Santander, February 19th-22nd 2008 - Aranzadi
Santander, February 19th-22nd 2008 - Aranzadi
Santander, February 19th-22nd 2008 - Aranzadi
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
24<br />
KATERINA DOUKA, THOMAS F. G. HIGHAM AND ROBERT E. M. HEDGES<br />
unsuitable for dating. With the help of Low-<br />
Vacuum SEM (LV-SEM) or Environmental SEM<br />
(ESEM) that do not require surface coating, we<br />
are able to use the same fragments for imaging<br />
and AMS 14 C dating.<br />
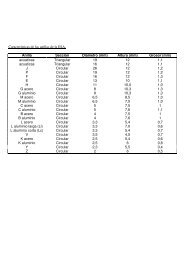
Figure 2. Close-up photograph and ESEM micrograph of a diagenetic<br />
Glycymeris sp. specimen from a Palaeolithic context. The mineralogical changes<br />
are observed macroscopically (left, red rectangle) and are confirmed<br />
microscopically (right), in the form of distinct calcite crystals that cover the entire<br />
surface of the sample and replace the original aragonite microstructure.<br />
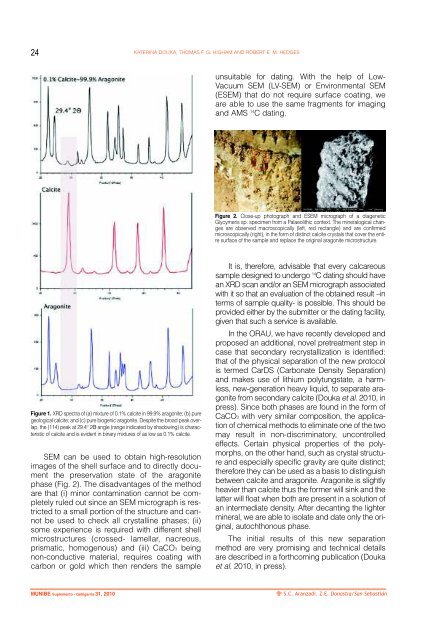
Figure 1. XRD spectra of (a) mixture of 0.1% calcite in 99.9% aragonite; (b) pure<br />
geological calcite; and (c) pure biogenic aragonite. Despite the broad peak overlap,<br />
the (114) peak at 29.4 O 2Θ angle (range indicated by shadowing) is characteristic<br />
of calcite and is evident in binary mixtures of as low as 0.1% calcite.<br />
SEM can be used to obtain high-resolution<br />
images of the shell surface and to directly document<br />
the preservation state of the aragonite<br />
phase (Fig. 2). The disadvantages of the method<br />
are that (i) minor contamination cannot be completely<br />
ruled out since an SEM micrograph is restricted<br />
to a small portion of the structure and cannot<br />
be used to check all crystalline phases; (ii)<br />
some experience is required with different shell<br />
microstructures (crossed- lamellar, nacreous,<br />
prismatic, homogenous) and (iii) CaCO3 being<br />
non-conductive material, requires coating with<br />
carbon or gold which then renders the sample<br />
It is, therefore, advisable that every calcareous<br />
sample designed to undergo 14 C dating should have<br />
an XRD scan and/or an SEM micrograph associated<br />
with it so that an evaluation of the obtained result –in<br />
terms of sample quality- is possible. This should be<br />
provided either by the submitter or the dating facility,<br />
given that such a service is available.<br />
In the ORAU, we have recently developed and<br />
proposed an additional, novel pretreatment step in<br />
case that secondary recrystallization is identified:<br />
that of the physical separation of the new protocol<br />
is termed CarDS (Carbonate Density Separation)<br />
and makes use of lithium polytungstate, a harmless,<br />
new-generation heavy liquid, to separate aragonite<br />
from secondary calcite (Douka et al. 2010, in<br />
press). Since both phases are found in the form of<br />
CaCO3 with very similar composition, the application<br />
of chemical methods to eliminate one of the two<br />
may result in non-discriminatory, uncontrolled<br />
effects. Certain physical properties of the polymorphs,<br />
on the other hand, such as crystal structure<br />
and especially specific gravity are quite distinct;<br />
therefore they can be used as a basis to distinguish<br />
between calcite and aragonite. Aragonite is slightly<br />
heavier than calcite thus the former will sink and the<br />
latter will float when both are present in a solution of<br />
an intermediate density. After decanting the lighter<br />
mineral, we are able to isolate and date only the original,<br />
autochthonous phase.<br />
The initial results of this new separation<br />
method are very promising and technical details<br />
are described in a forthcoming publication (Douka<br />
et al. 2010, in press).<br />
MUNIBE Suplemento - Gehigarria 31, 2010<br />
S.C. <strong>Aranzadi</strong>. Z.E. Donostia/San Sebastián