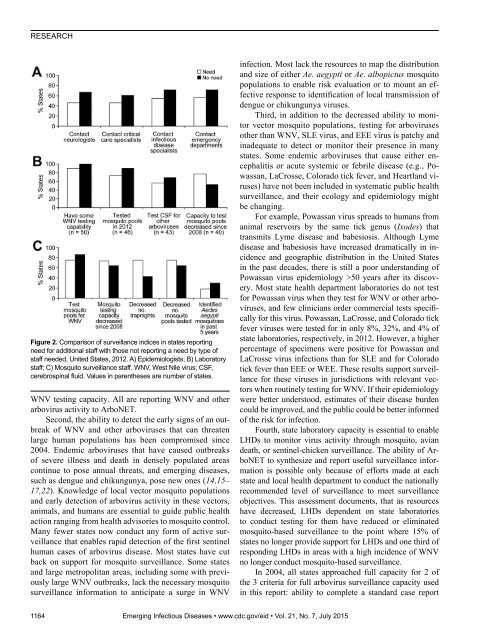
RESEARCHFigure 2. Comparison of surveillance indices in states reportingneed for additional staff with those not reporting a need by type ofstaff needed, United States, 2012. A) Epidemiologists; B) Laboratorystaff; C) Mosquito surveillance staff. WNV, West Nile virus; CSF,cerebrospinal fluid. Values in parentheses are number of states.WNV testing capacity. All are reporting WNV and otherarbovirus activity to ArboNET.Second, the ability to detect the early signs of an outbreakof WNV and other arboviruses that can threatenlarge human populations has been compromised since2004. Endemic arboviruses that have caused outbreaksof severe illness and death in densely populated areascontinue to pose annual threats, and emerging diseases,such as dengue and chikungunya, pose new ones (14,15–17,22). Knowledge of local vector mosquito populationsand early detection of arbovirus activity in these vectors,animals, and humans are essential to guide public healthaction ranging from health advisories to mosquito control.Many fewer states now conduct any form of active surveillancethat enables rapid detection of the first sentinelhuman cases of arbovirus disease. Most states have cutback on support for mosquito surveillance. Some statesand large metropolitan areas, including some with previouslylarge WNV outbreaks, lack the necessary mosquitosurveillance information to anticipate a surge in WNVinfection. Most lack the resources to map the distributionand size of either Ae. aegypti or Ae. albopictus mosquitopopulations to enable risk evaluation or to mount an effectiveresponse to identification of local transmission ofdengue or chikungunya viruses.Third, in addition to the decreased ability to monitorvector mosquito populations, testing for arbovirusesother than WNV, SLE virus, and EEE virus is patchy andinadequate to detect or monitor their presence in manystates. Some endemic arboviruses that cause either encephalitisor acute systemic or febrile disease (e.g., Powassan,LaCrosse, Colorado tick fever, and Heartland viruses)have not been included in systematic public healthsurveillance, and their ecology and epidemiology mightbe changing.For example, Powassan virus spreads to humans fromanimal reservoirs by the same tick genus (Ixodes) thattransmits Lyme disease and babesiosis. Although Lymedisease and babesiosis have increased dramatically in incidenceand geographic distribution in the United Statesin the past decades, there is still a poor understanding ofPowassan virus epidemiology >50 years after its discovery.Most state health department laboratories do not testfor Powassan virus when they test for WNV or other arboviruses,and few clinicians order commercial tests specificallyfor this virus. Powassan, LaCrosse, and Colorado tickfever viruses were tested for in only 8%, 32%, and 4% ofstate laboratories, respectively, in 2012. However, a higherpercentage of specimens were positive for Powassan andLaCrosse virus infections than for SLE and for Coloradotick fever than EEE or WEE. These results support surveillancefor these viruses in jurisdictions with relevant vectorswhen routinely testing for WNV. If their epidemiologywere better understood, estimates of their disease burdencould be improved, and the public could be better informedof the risk for infection.Fourth, state laboratory capacity is essential to enableLHDs to monitor virus activity through mosquito, aviandeath, or sentinel-chicken surveillance. The ability of ArboNETto synthesize and report useful surveillance informationis possible only because of efforts made at eachstate and local health department to conduct the nationallyrecommended level of surveillance to meet surveillanceobjectives. This assessment documents, that as resourceshave decreased, LHDs dependent on state laboratoriesto conduct testing for them have reduced or eliminatedmosquito-based surveillance to the point where 15% ofstates no longer provide support for LHDs and one third ofresponding LHDs in areas with a high incidence of WNVno longer conduct mosquito-based surveillance.In 2004, all states approached full capacity for 2 ofthe 3 criteria for full arbovirus surveillance capacity usedin this report: ability to complete a standard case report1164 Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015
