Homoleptische Methylverbindungen von Elementen der 4. und 5 ...
Homoleptische Methylverbindungen von Elementen der 4. und 5 ...
Homoleptische Methylverbindungen von Elementen der 4. und 5 ...
Sie wollen auch ein ePaper? Erhöhen Sie die Reichweite Ihrer Titel.
YUMPU macht aus Druck-PDFs automatisch weboptimierte ePaper, die Google liebt.
Praktischer Teil<br />
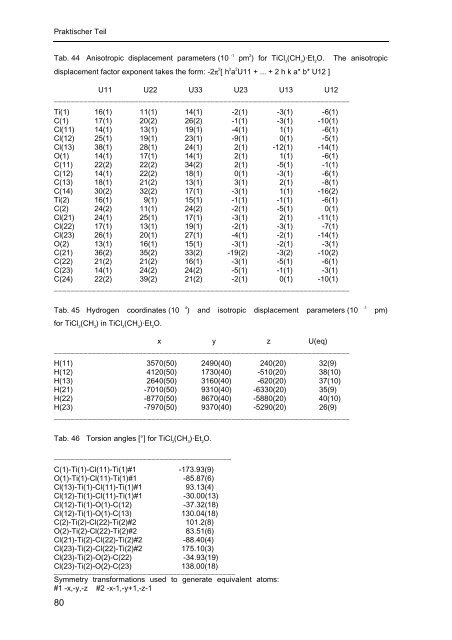
Tab. 44 Anisotropic displacement parameters (10 -1 pm 2 ) for TiCl (CH )·Et O. The anisotropic<br />
3 3 2<br />
displacement factor exponent takes the form: -2π 2 [ h 2 a 2 U11 + ... + 2 h k a* b* U12 ]<br />
U11 U22 U33 U23 U13 U12<br />
______________________________________________________________________<br />
Ti(1) 16(1) 11(1) 14(1) -2(1) -3(1) -6(1)<br />
C(1) 17(1) 20(2) 26(2) -1(1) -3(1) -10(1)<br />
Cl(11) 14(1) 13(1) 19(1) -4(1) 1(1) -6(1)<br />
Cl(12) 25(1) 19(1) 23(1) -9(1) 0(1) -5(1)<br />
Cl(13) 38(1) 28(1) 24(1) 2(1) -12(1) -14(1)<br />
O(1) 14(1) 17(1) 14(1) 2(1) 1(1) -6(1)<br />
C(11) 22(2) 22(2) 34(2) 2(1) -5(1) -1(1)<br />
C(12) 14(1) 22(2) 18(1) 0(1) -3(1) -6(1)<br />
C(13) 18(1) 21(2) 13(1) 3(1) 2(1) -8(1)<br />
C(14) 30(2) 32(2) 17(1) -3(1) 1(1) -16(2)<br />
Ti(2) 16(1) 9(1) 15(1) -1(1) -1(1) -6(1)<br />
C(2) 24(2) 11(1) 24(2) -2(1) -5(1) 0(1)<br />
Cl(21) 24(1) 25(1) 17(1) -3(1) 2(1) -11(1)<br />
Cl(22) 17(1) 13(1) 19(1) -2(1) -3(1) -7(1)<br />
Cl(23) 26(1) 20(1) 27(1) -4(1) -2(1) -14(1)<br />
O(2) 13(1) 16(1) 15(1) -3(1) -2(1) -3(1)<br />
C(21) 36(2) 35(2) 33(2) -19(2) -3(2) -10(2)<br />
C(22) 21(2) 21(2) 16(1) -3(1) -5(1) -6(1)<br />
C(23) 14(1) 24(2) 24(2) -5(1) -1(1) -3(1)<br />
C(24) 22(2) 39(2) 21(2) -2(1) 0(1) -10(1)<br />
______________________________________________________________________<br />
Tab. 45 Hydrogen coordinates (10 4 ) and isotropic displacement parameters (10 -1 pm)<br />
for TiCl (CH ) in TiCl (CH )·Et O.<br />
3 3 3 3 2<br />
x y z U(eq)<br />
______________________________________________________________________<br />
H(11) 3570(50) 2490(40) 240(20) 32(9)<br />
H(12) 4120(50) 1730(40) -510(20) 38(10)<br />
H(13) 2640(50) 3160(40) -620(20) 37(10)<br />
H(21) -7010(50) 9310(40) -6330(20) 35(9)<br />
H(22) -8770(50) 8670(40) -5880(20) 40(10)<br />
H(23) -7970(50) 9370(40) -5290(20) 26(9)<br />
______________________________________________________________________<br />
Tab. 46 Torsion angles [°] for TiCl 3 (CH 3 )·Et 2 O.<br />
__________________________________________<br />
C(1)-Ti(1)-Cl(11)-Ti(1)#1 -173.93(9)<br />
O(1)-Ti(1)-Cl(11)-Ti(1)#1 -85.87(6)<br />
Cl(13)-Ti(1)-Cl(11)-Ti(1)#1 93.13(4)<br />
Cl(12)-Ti(1)-Cl(11)-Ti(1)#1 -30.00(13)<br />
Cl(12)-Ti(1)-O(1)-C(12) -37.32(18)<br />
Cl(12)-Ti(1)-O(1)-C(13) 130.04(18)<br />
C(2)-Ti(2)-Cl(22)-Ti(2)#2 101.2(8)<br />
O(2)-Ti(2)-Cl(22)-Ti(2)#2 83.51(6)<br />
Cl(21)-Ti(2)-Cl(22)-Ti(2)#2 -88.40(4)<br />
Cl(23)-Ti(2)-Cl(22)-Ti(2)#2 175.10(3)<br />
Cl(23)-Ti(2)-O(2)-C(22) -3<strong>4.</strong>93(19)<br />
Cl(23)-Ti(2)-O(2)-C(23) 138.00(18)<br />
___________________________________________<br />
Symmetry transformations used to generate equivalent atoms:<br />
#1 -x,-y,-z #2 -x-1,-y+1,-z-1<br />
80