Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Medical Microbiology Section<br />
Murine Chagas’ disease: Requirements for Th1 inducing<br />
cytokines and NK cell activity in an effective immune response<br />
Zusammenfassung<br />
Der einzellige Parasit Trypanosoma cruzi ist der Erreger<br />
der Chagas-Krankheit des Menschen. Immunität<br />
des Wirtes ist abhängig von einer proinflammatorischen<br />
T-lymphozytären Immunreaktion. Wir haben in<br />
IL-12- bzw. IL-18- defizienten Mäusen untersucht, ob<br />
diese unterschiedlich suszeptibel gegenüber dem<br />
Parasiten sind. Während IL-12-/- Mäuse auch sehr<br />
niedrigen Infektionsdosen erlagen, waren IL-18-/-<br />
Mäuse vergleichbar resistent wie Wildtyp Kontrollen.<br />
Parasitäre Gewebslasten wurden an Tag 14 in einer<br />
quantitative PCR verglichen. In Geweben von IL-12-/-<br />
Mäuse waren sie deutlich erhöht, v.a. in der Leber;<br />
bei IL-18-/- Mäusen waren sie mit denen von Kontrollmäusen<br />
vergleichbar. Die Expression von IL-12<br />
ist demnach essentiell in der Immunabwehr gegenüber<br />
T. cruzi, während die Expression von IL-18 nicht<br />
zu einer Steigerung der Resistenz führt. IL-12 ist u.a.<br />
<strong>für</strong> die Aktivierung von NK-Zellen notwendig. Die Parasitämie<br />
in NK-Zell-depletierten Mäuse war signifikant<br />
erhöht, wenngleich Gewebslasten nicht verändert<br />
waren. Dieser Effekt war Perforin-unabhängig.<br />
Summary<br />
The protozoan pathogen Trypanosoma cruzi causes<br />
Chagas’ disease in man. A protective immune response<br />
by the host depends on pro-inflammatory Tcell reactivity.<br />
We investigated the requirements for expression of IL-<br />
12 and IL-18 in C57BL/6 mice in instigating such a<br />
response. IL-12-/- mice were susceptible to very low<br />
infectious doses, whereas IL-18-/- mice resisted an<br />
infection to the same extent as wildtype controls.<br />
Parasite tissue burdens were compared on day 14<br />
by real time quantitative PCR. Parasite infiltration was<br />
vastly increased in tissues of IL-12-/- mice, most notably<br />
in the liver, wheras in IL-18-/- mice, it was comparable<br />
to wildtype controls. Thus, IL-12 is essential for protective<br />
immunity to T. cruzi in this model, while IL-18 is<br />
dispensable. NK cells (that are activated by IL-12)<br />
showed contact-dependent effector function against<br />
free trypanosomes in vitro. Parasitaemia in NK celldepleted<br />
mice was significantly increased, but tissue<br />
parasite burdens were not altered. This effect was<br />
found to be perforin-independent.<br />
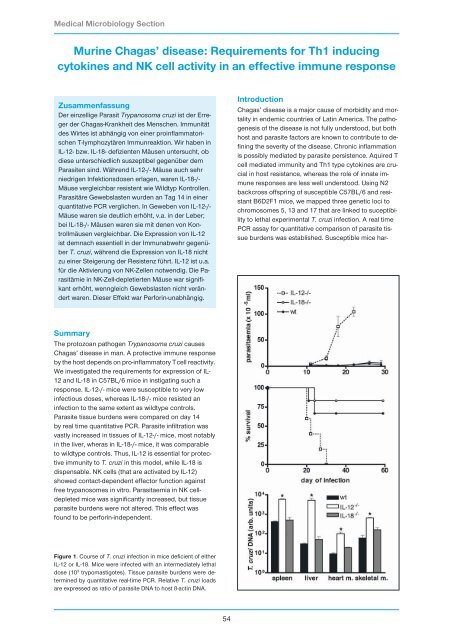
Figure 1. Course of T. cruzi infection in mice deficient of either<br />
IL-12 or IL-18. Mice were infected with an intermediately lethal<br />
dose (10 3 trypomastigotes). Tissue parasite burdens were determined<br />
by quantitative real-time PCR. Relative T. cruzi loads<br />
are expressed as ratio of parasite DNA to host ß-actin DNA.<br />
54<br />
Introduction<br />
Chagas’ disease is a major cause of morbidity and mortality<br />
in endemic countries of Latin America. The pathogenesis<br />
of the disease is not fully understood, but both<br />
host and parasite factors are known to contribute to defining<br />
the severity of the disease. Chronic inflammation<br />
is possibly mediated by parasite persistence. Aquired T<br />
cell mediated immunity and Th1 type cytokines are crucial<br />
in host resistance, whereas the role of innate immune<br />
responses are less well understood. Using N2<br />
backcross offspring of susceptible C57BL/6 and resistant<br />
B6D2F1 mice, we mapped three genetic loci to<br />
chromosomes 5, 13 and 17 that are linked to suceptibility<br />
to lethal experimental T. cruzi infection. A real time<br />
PCR assay for quantitative comparison of parasite tissue<br />
burdens was established. Susceptible mice har-