Arbovirus Surveillance in the United Statesform on every suspected/confirmed mosquito-borne arboviraldisease case and report it to ArboNET and having anenvironmental surveillance system that includes mosquitosurveillance “to routinely monitor arboviral activity in allparts of the jurisdiction in which there is the potential forhuman outbreaks of arboviral disease based on past experience.”In 2012, although the first criterion continued to bemet, the second criteria was no longer met. Although the2004 assessment did not measure the ability to test for IgMfor all relevant arboviruses (including dengue viruses) onany CSF or serum specimen submitted to the state or city/county laboratory on a suspected case of arboviral disease,this assessment found that many states are not meeting thisremaining criterion.This assessment has several major limitations. First,not all jurisdictions answered all questions. Second, additionalpersonnel needs were based on state and local healthdepartment self-assessment and are subjective. In addition,because of the way the assessment was worded andresponded to, we assumed states not specifying a need foradditional personnel had no need. Thus, results showingthat states that identified a need also performed far fewersurveillance activities than states with no reported additionalneed are subject to possible inaccuracies in this assumption.Third, the relative role of different surveillancemethods shifted between 2004 and 2012. Whereas needsfor human surveillance and laboratory testing capabilityand capacity are largely unchanged, the need for aviandeath and equine surveillance data in many jurisdictionshas decreased, but the need for mosquito surveillance datahas increased. Jurisdictions have adjusted resources to accommodatethese changes. This adjustment may explain,in part, the generally high US WNV surveillance capacity,despite federal funding cuts of more than 50%. Fourth,measures of workload and staffing need may be difficultto compare among years because they depend, in part, onlevels of WNV activity. The human WNV burden in 2012was more than double that in 2004, which may have influencedestimates of need. Finally, the 2012 assessment didnot solicit information on funding or unmet needs for anythingother than staff. For example, limited fiscal resourcesmight preclude purchase of updated laboratory equipmentand testing reagents, thereby limiting laboratory testingof mosquito pools and testing of human and nonhumanspecimens for arboviruses other than WNV. Unmet nonpersonnelneeds might have contributed to loss of arbovirussurveillance capacity and would need to be addressed inany effort to maintain or improve it.In summary, WNV emergence in the United Statesstimulated building of a robust national arbovirus surveillancesystem with human and vector early detection componentsand laboratory services. This system, althoughstill highly functional, has become less robust and mightbe near a large-scale tipping point, especially in areas ofvector surveillance and laboratory support for human diagnosticand mosquito testing. Already, arboviral surveillanceis inadequate in many states to rapidly detect andcontrol outbreaks and to give the public the critical informationit needs for prevention.AcknowledgmentsWe thank the workgroup for developing the assessment tooland providing valuable insights on interpretation of findings. Inaddition to the authors, workgroup members are Jane Getchelland Kelly Wroblewski (Association of Public Health Laboratories),James Blumenstock and Abraham Kulungara (Associationof State and Territorial Health Officials), and Alfred DeMaria,Catherine Brown, Carina Blackmore, and Jennifer Lemmings(CSTE). We also thank Kimberly Miller, Rebecca Rutledge andJessica Wurster for assistance with data analysis.This study was supported by Cooperative Agreement no.1U38HM000414-05 from the Centers for Disease Control