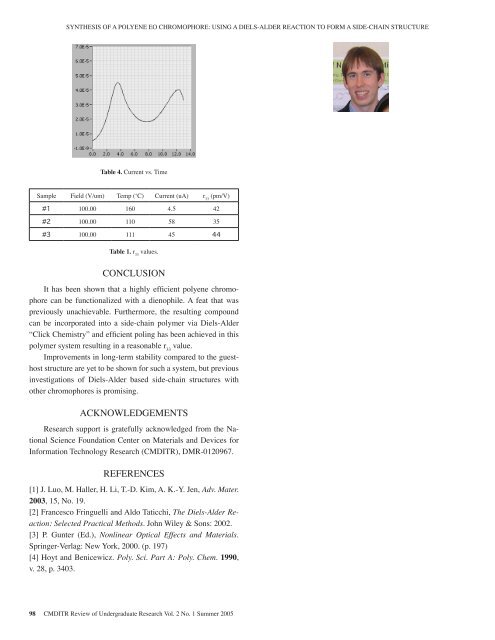
SYNTHESIS OF A POLYENE EO CHROMOPHORE: USING A DIELS-ALDER REACTION TO FORM A SIDE-CHAIN STRUCTUREe poling voltageg at 50 C. Thever time as thedication <strong>of</strong> whenreaches a localbreakdown andto the ITO).g with 100V/umhigh as 44pm/Vthe less efficientch a side-chainFig. 4. Current Vs. Time.dication that theTable 4. Current vs. Timeity <strong>of</strong> the systemrent at such high temperatures (4.5 uA at 160 C)etween the polymer and the chromophores, whichain structure.Sample Field (V/um) Temp (‘C) Current (uA) r 33(pm/V)#1 100.00 160 4.5 42#2 100.00 110 58 35('C) Current (uA) r 33 (pm/V)0 4.5 Table 1. r4233values.#3 100.00 111 45 44CONCLUSIONIt has been shown that a highly efficient 6 <strong>of</strong> polyene 7 chromophorecan be functionalized with a dienophile. A feat that waspreviously unachievable. Furthermore, the resulting compoundcan be incorporated into a side-chain polymer via Diels-Alder“Click Chemistry” and efficient poling has been achieved in thispolymer system resulting in a reasonable r 33value.Improvements in long-term stability compared to the guesthoststructure are yet to be shown for such a system, but previousinvestigations <strong>of</strong> Diels-Alder based side-chain structures withother chromophores is promising.ACKNOWLEDGEMENTSResearch support is gratefully acknowledged from the NationalScience Foundation Center on Materials and Devices forInformation Technology Research (CMDITR), DMR-0120967.REFERENCES[1] J. Luo, M. Haller, H. Li, T.-D. Kim, A. K.-Y. Jen, Adv. Mater.2003, 15, No. 19.[2] Francesco Fringuelli and Aldo Taticchi, The Diels-Alder Reaction:Selected Practical Methods. John Wiley & Sons: 2002.[3] P. Gunter (Ed.), Nonlinear Optical Effects and Materials.Springer-Verlag: New York, 2000. (p. 197)[4] Hoyt and Benicewicz. Poly. Sci. Part A: Poly. Chem. 1990,v. 28, p. 3403.98 CMDITR Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong>
Spectroscopic Investigations <strong>of</strong> Chromophores in Dyed Salt CrystalsStacy A. OliphantEdmonds Community CollegeKristin L. Wustholz, Bart Kahr and Philip J. ReidDepartment <strong>of</strong> Chemistry<strong>University</strong> <strong>of</strong> <strong>Washington</strong>INTRODUCTIONThe Science and Technology Center (STC) is committed tothe development <strong>of</strong> next-generation electro-optic (EO) devices,typically based on dye-doped polymers. Polymer-based switcheshave demonstrated greater device efficiencies when compared totraditional inorganics. Since the EO effect relies on the nonlinearmaterial response, the dye-doped polymers must be noncentrosymmetric.This is typically achieved via the application <strong>of</strong> apoling field. Ultimately, EO activity is given by:alignment and photophysics <strong>of</strong> single chromphores in dye-dopedcrystal hosts using confocal microscopy. Furthermore, by comparingthe orientations <strong>of</strong> dyes to their energetics, the environmentalheterogeneity among individual dye molecules within thecrystal can be studied.|Nµβ|where the EO response is proportional to the number <strong>of</strong> dye molecules(N), the molecular dipole moment (μ), hyperpolarizability(β), and the ordering parameter (cos 3 θ) that represents the extent<strong>of</strong> alignment afforded by poling. Understanding the factors thatinfluence chromophore orientation has taken a backseat to the development<strong>of</strong> enhanced molecular properties (i.e. β). Yet, recentwork suggests that poling is only partially effective in achievingmolecular alignment 1 – making it quite clear that the poling procedureand ordering parameter are not well understood.To experimentally test the efficacy <strong>of</strong> poling, and hencebulk EO device efficiency, it makes sense to begin with a systemin which the chromophores are intrinsically aligned by thehost. When grown in the presence <strong>of</strong> many organic dyes, aqueoussolutions <strong>of</strong> potassium acid phthalate (KAP) and sodium potassiumtartrate tetrahydrate (commonly known as Rochelle salt)frequently deposit “dyed crystals” wherein chromophores are orientedand overgrown by the host lattice. In these crystals, dyesselectively adsorb to particular sub-volumes within the latticeand bulk spectroscopic studies suggest that they are intrinsicallyaligned during the growth process. For example, Figure 1 showsthat 4-(Dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran(DCM) adsorbs to the {11 - 1} growth sector, whileViolamine R (VR) adds to the {010} growth sector <strong>of</strong> the hostcrystal, KAP.Here, a natural comparison presents itself in the study <strong>of</strong> theNLO properties <strong>of</strong> chromophore-polymer composites and dyedopedsingle crystals. We use single-molecule spectroscopy tomeasure the orientations and fluorescence spectra <strong>of</strong> embeddeddye molecules. The focus <strong>of</strong> this work was to interrogate theFigure 1. Dye-Doped KAP Crystals. Violamine R (1) and DCM(2) ad3sorb to different growth sectors <strong>of</strong> the KAP crystal host.EXPERIMENTALDyed crystals were grown by slow evaporation from aqueoussolution, in a temperature controlled air chamber (30°C) withdye concentrations <strong>of</strong> 10 -4 M to 10 -8 M. The latter concentrationwas used to ensure single-molecule resolution.Single-molecule experiments were performed using a confocalmicroscope with the following parameters: an invertedmicroscope (Nikon, TE2000). Excitation from a 532 nm solidstatelaser (NovaLux) was filtered (Chroma) and the excitationpolarization was manipulated using a half-waveplate. The laserwas focused to a diffraction-limited spot with an objective (100x,Nikon, 1.3 NA). Cleaved crystal samples (~1 mm) were mountedon a closed loop x-y piezo scanning stage (Queensgate). Emissionwas spectrally filtered by a dichroic mirror and emissionfilters (Chroma) and spatially filtered with a confocal pinhole(ThorLabs: 50 μm). The fluorescence intensity was imaged ontoa single-photon counting APD (PerkinElmer). Single-moleculefluorescence spectra were measured by adding a monochromator(Acton) attached to a liquid N 2CCD camera (Princeton Instruments)to the confocal instrument.CMDITR Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong> 99
- Page 2 and 3:
The material is based upon work sup
- Page 4 and 5:
TABLE OF CONTENTSSynthesis of Dendr
- Page 6 and 7:
6 CMDITR Review of Undergraduate Re
- Page 8 and 9:
SYNTHESIS OF DENDRIMER BUILDING BLO
- Page 10 and 11:
throughout the work period. Five su
- Page 12 and 13:
12 CMDITR Review of Undergraduate R
- Page 14 and 15:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 16 and 17:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 18 and 19:
SYNTHESIS OF NORBORNENE MONOMER OF
- Page 20:
20 CMDITR Review of Undergraduate R
- Page 23 and 24:
using different reaction conditions
- Page 25 and 26:
Synthesis of Nonlinear Optical-Acti
- Page 27 and 28:
quality of the XRD structures wasca
- Page 29 and 30:
Behavioral Properties of Colloidal
- Page 32 and 33:
Transmission electron microscopy ha
- Page 34 and 35:
34 CMDITR Review of Undergraduate R
- Page 36 and 37:
areorient themselves with the elect
- Page 38 and 39:
Fabry-Perot modulators with electro
- Page 40 and 41:
40 CMDITR Review of Undergraduate R
- Page 42 and 43:
QUANTIZED HAMILTON DYNAMICS APPLIED
- Page 44 and 45:
44 CMDITR Review of Undergraduate R
- Page 46 and 47:
INVESTIGATING NEW CLADDING AND CORE
- Page 48 and 49: Dr. Robert NorwoodChris DeRoseAmir
- Page 50 and 51: SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 52 and 53: SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 54 and 55: OPTIMIZING HYBRID WAVEGUIDESpropaga
- Page 56 and 57: At closer spaces the second undesir
- Page 58 and 59: SYNTHESIS AND ANALYSIS OF THIOL-STA
- Page 60 and 61: 60 CMDITR Review of Undergraduate R
- Page 62 and 63: QUINOXALINE-CONTAINING POLYFLUORENE
- Page 64 and 65: QUINOXALINE-CONTAINING POLYFLUORENE
- Page 66 and 67: 66 CMDITR Review of Undergraduate R
- Page 68 and 69: SYNTHESIS OF DENDRON-FUNCTIONALIZED
- Page 70 and 71: 70 CMDITR Review of Undergraduate R
- Page 72 and 73: BUILDING AN OPTICAL OXIMETER TO MEA
- Page 74 and 75: 74 CMDITR Review of Undergraduate R
- Page 76 and 77: 76 CMDITR Review of Undergraduate R
- Page 78 and 79: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 80 and 81: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 82 and 83: SYNTHESIS AND CHARACTERIZATION OF E
- Page 84 and 85: My name is Aaron Montgomery and I a
- Page 86 and 87: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 88 and 89: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 90 and 91: EFFECTS OF SURFACE CHEMISTRY ON CAD
- Page 92 and 93: EFFECTS OF SURFACE CHEMISTRY ON CAD
- Page 94 and 95: 94 CMDITR Review of Undergraduate R
- Page 96 and 97: SYNTHESIS OF A POLYENE EO CHROMOPHO
- Page 102 and 103: 102 CMDITR Review of Undergraduate
- Page 104 and 105: CHARACTERIZATION OF THE MOLECULAR P
- Page 106 and 107: 106 CMDITR Review of Undergraduate
- Page 108 and 109: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 110 and 111: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 112 and 113: CHARACTERIZATION OF THE PHOTODECOMP
- Page 114 and 115: 114 CMDITR Review of Undergraduate
- Page 116 and 117: ELECTROLUMINESCENT PROPERTIES OF OR
- Page 118 and 119: 118 CMDITR Review of Undergraduate
- Page 120 and 121: DETERMINATION OF MOLECULAR ORIENTAT
- Page 122 and 123: DETERMINATION OF MOLECULAR ORIENTAT
- Page 124 and 125: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 126 and 127: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 128 and 129: THE DESIGN OF A FLUID DELIVERY SYST
- Page 130: THE DESIGN OF A FLUID DELIVERY SYST