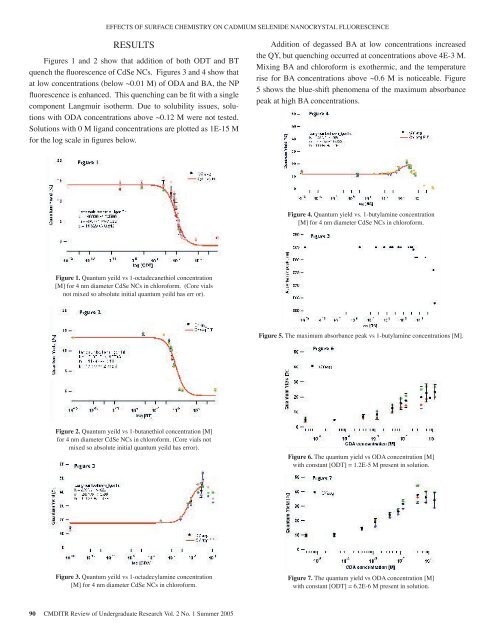
EFFECTS OF SURFACE CHEMISTRY ON CADMIUM SELENIDE NANOCRYSTAL FLUORESCENCERESULTSFigures 1 and 2 show that addition <strong>of</strong> both ODT and BTquench the fluorescence <strong>of</strong> CdSe NCs. Figures 3 and 4 show thatat low concentrations (below ~0.01 M) <strong>of</strong> ODA and BA, the NPfluorescence is enhanced. This quenching can be fit with a singlecomponent Langmuir isotherm. Due to solubility issues, solutionswith ODA concentrations above ~0.12 M were not tested.Solutions with 0 M ligand concentrations are plotted as 1E-15 Mfor the log scale in figures below.Addition <strong>of</strong> degassed BA at low concentrations increasedthe QY, but quenching occurred at concentrations above 4E-3 M.Mixing BA and chlor<strong>of</strong>orm is exothermic, and the temperaturerise for BA concentrations above ~0.6 M is noticeable. Figure5 shows the blue-shift phenomena <strong>of</strong> the maximum absorbancepeak at high BA concentrations.Figure 4. Quantum yield vs. 1-butylamine concentration[M] for 4 nm diameter CdSe NCs in chlor<strong>of</strong>orm.Figure 1. Quantum yeild vs 1-octadecanethiol concentration[M] for 4 nm diameter CdSe NCs in chlor<strong>of</strong>orm. (Core vialsnot mixed so absolute initial quantum yeild has err or).Figure 5. The maximum absorbance peak vs 1-butylamine concentrations [M].Figure 2. Quantum yeild vs 1-butanethiol concentration [M]for 4 nm diameter CdSe NCs in chlor<strong>of</strong>orm. (Core vials notmixed so absolute initial quantum yeild has error).Figure 6. The quantum yield vs ODA concentration [M]with constant [ODT] = 1.2E-5 M present in solution.Figure 3. Quantum yeild vs 1-octadecylamine concentration[M] for 4 nm diameter CdSe NCs in chlor<strong>of</strong>orm.Figure 7. The quantum yield vs ODA concentration [M]with constant [ODT] = 6.2E-6 M present in solution.90 CMDITR Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong>
H 2 H 2 H 2 HC2 HC2CHC2 HC2 HHC22N CCCCCCH C2 HC2C CH2 H2 H C2CHH2 H2 H 32H 2 H 2 H 2 HC2 HC2CHC2 HC2 HHC22N CCCCCCH C2 HC2C CH2 H2 HC2CHH2 H2 H 32The QY was enhanced more by the addition <strong>of</strong> ODA than BA.To investigate the reversibility <strong>of</strong> ligand binding, ODA wasadded to CdSe NCs already partially exchanged with ODT. The QY<strong>of</strong> CdSe NCs in 1.2E-5 M ODT was ~6%. Figure 6 shows that uponaddition <strong>of</strong> ~0.1M ODA, the QY was increased to ~21%. The QY<strong>of</strong> CdSe NCs in 6.2E-6 M ODT was ~10% and figure 7 shows thatthe addition <strong>of</strong> ~0.04 M ODA increased QY up to ~35%.Figure 8, 9, and 10 show the H-NMR 9,10 spectra that wasperformed to attempt to determine the relative amounts <strong>of</strong> eachFigure 10. shows the structure <strong>of</strong> 1-octadecanethiol (a) andcorresponding peak assignments (b) on the solution state H-NMRspectra <strong>of</strong> ODT. Figure 10 (c) shows the H-NMR spectra whenODT is bound to 1E-5 mmol CdSe in deuterated chlor<strong>of</strong>orm.In the case <strong>of</strong> ODA