Protein Expression and Purification Series - Bio-Rad
Protein Expression and Purification Series - Bio-Rad
Protein Expression and Purification Series - Bio-Rad
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
CHAPTER 4<br />
11 ml CULTURE<br />
PROTOCOL<br />
<strong>Protein</strong> <strong>Expression</strong> <strong>and</strong> <strong>Purification</strong> <strong>Series</strong><br />
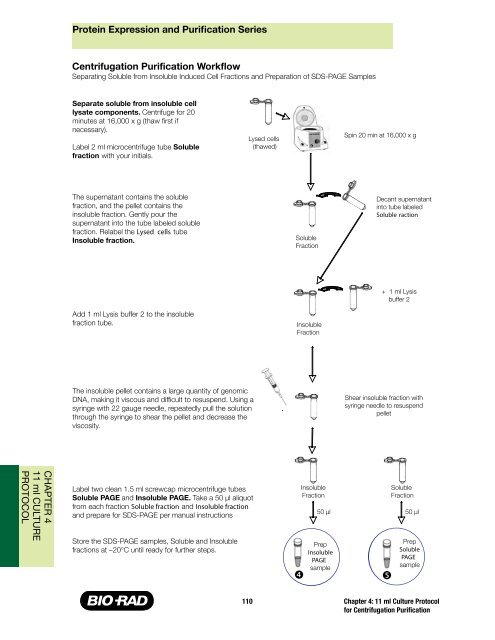
Centrifugation <strong>Purification</strong> Workflow<br />
Separating Soluble from Insoluble Induced Cell Fractions <strong>and</strong> Preparation of SDS-PAGE Samples<br />
Separate soluble from insoluble cell<br />
lysate components. Centrifuge for 20<br />
minutes at 16,000 x g (thaw first if<br />
necessary).<br />
Label 2 ml microcentrifuge tube Soluble<br />
fraction with your initials.<br />
The supernatant contains the soluble<br />
fraction, <strong>and</strong> the pellet contains the<br />
insoluble fraction. Gently pour the<br />
supernatant into the tube labeled soluble<br />
fraction. Relabel the Lysed cells tube<br />
Insoluble fraction.<br />
Add 1 ml Lysis buffer 2 to the insoluble<br />
fraction tube.<br />
The insoluble pellet contains a large quantity of genomic<br />
DNA, making it viscous <strong>and</strong> difficult to resuspend. Using a<br />
syringe with 22 gauge needle, repeatedly pull the solution<br />
through the syringe to shear the pellet <strong>and</strong> decrease the<br />
viscosity.<br />
Label two clean 1.5 ml screwcap microcentrifuge tubes<br />
Soluble PAGE <strong>and</strong> Insoluble PAGE. Take a 50 µl aliquot<br />
from each fraction Soluble fraction <strong>and</strong> Insoluble fraction<br />
<strong>and</strong> prepare for SDS-PAGE per manual instructions<br />
Store the SDS-PAGE samples, Soluble <strong>and</strong> Insoluble<br />
fractions at –20°C until ready for further steps.<br />
Lysed cells<br />
(thawed)<br />
Soluble<br />
Fraction<br />
Insoluble<br />
Fraction<br />
Insoluble<br />
Fraction<br />
50 µl<br />
Prep<br />
Insoluble<br />
PAGE<br />
sample<br />
Spin 20 min at 16,000 x g<br />
Decant supernatant<br />
into tube labeled<br />
Soluble raction<br />
+ 1 ml Lysis<br />
buffer 2<br />
Shear insoluble fraction with<br />
syringe needle to resuspend<br />
pellet<br />
<br />
Soluble<br />
Fraction<br />
50 µl<br />
Prep<br />
Soluble<br />
PAGE<br />
sample<br />
110 Chapter 4: 11 ml Culture Protocol<br />
for Centrifugation <strong>Purification</strong>