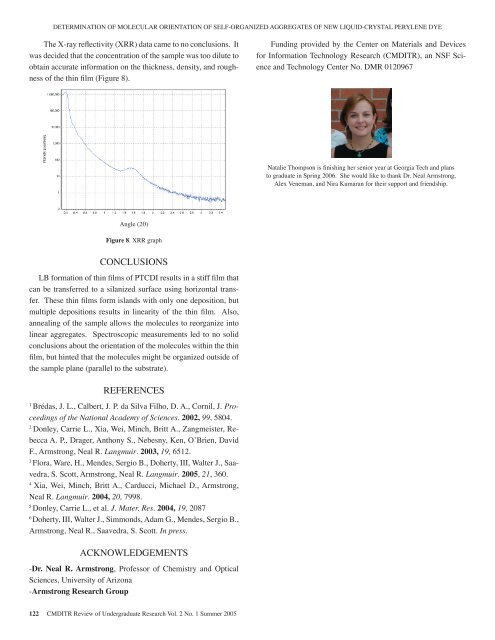
DETERMINATION OF MOLECULAR ORIENTATION OF SELF-ORGANIZED AGGREGATES OF NEW LIQUID-CRYSTAL PERYLENE DYEThe X-ray reflectivity (XRR) data came to no conclusions. Itwas decided that the concentration <strong>of</strong> the sample was too dilute toobtain accurate information on the thickness, density, and roughness<strong>of</strong> the thin film (Figure 8).Funding provided by the Center on Materials and Devicesfor Information Technology Research (CMDITR), an NSF Scienceand Technology Center No. DMR 0120967Natalie Thompson is finishing her senior year at Georgia Tech and plansto graduate in Spring 2006. She would like to thank Dr. Neal Armstrong,Alex Veneman, and Nira Kumaran for their support and friendship.Angle (2θ)Figure 8. XRR graphCONCLUSIONSLB formation <strong>of</strong> thin films <strong>of</strong> PTCDI results in a stiff film thatcan be transferred to a silanized surface using horizontal transfer.These thin films form islands with only one deposition, butmultiple depositions results in linearity <strong>of</strong> the thin film. Also,annealing <strong>of</strong> the sample allows the molecules to reorganize intolinear aggregates. Spectroscopic measurements led to no solidconclusions about the orientation <strong>of</strong> the molecules within the thinfilm, but hinted that the molecules might be organized outside <strong>of</strong>the sample plane (parallel to the substrate).REFERENCES1Brédas, J. L., Calbert, J. P. da Silva Filho, D. A., Cornil, J. Proceedings<strong>of</strong> the National Academy <strong>of</strong> Sciences. 2002, 99, 5804.2Donley, Carrie L., Xia, Wei, Minch, Britt A., Zangmeister, RebeccaA. P., Drager, Anthony S., Nebesny, Ken, O’Brien, DavidF., Armstrong, Neal R. Langmuir. 2003, 19, 6512.3Flora, Ware, H., Mendes, Sergio B., Doherty, III, Walter J., Saavedra,S. Scott, Armstrong, Neal R. Langmuir. <strong>2005</strong>, 21, 360.4Xia, Wei, Minch, Britt A., Carducci, Michael D., Armstrong,Neal R. Langmuir. 2004, 20, 7998.5Donley, Carrie L., et al. J. Mater. Res. 2004, 19, 20876Doherty, III, Walter J., Simmonds, Adam G., Mendes, Sergio B.,Armstrong, Neal R., Saavedra, S. Scott. In press.ACKNOWLEDGEMENTS-Dr. Neal R. Armstrong, Pr<strong>of</strong>essor <strong>of</strong> Chemistry and OpticalSciences, <strong>University</strong> <strong>of</strong> Arizona-Armstrong Research Group122 CMDITR Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong>
Hydrogel Materials for Two-Photon Micr<strong>of</strong>abricationMayen UdoetukNorfolk State <strong>University</strong>Dr. Joe Perry and Vincent ChenGeorgia Institute <strong>of</strong> TechnologyOBJECTIVEThe purpose <strong>of</strong> the research is to develop a hydrogel materialsystem for two-photon micr<strong>of</strong>abrication. Problems that were addressedin this research project are:1) For what composition can sufficient crosslinking beachieved by ultra-violet (UV) exposure such that the patternedstructure will not wash away upon development?2) Can hydrophilic monomers be crosslinked using currenttwo-photon absorbing dyes?3) What are the requirements <strong>of</strong> the material and the structureto ensure that pores and other desired features within thestructure are maintained after swelling?Potential practical applications <strong>of</strong> these materials are, for example,microstructures to be used for cell adhesion, which canfind applications in tissue engineering, and structures that respondto the solvent environment for sensors or micr<strong>of</strong>luidics. Mostresearch in micr<strong>of</strong>abrication to date has been carried out with acrylateand epoxy type polymers. 