Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Research Group Heussler (Malaria I) - Bernhard-Nocht-Institut für ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
low level chronic replication of the SIV∆nef, expands<br />
the numbers of γδ T cells at the portal of entry.<br />
One of the most important immunoarchitectural<br />
changes was that the number of mature DCs in the<br />
tonsils strikingly expanded in the vaccinated animals<br />
but not in the unvaccinated controls, as assessed by<br />
staining for the DC-LAMP and the CD83 markers. In<br />
naïve challenged controls, and in vaccinated animals<br />
prior to challenge, the frequency of mature DCs was<br />
similar to normal tonsillar tissue. However, in the vaccinated<br />
challenged animals, the frequency of mature DCs<br />
increased almost 2 fold at day 4. Then DC numbers<br />
began to fall at day 7/8 and were back to baseline at<br />
day 14/15. In the naïve controls, there was no change<br />
in DC numbers at day 4/7, but these began to fall below<br />
normal at a later time point. Therefore one of the<br />
distinctive features of the portal of entry of vaccinated<br />
challenged animals is an acute expansion of mature<br />
DCs, whereas in unvaccinated monkeys, DC numbers<br />
actually drop considerably during acute infection (3).<br />
In conclusion, SIV∆NU vaccination can selectively block<br />
early infection at the entry site of an immunodeficiency<br />
virus, which we propose depends at least in part upon<br />
innate protective functions by dendritic and γδ T cells.<br />
γδ Tcells can produce chemokines and cytokines that<br />
promote DC maturation (Leslie D.S. et al. J. Exp. Med.<br />
2002; 196:1575). Mature DC could shift the host toward<br />
adoptive immune responses that impede replication<br />
of the highly pathogenic wild-type virus during primary<br />
infection.<br />
87<br />
Selected Publications<br />
• Stahl-Hennig C et al. (1999). Science 285: 1261-1265<br />
• Stahl-Hennig, C et al. (2002). J. Virol. 76: 688-696<br />
• Tenner-Racz K et al. (2004). Proc. Natl. Acad. Sci.<br />
USA 101: 3017-3022<br />
Funding<br />
• European Community Contract QLK2-CT-1999-01215<br />
Cooperating Partners<br />
• Ralph M. Steinman, Rockefeller University,<br />
New York, USA<br />
• Christiane Stahl-Hennig, German Primate Centre,<br />
Göttingen (DPZ)<br />
• Klaus Überla, Ruhr-University Bochum<br />
• Manfred Dierich /Heribert Stoiber, <strong>Institut</strong>e of Hygiene<br />
and Social Medicine, Innsbruck, Austria<br />
• Ralf Ignatius, Charité - University Medicine Berlin<br />
• Gudmundur Gerogsson, University of Iceland,<br />
Reykjavik, Iceland<br />
• Gianni Pozzi, University of Siena, Italy<br />
• Carlo Baroni, University „La Sapienza”, Roma, Italy<br />
• Marco Baggiolini, Mariagrazia Uguccioni, <strong>Institut</strong>e for<br />
<strong>Research</strong> in Biomedicine, Bellinzona, Switzerland<br />
• Jonathan Heeney, Biomedical Primate <strong>Research</strong><br />
Center, Rijswijk, Netherlands<br />
Investigators<br />
• Paul Racz<br />
• Klara Tenner-Racz<br />
Tropical Medicine Section<br />
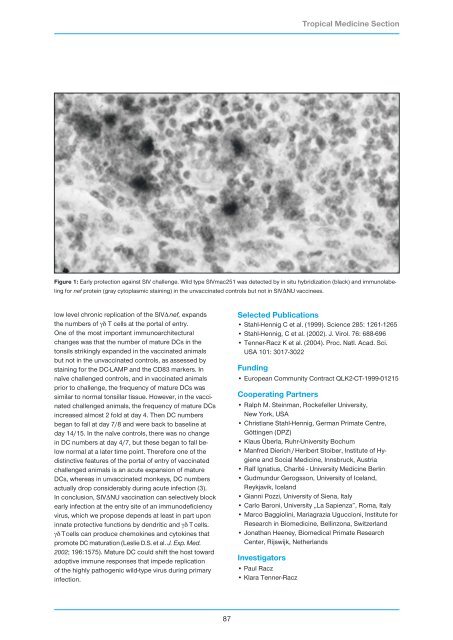
Figure 1: Early protection against SIV challenge. Wild type SIVmac251 was detected by in situ hybridization (black) and immunolabeling<br />
for nef protein (gray cytoplasmic staining) in the unvaccinated controls but not in SIV∆NU vaccinees.