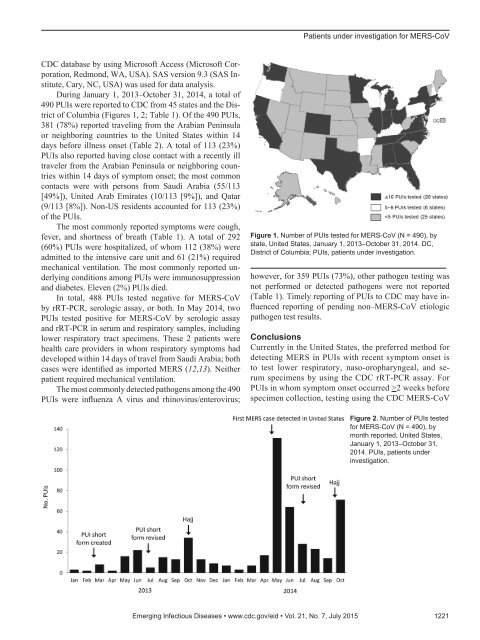
DISPATCHESEvaluation of Patients under Investigationfor MERS-CoV Infection, United States,January 2013–October 2014Eileen Schneider, Christina Chommanard,Jessica Rudd, Brett Whitaker, Luis Lowe,Susan I. GerberMiddle East respiratory syndrome (MERS) cases continueto be reported from the Middle East. Evaluation and testingof patients under investigation (PUIs) for MERS are recommended.In 2013–2014, two imported cases were detectedamong 490 US PUIs. Continued awareness is neededfor early case detection and implementation of infectioncontrol measures.Middle East respiratory syndrome coronavirus(MERS-CoV) infection was first reported in September2012 in a patient with fatal pneumonia in Saudi Arabia(1). Subsequent investigation showed that earlier MERS-CoV infection had occurred in Jordan in April 2012 amonga cluster of patients with pneumonia (2,3). As of February5, 2015, the World Health Organization had reported 971laboratory-confirmed cases worldwide and at least 356 relateddeaths (4). All known reported cases have been linkeddirectly or indirectly to the Middle East region; most havebeen reported by Saudi Arabia and the United Arab Emirates.Typically, the initial symptoms for MERS patientsseeking medical care are fever, chills, cough, shortness ofbreath, and myalgia. These symptoms often progress to severelower respiratory tract infection, which may requiremechanical ventilation and intensive care (5,6). Several asymptomaticor mild MERS cases have been reported (7),particularly in healthy young adults. Little is known abouttransmission routes, virus shedding, risk factors, and animalreservoirs, although bats and camels have been implicatedin transmission and as reservoirs (8,9). Clusters ofhuman-to-human transmission have been associated withhousehold and health care settings (2,3,5).Using World Health Organization guidelines and definitions(4), CDC developed guidance for evaluating a patientunder investigation (PUI) for MERS-CoV infection,collecting specimens, conducting laboratory testing, andmanaging infection control (http://www.cdc.gov/coronavirus/mers/index.html).The PUI guidance was created to assisthealth care providers determine which patients shouldAuthor affiliation: Centers for Disease Control and Prevention,Atlanta, Georgia, USADOI: http://dx.doi.org/10.3201/eid2107.141888be considered for MERS-CoV evaluation and testing. Toinform state and local health departments of the basic demographicand clinical characteristics of PUIs and on assayuse, we summarized the descriptive analysis of PUIs in theUnited States.The StudyIn October 2012, CDC developed real-time reverse transcriptionPCR (rRT-PCR) assays for detection of MERS-CoV (10). CDC initially performed the testing, but on June5, 2013, a Food and Drug Administration–issued EmergencyUse Authorization allowed for assay deployment in a kitto laboratories through the Laboratory Response Network.As of March 12, 2015, a total of 47 states and the Districtof Columbia had MERS-CoV testing capability. The assaykit is intended for detection of MERS-CoV RNA inrespiratory, serum, and stool samples. CDC also developedserologic tests for detecting MERS-CoV antibodies;these tests have been used by CDC since the summer of2013. Because MERS-CoV is an emerging pathogen, CDCguidelines and guidance regarding PUI characteristics areperiodically updated as new MERS-CoV information andrisk factors are identified. CDC recommends evaluatingand testing PUIs for MERS-CoV and for other commonrespiratory pathogens.On January 1, 2013, CDC began collecting data onPUIs for MERS-CoV infection. Health care providers forpersons suspected of having MERS were to contact theirstate or local health department for consultation and to arrangefor MERS-CoV testing, if indicated. PUIs were reportedto CDC through state and local health departmentsby using the single-page PUI short form, which containsno personal identifiers (11). Since its implementation, theshort form has been revised 3 times to reflect modificationsto the PUI guidance. The short form collects informationon basic demographic data, symptoms, disease severity,hospitalization, travel history, risk factors, and laboratorytest results at the time of MERS-CoV testing. Follow-updata collection on missing information was not routinelyperformed. At least 370 (76%) PUIs met the guidance characteristicsfor PUI for MERS. The remaining 120 (24%)PUIs had incomplete clinical or travel data; the most commonmissing information was pneumonia data for personswith respiratory symptoms and a recent travel history. Theshort form was sent electronically to CDC by secure fax oremail. Data collected on the short form was entered into a1220 Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015
