Strains in the Amer
- Page 119 and 120: Novel Arenavirus Isolates from Nama
- Page 121 and 122: Novel Arenaviruses, Southern Africa
- Page 123 and 124: Readability of Ebola Informationon
- Page 125 and 126: Readability of Ebola Information on
- Page 127 and 128: Patients under investigation for ME
- Page 129 and 130: Patients under investigation for ME
- Page 131 and 132: Wildlife Reservoir for Hepatitis E
- Page 133 and 134: Asymptomatic Malaria and Other Infe
- Page 135 and 136: Asymptomatic Malaria in Children fr
- Page 137 and 138: Bufavirus in Wild Shrews and Nonhum
- Page 139 and 140: Bufavirus in Wild Shrews and Nonhum
- Page 141 and 142: Range Expansion for Rat Lungworm in
- Page 143 and 144: Slow Clearance of Plasmodium falcip
- Page 145 and 146: Slow Clearance of Plasmodium falcip
- Page 147 and 148: Gastroenteritis Caused by Norovirus
- Page 149 and 150: Ebola Virus Stability on Surfaces a
- Page 151 and 152: Ebola Virus Stability on Surfaces a
- Page 153 and 154: Outbreak of Ciprofloxacin-Resistant
- Page 155 and 156: Outbreak of S. sonnei, South KoreaT
- Page 157 and 158: Rapidly Expanding Range of Highly P
- Page 159 and 160: Cluster of Ebola Virus Disease, Bon
- Page 161 and 162: Cluster of Ebola Virus Disease, Lib
- Page 163 and 164:
ANOTHER DIMENSIONThe Past Is Never
- Page 165 and 166:
Measles Epidemic, Boston, Massachus
- Page 167 and 168:
LETTERSInfluenza A(H5N6)Virus Reass
- Page 169 and 170:
LETTERSsystem (8 kb-span paired-end
- Page 171 and 172:
LETTERS3. Van Hong N, Amambua-Ngwa
- Page 173 and 174:
LETTERSTable. Prevalence of Bartone
- Page 175 and 176:
LETTERSavian influenza A(H5N1) viru
- Page 177 and 178:
LETTERSprovinces and a total of 200
- Page 179 and 180:
LETTERS7. Manian FA. Bloodstream in
- Page 181 and 182:
LETTERSforward projections. N Engl
- Page 183 and 184:
LETTERS3. Guindon S, Gascuel OA. Si
- Page 185 and 186:
BOOKS AND MEDIAin the port cities o
- Page 187 and 188:
ABOUT THE COVERNorth was not intere
- Page 189 and 190:
Earning CME CreditTo obtain credit,
- Page 191:
Emerging Infectious Diseases is a p