binding to CdSe, the triplet peak (b) at~2.66 ppm corresponding to the alpha-protons (circled in Figure9) is broadened. The pentet at ~3.19 ppm leans towards the upfieldpeaks indicating that it is interacting with the ODA. *Acetone andstearicFigure 10acid impurities. o Peaks are a result <strong>of</strong> amine bound to NC.Figure 10 shows the structure <strong>of</strong> 1-octadecanethiol (a) andcorresponding peak assignments (b) on the solution state H-NMRspectra <strong>of</strong> ODT. Figure 10 (c) shows the H-NMR spectra whenODT Figure is 10 bound to 1E-5 mmol CdSe in deuterated chlor<strong>of</strong>orm.2 4 51 3 62 4 54In 1 H-NMR studies <strong>of</strong> ODT, the proton on the sulfur atom(circled region in Figure 10) is presumed to have a chemical shiftnear the methylene peak at ~1.2 ppm making the peak difficult toresolve.DISCUSSIONWe hypothesized that the ligand binding could be describedby a Langmuir isotherm (eqn 1). The Langmuir isotherm relatesthe fractional surface coverage, θ, <strong>of</strong> ligand on surface sites to theconcentration <strong>of</strong> ligand in solution and can be derived from anequilibrium reaction given the equilibrium binding constant, K,and ligand concentration, [ligand].ligand Figure in 8, solution 9, and 10 that show is free the or H-NMR bound to 9,10 a spectra CdSe NC. that H-NMR was performed to attempt to determine thespectra relative <strong>of</strong> amounts 1E-5 mmol <strong>of</strong> each washed ligand CdSe in solution NCs, ODA, that is ODA free or bound bound to to a CdSe NC. H-NMR spectra <strong>of</strong>1E-5 mmol washed CdSe NCs, ODA, ODA bound to CdSe, ODT, and ODT bound to CdSe inCdSe,deuteratedODT,chlor<strong>of</strong>ormand ODT boundwere conducted.to CdSe in deuterated chlor<strong>of</strong>ormwere conducted.Figure 8 8, 9, and 10 show the H-NMR 9,10 spectra that was performed to attempt to determine therelative amounts <strong>of</strong> each ligand in solution that is free or bound to a CdSe NC. H-NMR spectra <strong>of</strong>1E-5 mmol washed CdSe NCs, ODA, ODA bound to CdSe, ODT, Figure and 8 ODT shows bound the 300 to CdSe MHzindeuterated chlor<strong>of</strong>orm were conducted.solution state H-NMR spectra <strong>of</strong> 1E-5mmol CdSe NCs in deuteratedFigure 8In order to use the isotherm, four assumptions were made: (1)the reaction is reversible, (2) only one monolayer can be formed,Figure 9Figure 8 shows the 300 MHz(a) Figure 8. shows the 300 MHz solution state H-NMR spectra solution (3) binding state H-NMR <strong>of</strong> a ligand spectra to <strong>of</strong> a 1E-5 surface site does not affect the binding<strong>of</strong> 1E-5 mmol CdSe NCs in deuterated chlor<strong>of</strong>orm.bdmmol at any CdSe other NCs sites, in and deuterated (4) the QY is linearly dependent on theeacfractional surface coverage, θ. We also assumed that the NCsfFigure 9started with all empty surface sites, which is not true because(a)there are stearic acid, TOPO, ODA bound to the surface.bdFigure 9. shows the structure <strong>of</strong> 