1 However, in applications thatinvolve biological molecules or require the adhesion <strong>of</strong> cells ontopolymeric structures, biocompatible materials need to be used.Hydrogel systems have hydrophilic surfaces and are <strong>of</strong> particularinterest due to their ability to swell and de-swell, depending ontheir environment.RESEARCH METHODSThe research problems were approached by testing resins basedon hydrophilic monomers and two-photon absorbing dyes or UV initiators.Photolithography is a process <strong>of</strong> transferring spatial patternsonto a surface <strong>of</strong> a material by exposure to light with a mask. Stepsusually involved in this process include substrate cleaning, adhesionlayer formation, photoresist application, s<strong>of</strong>t baking, mask alignment,exposure, development, and hard-baking.The hydrophilic monomers and crosslinker that were usedinclude N-vinylpyrrolidone (VP), hydroxyethylmethacrylate(HEMA), and ethyleneglycol-bis-methacrylate (EGMA). Resinscontaining various amounts <strong>of</strong> these compounds were prepared.The initiator used for UV exposure was 1-[4-(methylthio)phenyl]-2-methyl-2-morpholinopropan-1-one, (UV initiator #7).The amounts <strong>of</strong> initiator used were 0.1, 0.2, and 0.5 wt%, seetable 1.1). To ensure that the structures remained attached to thesubstrate, the surface <strong>of</strong> the substrate was modified with an adhesionpromoter (95% methanol and 5% 3-(trimethoxysilyl)propylmethacrylate). The adhesion promoter solution was spin-coatedonto microscope slides and cover slips, and baked on a hotplateat 90°C for 2 minutes.Five drops <strong>of</strong> the hydrophilic resin were placed on surfacetreated microscope slides and cover slips, and were exposed toUV light from a mercury UV lamp (254nm) for various times.Solubility tests were done in order to find a solvent that wouldwash away any resin left after exposure, while leaving the polymerizedstructure intact. All <strong>of</strong> the following were found toperform well as development solvents: dichloromethane, toluene,ethanol, dioxane, cyclohexane, dimethyl formamide, and 4-methyl-2-pentanone. 4-Methyl-2-pentanone was chosen for usein the experiments.Several sample geometries were tested but some gave an inadequateamount <strong>of</strong> crosslinking. The initial setup included theresin on top <strong>of</strong> a microscope slide modified with adhesion promoter,with the UV light shining down on it. Due to the thickness<strong>of</strong> the resin, the UV light did not penetrate to the resin-substrateinterface in order to yield sufficient crosslinking. Two geometriesthat were found to provide sufficient exposure and crosslinking atthe interface are shown in diagram 1.1.Experiments were conducted to determine the swelling properties<strong>of</strong> the crosslinked materials. The dimensions <strong>of</strong> the lineswere measured before and after swelling with water at room temperature.The feature dimensions were measured using opticalmicroscopy.Bulk materials <strong>of</strong> the hydrogels were prepared using setup 1in order to better visualize the swelling. Fluorescence and transmissionimaging were used to characterize and document thefabricated structures. A laser was used for 3D micr<strong>of</strong>abrication<strong>of</strong> structures 3 . A test structure consisting <strong>of</strong> a vertical stack <strong>of</strong>crossing lines was designed and fabricated using two-photon micr<strong>of</strong>abrication.1-[4-(methylthiophenyl]-2-methyl-2-morpholinopropane-1-one (UV #7)was varied in the compositions. 0.1, 0.2, and 0.5 wt% were used.CMDITR Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong> 123