Patients under investigation for MERS-CoVCDC database by using Microsoft Access (Microsoft Corporation,Redmond, WA, USA). SAS <strong>version</strong> 9.3 (SAS Institute,Cary, NC, USA) was used for data analysis.During January 1, 2013–October 31, 2014, a total of490 PUIs were reported to CDC from 45 states and the Districtof Columbia (Figures 1, 2; Table 1). Of the 490 PUIs,381 (78%) reported traveling from the Arabian Peninsulaor neighboring countries to the United States within 14days before illness onset (Table 2). A total of 113 (23%)PUIs also reported having close contact with a recently illtraveler from the Arabian Peninsula or neighboring countrieswithin 14 days of symptom onset; the most commoncontacts were with persons from Saudi Arabia (55/113[49%]), United Arab Emirates (10/113 [9%]), and Qatar(9/113 [8%]). Non-US residents accounted for 113 (23%)of the PUIs.The most commonly reported symptoms were cough,fever, and shortness of breath (Table 1). A total of 292(60%) PUIs were hospitalized, of whom 112 (38%) wereadmitted to the intensive care unit and 61 (21%) requiredmechanical ventilation. The most commonly reported underlyingconditions among PUIs were immunosuppressionand diabetes. Eleven (2%) PUIs died.In total, 488 PUIs tested negative for MERS-CoVby rRT-PCR, serologic assay, or both. In May 2014, twoPUIs tested positive for MERS-CoV by serologic assayand rRT-PCR in serum and respiratory samples, includinglower respiratory tract specimens. These 2 patients werehealth care providers in whom respiratory symptoms haddeveloped within 14 days of travel from Saudi Arabia; bothcases were identified as imported MERS (12,13). Neitherpatient required mechanical ventilation.The most commonly detected pathogens among the 490PUIs were influenza A virus and rhinovirus/enterovirus;Figure 1. Number of PUIs tested for MERS-CoV (N = 490), bystate, United States, January 1, 2013–October 31, 2014. DC,District of Columbia; PUIs, patients under investigation.however, for 359 PUIs (73%), other pathogen testing wasnot performed or detected pathogens were not reported(Table 1). Timely reporting of PUIs to CDC may have influencedreporting of pending non–MERS-CoV etiologicpathogen test results.ConclusionsCurrently in the United States, the preferred method fordetecting MERS in PUIs with recent symptom onset isto test lower respiratory, naso-oropharyngeal, and serumspecimens by using the CDC rRT-PCR assay. ForPUIs in whom symptom onset occurred >2 weeks beforespecimen collection, testing using the CDC MERS-CoVFigure 2. Number of PUIs testedfor MERS-CoV (N = 490), bymonth reported, United States,January 1, 2013–October 31,2014. PUIs, patients underinvestigation.Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015 1221
- Page 3 and 4:
July 2015SynopsisOn the CoverMarian
- Page 5 and 6:
1240 Gastroenteritis OutbreaksCause
- Page 7 and 8:
SYNOPSISDisseminated Infections wit
- Page 9 and 10:
Disseminated Infections with Talaro
- Page 11 and 12:
Disseminated Infections with Talaro
- Page 13 and 14:
Macacine Herpesvirus 1 inLong-Taile
- Page 15 and 16:
Macacine Herpesvirus 1 in Macaques,
- Page 17 and 18:
Macacine Herpesvirus 1 in Macaques,
- Page 19:
Macacine Herpesvirus 1 in Macaques,
- Page 23:
Malaria among Young Infants, Africa
- Page 26 and 27:
RESEARCHFigure 3. Dynamics of 19-kD
- Page 28 and 29:
Transdermal Diagnosis of MalariaUsi
- Page 30 and 31:
RESEARCHFigure 2. A) Acoustic trace
- Page 32 and 33:
RESEARCHof malaria-infected mosquit
- Page 34 and 35:
Lack of Transmission amongClose Con
- Page 36 and 37:
RESEARCH(IFA) and microneutralizati
- Page 38 and 39:
RESEARCHoropharyngeal, and serum sa
- Page 40 and 41:
RESEARCH6. Assiri A, McGeer A, Perl
- Page 42 and 43:
RESEARCHadvanced genomic sequencing
- Page 44 and 45:
RESEARCHTable 2. Next-generation se
- Page 46 and 47:
RESEARCHTable 3. Mutation analysis
- Page 48 and 49:
RESEARCHReferences1. Baize S, Panne
- Page 50 and 51:
Parechovirus Genotype 3 Outbreakamo
- Page 52 and 53:
RESEARCHFigure 1. Venn diagramshowi
- Page 54 and 55:
RESEARCHTable 2. HPeV testing of sp
- Page 56 and 57:
RESEARCHFigure 5. Distribution of h
- Page 58 and 59:
RESEARCHReferences1. Selvarangan R,
- Page 60 and 61:
RESEARCHthe left lobe was sampled b
- Page 62 and 63:
RESEARCHTable 2. Middle East respir
- Page 64 and 65:
RESEARCHseroprevalence in domestic
- Page 66 and 67:
RESEARCHmeasure their current surve
- Page 68 and 69:
RESEARCHTable 2. States with labora
- Page 70 and 71:
RESEARCHFigure 2. Comparison of sur
- Page 72 and 73:
RESEARCH9. Centers for Disease Cont
- Page 74 and 75:
RESEARCHthe analyses. Cases in pers
- Page 76 and 77: RESEARCHTable 3. Sampling results (
- Page 78 and 79: RESEARCHpresence of Legionella spp.