1-octadecylamine (a) ande A more accurate model would use a multiple componenta corresponding c peak assignments b (b) on the colution d,e state H-NMR Langmuir isotherm in which the ligand already bound to the surfaceis taken (a) into and account. corresponding The fluorescence quenching that occursspectra <strong>of</strong> ODA. Figure 9 (c) shows the H-NMR spectrafFigure 9 showswhenfstructure <strong>of</strong> 1-(b)aoctadecylamineODA is bound to IE-5 mmol CdSe deuterated chlor<strong>of</strong>orm.cpeak assignments (b) on the solutionat high amine concentrations does not agree with the assumptionstate H-NMR spectra <strong>of</strong> ODA.Figure (4) that 9 (c) the shows QY increases the H-NMR linearly as the surface is passivated withbd,e* <strong>of</strong>spectra Figure more when 9 amine. shows ODA the For is structure bound the amine to <strong>of</strong> 1E-5 1- curves the fits were made with data at(b)o ammol octadecylamine CdSe deuterated (a) and correspondingchlor<strong>of</strong>orm.(c)camine concentrations below the solution saturation point <strong>of</strong> ODApeak assignments (b) on the solutionstate (~0.01 H-NMR M). spectra <strong>of</strong> ODA.In the case <strong>of</strong> ODA binding to CdSe, the triplet peak (b) at ~2.66 Figure ppm corresponding 9 To (c) improve shows to the themodel, H-NMR alphaprotons(circled in Figure 9) is broadened. The pentet at ~3.19 ppm leans towards the upfield peaksfits with the Hill equation (eqn 2)indicating that it is interacting with* ospectra when ODA is bound to 1E-5the ODA. o *Acetone and stearic mmol were acid CdSe impurities. performed. in deuterated The o chlor<strong>of</strong>orm. Peaks Hill are equation a is widely used as an empiricalresult (c) <strong>of</strong> amine bound to NC.fitting function when some degree <strong>of</strong> deviation from the Langmuirisotherm assumptions is expected.In the case <strong>of</strong> ODA binding to CdSe, the triplet peak (b) at ~2.66 ppm corresponding to the alphaprotons(circled in Figure 9) is broadened. The pentet at ~3.19 ppm leans towards the upfield peaksindicating that it is interacting with the ODA. *Acetone and stearic acid impurities. o Peaks are aresult <strong>of</strong> amine bound to NC.NGTable 1 Langmuir isotherm- K Hill equation- K Hill equation- nCdSe + ODT 106980 +/- 24500 94877 2CdSe + BT 101800+/- 4300 186470 2CdSe + ODA 2001.7 +/- 522 4180 1CdSe + BA 1787.3 +/- 769 2000 1Table 1. shows the equilibrium binding constant (K) valuesfound for least-squares fits <strong>of</strong> the Langmuir isothermand Hill equations to the experimental data.1 3 CMDITR 6 Review <strong>of</strong> Undergraduate Research Vol. 2 No. 1 Summer <strong>2005</strong> 91
- Page 2 and 3:
The material is based upon work sup
- Page 4 and 5:
TABLE OF CONTENTSSynthesis of Dendr
- Page 6 and 7:
6 CMDITR Review of Undergraduate Re
- Page 8 and 9:
SYNTHESIS OF DENDRIMER BUILDING BLO
- Page 10 and 11:
throughout the work period. Five su
- Page 12 and 13:
12 CMDITR Review of Undergraduate R
