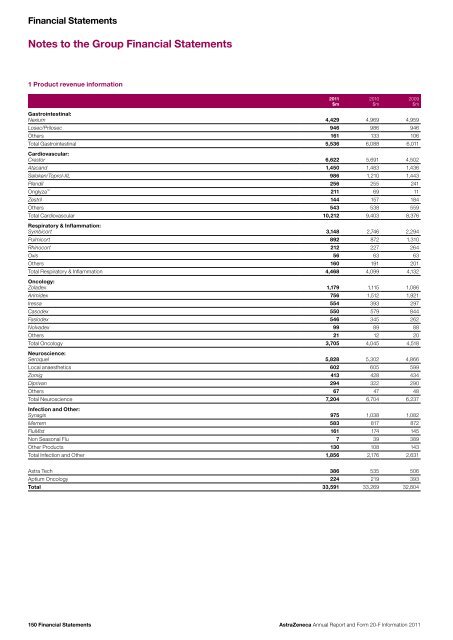
Financial StatementsNotes to the Group Financial Statements1 Product revenue information<strong>20</strong>11$m<strong>20</strong>10$m<strong>20</strong>09$mGastrointestinal:Nexium 4,429 4,969 4,959Losec/Prilosec 946 986 946Others 161 133 106Total Gastrointestinal 5,536 6,088 6,011Cardiovascular:Crestor 6,622 5,691 4,502Atac<strong>and</strong> 1,450 1,483 1,436Seloken/Toprol-XL 986 1,210 1,443Plendil 256 255 241Onglyza 211 69 11Zestril 144 157 184Others 543 538 559Total Cardiovascular 10,212 9,403 8,376Respiratory & Inflammation:Symbicort 3,148 2,746 2,294Pulmicort 892 872 1,310Rhinocort 212 227 264Oxis 56 63 63Others 160 191 <strong>20</strong>1Total Respiratory & Inflammation 4,468 4,099 4,132Oncology:Zoladex 1,179 1,115 1,086Arimidex 756 1,512 1,921Iressa 554 393 297Casodex 550 579 844Faslodex 546 345 262Nolvadex 99 89 88Others 21 12 <strong>20</strong>Total Oncology 3,705 4,045 4,518Neuroscience:Seroquel 5,828 5,302 4,866Local anaesthetics 602 605 599Zomig 413 428 434Diprivan 294 322 290Others 67 47 48Total Neuroscience 7,<strong>20</strong>4 6,704 6,237Infection <strong>and</strong> Other:Synagis 975 1,038 1,082Merrem 583 817 872FluMist 161 174 145Non Seasonal Flu 7 39 389Other Products 130 108 143Total Infection <strong>and</strong> Other 1,856 2,176 2,631Astra Tech 386 535 506Aptium Oncology 224 219 393Total 33,591 33,269 32,804150 Financial Statements<strong>AstraZeneca</strong> <strong>Annual</strong> <strong>Report</strong> <strong>and</strong> <strong>Form</strong> <strong>20</strong>-F <strong>Information</strong> <strong>20</strong>11
2 Operating profitOperating profit includes the following items:Research <strong>and</strong> developmentIn <strong>20</strong>11, research <strong>and</strong> development includes a $285m impairment charge related to the termination of development of the investigationalcompound olaparib <strong>and</strong> $150m impairment charge related to the intangible assets held in relation to TC-5214 (see Note 9). In <strong>20</strong>10, research<strong>and</strong> development included a $445m impairment of intangible assets related specifically to motavizumab.Selling, general <strong>and</strong> administrative costsIn <strong>20</strong>11, selling, general <strong>and</strong> administrative costs includes $135m of legal provision charges in respect of the ongoing Seroquel product liabilitylitigation, Average Wholesale Price litigation in the US <strong>and</strong> the Toprol-XL antitrust litigation. In <strong>20</strong>10, selling, general <strong>and</strong> administrative costsincluded legal provision of $612m of which $592m was in respect to Seroquel legal matters. The current status of these matters is describedin Note 25. These provisions constituted our best estimate at that time of losses expected for these matters.Also included within selling, general <strong>and</strong> administrative costs in <strong>20</strong>10 were gains of $791m arising from changes made to benefits under certainof the Group’s post-retirement benefit plans, chiefly the Group’s UK pension plan. Further details of this gain are included in Note 18.In <strong>20</strong>09, <strong>AstraZeneca</strong> was defending its interests in various federal <strong>and</strong> state investigations <strong>and</strong> civil litigation matters relating to drug marketing<strong>and</strong> pricing practices <strong>and</strong> in respect of which the Group made provisions in aggregate of $636m during <strong>20</strong>09. $524m of this was made inrespect of the US Attorney’s Office investigation into sales <strong>and</strong> marketing practices involving Seroquel <strong>and</strong> $112m related to average wholesaleprice litigation.Profit on disposal of subsidiaryThe profit on disposal of subsidiary in <strong>20</strong>11 of $1,483m relates to the sale of the Astra Tech business to DENTSPLY International Inc. Furtherdetails are included in Note 22.Other operating income <strong>and</strong> expense<strong>20</strong>11$m<strong>20</strong>10$m<strong>20</strong>09$mRoyaltiesIncome 610 522 255Amortisation (51) (59) (79)Impairment – (123) (150)Net gain on disposal of property, plant <strong>and</strong> equipment 33 66 8Gains on disposal of product rights – – 170Net (loss)/gain on disposal of other intangible assets – (1) 1Gains on divestments of non-core products – – 216Impairment of intangible assets relating to future licensing <strong>and</strong> contractual income – – (115)Other income 226 307 265Other expense (41) – (18)Other operating income <strong>and</strong> expense 777 712 553Royalty amortisation <strong>and</strong> impairment relates to income streams acquired with MedImmune.Restructuring costsDuring <strong>20</strong>11, the Group continued the restructuring programmes approved by the SET <strong>and</strong> announced in previous years. In addition, the Groupannounced further programmes during the year. The tables below show the costs that have been charged in respect of these programmes bycost category <strong>and</strong> type. Severance provisions are detailed in Note 17.Cost of sales 54 144 188Research <strong>and</strong> development 468 654 68Selling, general <strong>and</strong> administrative costs 639 404 403Total charge 1,161 1,<strong>20</strong>2 659Severance costs 403 505 262Accelerated depreciation <strong>and</strong> impairment 290 299 148Other 468 398 249Total charge 1,161 1,<strong>20</strong>2 659<strong>20</strong>11$m<strong>20</strong>11$m<strong>20</strong>10$m<strong>20</strong>10$m<strong>20</strong>09$m<strong>20</strong>09$mFinancial StatementsOther costs are those incurred in designing <strong>and</strong> implementing the Group’s various restructuring initiatives including internal project costs,external consultancy fees <strong>and</strong> staff relocation costs.<strong>AstraZeneca</strong> <strong>Annual</strong> <strong>Report</strong> <strong>and</strong> <strong>Form</strong> <strong>20</strong>-F <strong>Information</strong> <strong>20</strong>11 Financial Statements 151