- Page 80 and 81: Seroprevalence for Hepatitis Eand O
- Page 82 and 83: RESEARCHTable 1. Description of stu
- Page 84 and 85: RESEARCHTable 3. Crude and adjusted
- Page 86 and 87: RESEARCHrates by gender or HIV stat
- Page 88 and 89: RESEARCH25. Taha TE, Kumwenda N, Ka
- Page 90 and 91: POLICY REVIEWDutch Consensus Guidel
- Page 92 and 93: POLICY REVIEWTable 3. Comparison of
- Page 94 and 95: POLICY REVIEW6. Botelho-Nevers E, F
- Page 96 and 97: DISPATCHESFigure 1. Phylogenetic tr
- Page 98 and 99: DISPATCHESSevere Pediatric Adenovir
- Page 100 and 101: DISPATCHESTable 1. Demographics and
- Page 102 and 103: DISPATCHES13. Kim YJ, Hong JY, Lee
- Page 104 and 105: DISPATCHESTable. Alignment of resid
- Page 106 and 107: DISPATCHESFigure 2. Interaction of
- Page 108 and 109: DISPATCHESSchmallenberg Virus Recur
- Page 110 and 111: DISPATCHESFigure 2. Detection of Sc
- Page 112 and 113: DISPATCHESFigure 1. Histopathologic
- Page 114: DISPATCHESFigure 2. Detection of fo
- Page 117 and 118: Influenza Virus Strains in the Amer
- Page 119 and 120: Novel Arenavirus Isolates from Nama
- Page 121 and 122: Novel Arenaviruses, Southern Africa
- Page 123 and 124: Readability of Ebola Informationon
- Page 125: Readability of Ebola Information on
- Page 129 and 130: Patients under investigation for ME
- Page 131 and 132: Wildlife Reservoir for Hepatitis E
- Page 133 and 134: Asymptomatic Malaria and Other Infe
- Page 135 and 136: Asymptomatic Malaria in Children fr
- Page 137 and 138: Bufavirus in Wild Shrews and Nonhum
- Page 139 and 140: Bufavirus in Wild Shrews and Nonhum
- Page 141 and 142: Range Expansion for Rat Lungworm in
- Page 143 and 144: Slow Clearance of Plasmodium falcip
- Page 145 and 146: Slow Clearance of Plasmodium falcip
- Page 147 and 148: Gastroenteritis Caused by Norovirus
- Page 149 and 150: Ebola Virus Stability on Surfaces a
- Page 151 and 152: Ebola Virus Stability on Surfaces a
- Page 153 and 154: Outbreak of Ciprofloxacin-Resistant
- Page 155 and 156: Outbreak of S. sonnei, South KoreaT
- Page 157 and 158: Rapidly Expanding Range of Highly P
- Page 159 and 160: Cluster of Ebola Virus Disease, Bon
- Page 161 and 162: Cluster of Ebola Virus Disease, Lib
- Page 163 and 164: ANOTHER DIMENSIONThe Past Is Never
- Page 165 and 166: Measles Epidemic, Boston, Massachus
- Page 167 and 168: LETTERSInfluenza A(H5N6)Virus Reass
- Page 169 and 170: LETTERSsystem (8 kb-span paired-end
- Page 171 and 172: LETTERS3. Van Hong N, Amambua-Ngwa
- Page 173 and 174: LETTERSTable. Prevalence of Bartone
- Page 175 and 176: LETTERSavian influenza A(H5N1) viru
- Page 177 and 178:
LETTERSprovinces and a total of 200
- Page 179 and 180:
LETTERS7. Manian FA. Bloodstream in
- Page 181 and 182:
LETTERSforward projections. N Engl
- Page 183 and 184:
LETTERS3. Guindon S, Gascuel OA. Si
- Page 185 and 186:
BOOKS AND MEDIAin the port cities o
- Page 187 and 188:
ABOUT THE COVERNorth was not intere
- Page 189 and 190:
Earning CME CreditTo obtain credit,
- Page 191:
Emerging Infectious Diseases is a p