- Page 14 and 15:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 16 and 17:
BARIUM TITANATE DOPED SOL-GEL FOR E
- Page 18 and 19:
SYNTHESIS OF NORBORNENE MONOMER OF
- Page 20:
20 CMDITR Review of Undergraduate R
- Page 23 and 24:
using different reaction conditions
- Page 25 and 26:
Synthesis of Nonlinear Optical-Acti
- Page 27 and 28:
quality of the XRD structures wasca
- Page 29 and 30:
Behavioral Properties of Colloidal
- Page 32 and 33:
Transmission electron microscopy ha
- Page 34 and 35:
34 CMDITR Review of Undergraduate R
- Page 36 and 37:
areorient themselves with the elect
- Page 38 and 39:
Fabry-Perot modulators with electro
- Page 40 and 41: 40 CMDITR Review of Undergraduate R
- Page 42 and 43: QUANTIZED HAMILTON DYNAMICS APPLIED
- Page 44 and 45: 44 CMDITR Review of Undergraduate R
- Page 46 and 47: INVESTIGATING NEW CLADDING AND CORE
- Page 48 and 49: Dr. Robert NorwoodChris DeRoseAmir
- Page 50 and 51: SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 52 and 53: SYNTHESIS OF TPD-BASED COMPOUNDS FO
- Page 54 and 55: OPTIMIZING HYBRID WAVEGUIDESpropaga
- Page 56 and 57: At closer spaces the second undesir
- Page 58 and 59: SYNTHESIS AND ANALYSIS OF THIOL-STA
- Page 60 and 61: 60 CMDITR Review of Undergraduate R
- Page 62 and 63: QUINOXALINE-CONTAINING POLYFLUORENE
- Page 64 and 65: QUINOXALINE-CONTAINING POLYFLUORENE
- Page 66 and 67: 66 CMDITR Review of Undergraduate R
- Page 68 and 69: SYNTHESIS OF DENDRON-FUNCTIONALIZED
- Page 70 and 71: 70 CMDITR Review of Undergraduate R
- Page 72 and 73: BUILDING AN OPTICAL OXIMETER TO MEA
- Page 74 and 75: 74 CMDITR Review of Undergraduate R
- Page 76 and 77: 76 CMDITR Review of Undergraduate R
- Page 78 and 79: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 80 and 81: TOWARD MOLECULAR RESOLUTION C-AFM W
- Page 82 and 83: SYNTHESIS AND CHARACTERIZATION OF E
- Page 84 and 85: My name is Aaron Montgomery and I a
- Page 86 and 87: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 88 and 89: 1,1-DIPHENYL-2,3,4,5-TETRAKIS(9,9-D
- Page 92 and 93: EFFECTS OF SURFACE CHEMISTRY ON CAD
- Page 94 and 95: 94 CMDITR Review of Undergraduate R
- Page 96 and 97: SYNTHESIS OF A POLYENE EO CHROMOPHO
- Page 98 and 99: SYNTHESIS OF A POLYENE EO CHROMOPHO
- Page 102 and 103: 102 CMDITR Review of Undergraduate
- Page 104 and 105: CHARACTERIZATION OF THE MOLECULAR P
- Page 106 and 107: 106 CMDITR Review of Undergraduate
- Page 108 and 109: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 110 and 111: OPTIMIZATION OF SEMICONDUCTOR NANOP
- Page 112 and 113: CHARACTERIZATION OF THE PHOTODECOMP
- Page 114 and 115: 114 CMDITR Review of Undergraduate
- Page 116 and 117: ELECTROLUMINESCENT PROPERTIES OF OR
- Page 118 and 119: 118 CMDITR Review of Undergraduate
- Page 120 and 121: DETERMINATION OF MOLECULAR ORIENTAT
- Page 122 and 123: DETERMINATION OF MOLECULAR ORIENTAT
- Page 124 and 125: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 126 and 127: HYDROGEL MATERIALS FOR TWO-PHOTON M
- Page 128 and 129: THE DESIGN OF A FLUID DELIVERY SYST
- Page 130: THE DESIGN OF A FLUID DELIVERY SYST