andPrevention. K.B. is listed as principal investigator on the ELCcooperative agreement for Oklahoma.Dr. Hadler is clinical professor of epidemiology and public healthat Yale School of Public Health and consultant to the New YorkCity Department of Health and Mental Hygiene and to CSTE. Hismain research interests include the epidemiology and preventionof infectious diseases, disease surveillance, and health disparities.References1. Hadler JL, Patel D, Bradley K, Hughes JM, Blackmore C, Etkind P,et al. National capacity for surveillance, prevention, and controlof West Nile Virus and other arbovirus infections—United States,2004 and 2012. MMWR Morb Mortal Wkly Rep. 2014;63:281–4.2. Centers for Disease Control and Prevention. Arboviral infections ofthe central nervous system—United States, 1996–1997.MMWR Morb Mortal Wkly Rep. 1998;47:517–22.3. Centers for Disease Control and Prevention. Outbreak of WestNile–like viral encephalitis—New York. MMWR Morb MortalWkly Rep. 1999;48:845–9.4. Centers for Disease Control and Prevention. Guidelines forsurveillance, prevention, and control of West Nile virus infection—United States. MMWR Morb Mortal Wkly Rep. 2000;49:25–8.5. Centers for Disease Control and Prevention. Epidemic/epizooticWest Nile virus in the United States: guidelines forsurveillance, prevention, and control. 3rd revision, 2003[cited 2014 Apr 25]. http://www.michigan.gov/documents/wnv-guidelines-aug-2003_87983_7.<strong>pdf</strong>6. Petersen LR, Hayes EB. Westward ho?—The spread ofWest Nile virus. N Engl J Med. 2004;351:2257–9.http://dx.doi.org/10.1056/NEJMp0482617. Pealer LN, Marfin AA, Petersen LR, Lanciotti RS, Page PL,Stramer SL, et al. Transmission of West Nile virus throughblood transfusion in the United States in 2002. N Engl J Med.2003;349:1236–45. http://dx.doi.org/10.1056/NEJMoa0309698. Iwamoto M, Jernigan DB, Guasch A, Trepka MJ, Blackmore CG,Hellinger WC, et al; The West Nile Virus in Transplant RecipientsInvestigation Team. Transmission of West Nile virus from an organdonor to four transplant recipients. N Engl J Med. 2003;348:2196–203. http://dx.doi.org/10.1056/NEJMoa022987Emerging Infectious Diseases • www.cdc.gov/eid • Vol. 21, No. 7, July 2015 1165
- Page 3 and 4:
July 2015SynopsisOn the CoverMarian
- Page 5 and 6:
1240 Gastroenteritis OutbreaksCause
- Page 7 and 8:
SYNOPSISDisseminated Infections wit
- Page 9 and 10:
Disseminated Infections with Talaro
- Page 11 and 12:
Disseminated Infections with Talaro
- Page 13 and 14:
Macacine Herpesvirus 1 inLong-Taile
- Page 15 and 16:
Macacine Herpesvirus 1 in Macaques,
- Page 17 and 18:
Macacine Herpesvirus 1 in Macaques,
- Page 19: Macacine Herpesvirus 1 in Macaques,
- Page 23: Malaria among Young Infants, Africa
- Page 26 and 27: RESEARCHFigure 3. Dynamics of 19-kD
- Page 28 and 29: Transdermal Diagnosis of MalariaUsi
- Page 30 and 31: RESEARCHFigure 2. A) Acoustic trace
- Page 32 and 33: RESEARCHof malaria-infected mosquit
- Page 34 and 35: Lack of Transmission amongClose Con
- Page 36 and 37: RESEARCH(IFA) and microneutralizati
- Page 38 and 39: RESEARCHoropharyngeal, and serum sa
- Page 40 and 41: RESEARCH6. Assiri A, McGeer A, Perl
- Page 42 and 43: RESEARCHadvanced genomic sequencing
- Page 44 and 45: RESEARCHTable 2. Next-generation se
- Page 46 and 47: RESEARCHTable 3. Mutation analysis
- Page 48 and 49: RESEARCHReferences1. Baize S, Panne
- Page 50 and 51: Parechovirus Genotype 3 Outbreakamo
- Page 52 and 53: RESEARCHFigure 1. Venn diagramshowi
- Page 54 and 55: RESEARCHTable 2. HPeV testing of sp
- Page 56 and 57: RESEARCHFigure 5. Distribution of h
- Page 58 and 59: RESEARCHReferences1. Selvarangan R,
- Page 60 and 61: RESEARCHthe left lobe was sampled b
- Page 62 and 63: RESEARCHTable 2. Middle East respir
- Page 64 and 65: RESEARCHseroprevalence in domestic
- Page 66 and 67: RESEARCHmeasure their current surve
- Page 68 and 69: RESEARCHTable 2. States with labora
- Page 72 and 73: RESEARCH9. Centers for Disease Cont
- Page 74 and 75: RESEARCHthe analyses. Cases in pers
- Page 76 and 77: RESEARCHTable 3. Sampling results (
- Page 78 and 79: RESEARCHpresence of Legionella spp.