- Page 2 and 3:
The material is based upon work sup
- Page 4 and 5:
TABLE OF CONTENTSSynthesis of Dendr
- Page 6 and 7:
6 CMDITR Review of Undergraduate Re
- Page 8 and 9:
SYNTHESIS OF DENDRIMER BUILDING BLO
- Page 10 and 11:
throughout the work period. Five su
- Page 12 and 13:
12 CMDITR Review of Undergraduate R
- Page 14 and 15:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 16 and 17:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 18 and 19:
SYNTHESIS OF NORBORNENE MONOMER OF
- Page 20:
20 CMDITR Review of Undergraduate R
- Page 23 and 24:
using different reaction conditions
- Page 25 and 26:
Synthesis of Nonlinear Optical-Acti
- Page 27 and 28:
quality of the XRD structures wasca
- Page 29 and 30:
Behavioral Properties of Colloidal
- Page 32 and 33:
Transmission electron microscopy ha
- Page 34 and 35:
34 CMDITR Review of Undergraduate R
- Page 36 and 37:
areorient themselves with the elect
- Page 38 and 39:
Fabry-Perot modulators with electro
- Page 40 and 41:
40 CMDITR Review of Undergraduate R
- Page 42 and 43:
QUANTIZED HAMILTON DYNAMICS APPLIED
- Page 44 and 45:
44 CMDITR Review of Undergraduate R
- Page 46 and 47:
INVESTIGATING NEW CLADDING AND CORE
- Page 48 and 49:
Dr. Robert NorwoodChris DeRoseAmir
- Page 50 and 51:
SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 52 and 53:
SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 54 and 55:
OPTIMIZING HYBRID WAVEGUIDESpropaga
- Page 56 and 57:
At closer spaces the second undesir
- Page 58 and 59:
SYNTHESIS AND ANALYSIS OF THIOL-STA
- Page 60 and 61:
60 CMDITR Review of Undergraduate R
- Page 62 and 63:
QUINOXALINE-CONTAINING POLYFLUORENE
- Page 64 and 65:
QUINOXALINE-CONTAINING POLYFLUORENE
- Page 66 and 67:
66 CMDITR Review of Undergraduate R
- Page 68 and 69:
SYNTHESIS OF DENDRON-FUNCTIONALIZED
- Page 70 and 71:
70 CMDITR Review of Undergraduate R
- Page 72 and 73: BUILDING AN OPTICAL OXIMETER TO MEA
- Page 74 and 75: 74 CMDITR Review of Undergraduate R
- Page 76 and 77: 76 CMDITR Review of Undergraduate R
- Page 78 and 79: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 80 and 81: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 82 and 83: SYNTHESIS AND CHARACTERIZATION OF E
- Page 84 and 85: My name is Aaron Montgomery and I a
- Page 86 and 87: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 88 and 89: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 90 and 91: EFFECTS OF SURFACE CHEMISTRY ON CAD
- Page 92 and 93: EFFECTS OF SURFACE CHEMISTRY ON CAD
- Page 94 and 95: 94 CMDITR Review of Undergraduate R
- Page 96 and 97: SYNTHESIS OF A POLYENE EO CHROMOPHO
- Page 98 and 99: SYNTHESIS OF A POLYENE EO CHROMOPHO
- Page 102 and 103: 102 CMDITR Review of Undergraduate
- Page 104 and 105: CHARACTERIZATION OF THE MOLECULAR P
- Page 106 and 107: 106 CMDITR Review of Undergraduate
- Page 108 and 109: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 110 and 111: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 112 and 113: CHARACTERIZATION OF THE PHOTODECOMP
- Page 114 and 115: 114 CMDITR Review of Undergraduate
- Page 116 and 117: ELECTROLUMINESCENT PROPERTIES OF OR
- Page 118 and 119: 118 CMDITR Review of Undergraduate
- Page 120 and 121: DETERMINATION OF MOLECULAR ORIENTAT
- Page 124 and 125: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 126 and 127: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 128 and 129: THE DESIGN OF A FLUID DELIVERY SYST
- Page 130: THE DESIGN OF A FLUID DELIVERY SYST