School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
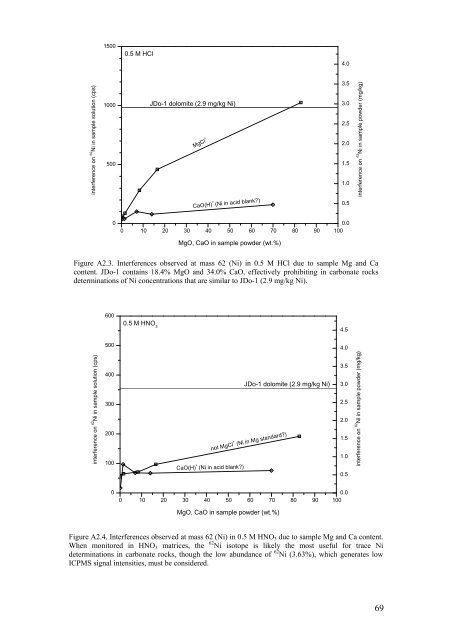
1500<br />
0.5 M HCl<br />
4.0<br />
interference on 62 Ni in sample solution (cps)<br />
1000<br />
500<br />
JDo-1 dolomite (2.9 mg/kg Ni)<br />
MgCl +<br />
CaO(H) + (Ni in acid blank?)<br />
3.5<br />
3.0<br />
2.5<br />
2.0<br />
1.5<br />
1.0<br />
0.5<br />
interference on 62 Ni in sample powder (mg/kg)<br />
0<br />
0.0<br />
0 10 20 30 40 50 60 70 80 90 100<br />
MgO, CaO in sample powder (wt.%)<br />
Figure A2.3. Interferences observed at mass 62 (Ni) in 0.5 M HCl due to sample Mg <strong>and</strong> Ca<br />
content. JDo-1 contains 18.4% MgO <strong>and</strong> 34.0% CaO, effectively prohibiting in carbonate rocks<br />
determinations <strong>of</strong> Ni concentrations that are similar to JDo-1 (2.9 mg/kg Ni).<br />
600<br />
4.5<br />
500<br />
4.0<br />
interference on 62 Ni in sample solution (cps)<br />
400<br />
300<br />
200<br />
100<br />
0.5 M HNO 3<br />
MgO, CaO in sample powder (wt.%)<br />
CaO(H) + (Ni in acid blank?)<br />
JDo-1 dolomite (2.9 mg/kg Ni)<br />
not MgCl + (Ni in Mg st<strong>and</strong>ard?)<br />
3.5<br />
3.0<br />
2.5<br />
2.0<br />
1.5<br />
1.0<br />
0.5<br />
interference on 62 Ni in sample powder (mg/kg)<br />
0<br />
0.0<br />
0 10 20 30 40 50 60 70 80 90 100<br />
Figure A2.4. Interferences observed at mass 62 (Ni) in 0.5 M HNO 3 due to sample Mg <strong>and</strong> Ca content.<br />
When monitored in HNO 3 matrices, the 62 Ni isotope is likely the most useful for trace Ni<br />
determinations in carbonate rocks, though the low abundance <strong>of</strong> 62 Ni (3.63%), which generates low<br />
ICPMS signal intensities, must be considered.<br />
69