- Page 80 and 81: Seroprevalence for Hepatitis Eand O
- Page 82 and 83: RESEARCHTable 1. Description of stu
- Page 84 and 85: RESEARCHTable 3. Crude and adjusted
- Page 86 and 87: RESEARCHrates by gender or HIV stat
- Page 88 and 89: RESEARCH25. Taha TE, Kumwenda N, Ka
- Page 90 and 91: POLICY REVIEWDutch Consensus Guidel
- Page 92 and 93: POLICY REVIEWTable 3. Comparison of
- Page 94 and 95: POLICY REVIEW6. Botelho-Nevers E, F
- Page 96 and 97: DISPATCHESFigure 1. Phylogenetic tr
- Page 98 and 99: DISPATCHESSevere Pediatric Adenovir
- Page 100 and 101: DISPATCHESTable 1. Demographics and
- Page 102 and 103: DISPATCHES13. Kim YJ, Hong JY, Lee
- Page 104 and 105: DISPATCHESTable. Alignment of resid
- Page 106 and 107: DISPATCHESFigure 2. Interaction of
- Page 108 and 109: DISPATCHESSchmallenberg Virus Recur
- Page 110 and 111: DISPATCHESFigure 2. Detection of Sc
- Page 112 and 113: DISPATCHESFigure 1. Histopathologic
- Page 114: DISPATCHESFigure 2. Detection of fo
- Page 117 and 118: Influenza Virus Strains in the Amer
- Page 119 and 120: Novel Arenavirus Isolates from Nama
- Page 121 and 122:
Novel Arenaviruses, Southern Africa
- Page 123 and 124:
Readability of Ebola Informationon
- Page 125 and 126:
Readability of Ebola Information on
- Page 127 and 128:
Patients under investigation for ME
- Page 129 and 130:
Patients under investigation for ME
- Page 131 and 132:
Wildlife Reservoir for Hepatitis E
- Page 133 and 134:
Asymptomatic Malaria and Other Infe
- Page 135 and 136:
Asymptomatic Malaria in Children fr
- Page 137 and 138:
Bufavirus in Wild Shrews and Nonhum
- Page 139 and 140:
Bufavirus in Wild Shrews and Nonhum
- Page 141 and 142:
Range Expansion for Rat Lungworm in
- Page 143 and 144:
Slow Clearance of Plasmodium falcip
- Page 145 and 146:
Slow Clearance of Plasmodium falcip
- Page 147 and 148:
Gastroenteritis Caused by Norovirus
- Page 149 and 150:
Ebola Virus Stability on Surfaces a
- Page 151 and 152:
Ebola Virus Stability on Surfaces a
- Page 153 and 154:
Outbreak of Ciprofloxacin-Resistant
- Page 155 and 156:
Outbreak of S. sonnei, South KoreaT
- Page 157 and 158:
Rapidly Expanding Range of Highly P
- Page 159 and 160:
Cluster of Ebola Virus Disease, Bon
- Page 161 and 162:
Cluster of Ebola Virus Disease, Lib
- Page 163 and 164:
ANOTHER DIMENSIONThe Past Is Never
- Page 165 and 166:
Measles Epidemic, Boston, Massachus
- Page 167 and 168:
LETTERSInfluenza A(H5N6)Virus Reass
- Page 169 and 170:
LETTERSsystem (8 kb-span paired-end
- Page 171 and 172:
LETTERS3. Van Hong N, Amambua-Ngwa
- Page 173 and 174:
LETTERSTable. Prevalence of Bartone
- Page 175 and 176:
LETTERSavian influenza A(H5N1) viru
- Page 177 and 178:
LETTERSprovinces and a total of 200
- Page 179 and 180:
LETTERS7. Manian FA. Bloodstream in
- Page 181 and 182:
LETTERSforward projections. N Engl
- Page 183 and 184:
LETTERS3. Guindon S, Gascuel OA. Si
- Page 185 and 186:
BOOKS AND MEDIAin the port cities o
- Page 187 and 188:
ABOUT THE COVERNorth was not intere
- Page 189 and 190:
Earning CME CreditTo obtain credit,
- Page 191:
Emerging Infectious Diseases is a p