School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Author's personal copy<br />
B.W. Alex<strong>and</strong>er et al. / Earth <strong>and</strong> Planetary <strong>Science</strong> Letters 283 (2009) 144–155<br />
147<br />
varying between 5 <strong>and</strong> 10 cm in length <strong>and</strong> labelled as IF1 to IF7. The<br />
entire length <strong>of</strong> sampled drill core is highly magnetic. The IF is finely<br />
laminated on millimeter scales <strong>and</strong> displays thicker b<strong>and</strong>ing on<br />
centimeter scales. The longest individual pieces <strong>of</strong> drill core were<br />
subdivided two or three times <strong>and</strong> these samples are identified as<br />
such, e.g., IF6, IF6a, <strong>and</strong> IF6b are individual samples from the ~10 cm<br />
long IF6 piece <strong>of</strong> drill core. This resulted in a total <strong>of</strong> fourteen samples,<br />
each representing relatively homogenous ~1 cm thick b<strong>and</strong>s that<br />
contained varying amounts <strong>of</strong> Fe. Samples were crushed with a steel<br />
jaw crusher <strong>and</strong> powdered with an agate ball mill. Major element<br />
concentrations were determined by X-ray fluorescence (XRF) at the<br />
SpectRAU laboratory <strong>of</strong> the <strong>University</strong> <strong>of</strong> Johannesburg, South Africa.<br />
Trace element concentrations were determined at <strong>Jacobs</strong> <strong>University</strong><br />
Bremen using inductively-coupled plasma mass spectrometry<br />
(ICPMS) following the methods described by Dulski (2001). Briefly,<br />
for both trace metal <strong>and</strong> Sm–Nd isotopic analyses, powdered samples<br />
were digested under pressure at high temperature (TN160 °C) in<br />
Teflon vessels using concentrated ultrapure perchloric (HClO 4 ) <strong>and</strong><br />
hydr<strong>of</strong>luoric (HF) acids. Following HClO 4 –HF evaporation the samples<br />
were re-dissolved in hydrochloric acid prior to ICPMS analysis or REE<br />
separation. Analytical quality for major <strong>and</strong> trace element determinations<br />
was monitored using the iron-formation st<strong>and</strong>ard FeR-2<br />
(Geological Survey <strong>of</strong> Canada), analysis <strong>of</strong> which produced good<br />
agreement with reference values (Table 1).<br />
Samples for Sm–Nd isotope determinations were spiked prior<br />
to dissolution with a mixed 147 Sm/ 150 Nd spike, <strong>and</strong> analyzed at the<br />
Laboratory for Isotope Geology (LIG), Swedish Museum <strong>of</strong> Natural<br />
History. Samarium <strong>and</strong> Nd were separated using ion exchange chromatography<br />
techniques described in Andersson et al. (2008). The<br />
isotopic ratios were determined with a Thermo Scientific TritonTIMS<br />
(thermal ionization mass spectrometer) using multicollector static<br />
mode. Samarium <strong>and</strong> Nd were loaded on double rhenium filaments. The<br />
total Nd blank averaged 43 pg for three separate digestion batches <strong>and</strong> is<br />
an insignificant contribution to the sample isotopic composition. Measured<br />
isotope ratios were reduced assuming exponential fractionation.<br />
Samarium ratios were normalized to 149 Sm/ 152 Sm = 0.516747. Neodymium<br />
was run in static mode using rotating gain compensation, <strong>and</strong><br />
calculated ratios were normalized to 146 Nd/ 144 Nd=0.7219.<br />
The La Jolla st<strong>and</strong>ard yielded 143 Nd/ 144 Nd=0.511848 ±0.0000047<br />
(2σ, n=32). No corrections were applied to the measured ratios <strong>and</strong><br />
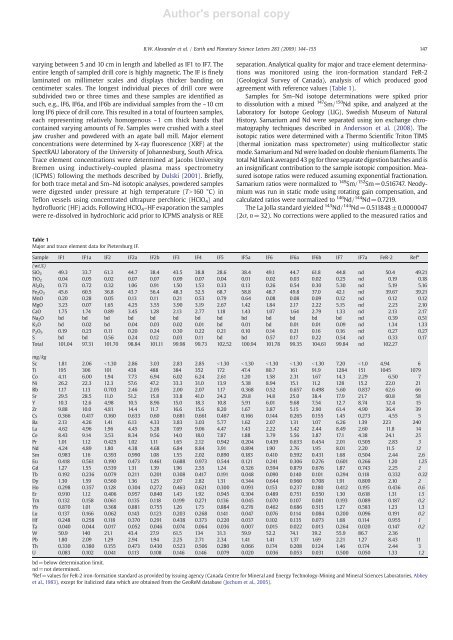
Table 1<br />
Major <strong>and</strong> trace element data for Pietersburg IF.<br />
Sample IF1 IF1a IF2 IF2a IF2b IF3 IF4 IF5 IF5a IF6 IF6a IF6b IF7 IF7a FeR-2 Ref a<br />
(wt.%)<br />
SiO 2 49.3 33.7 61.3 44.7 38.4 43.5 38.8 28.6 38.4 49.1 44.7 61.8 44.8 nd 50.4 49.21<br />
TiO 2 0.04 0.05 0.02 0.07 0.07 0.09 0.07 0.04 0.01 0.02 0.03 0.02 0.25 nd 0.19 0.18<br />
Al 2 O 3 0.73 0.72 0.32 1.06 0.91 1.50 1.53 0.33 0.13 0.26 0.54 0.30 5.30 nd 5.19 5.16<br />
Fe 2 O 3 45.6 60.5 36.8 43.7 56.4 48.3 52.5 68.7 58.8 48.7 49.8 37.0 42.1 nd 39.67 39.21<br />
MnO 0.20 0.28 0.05 0.13 0.11 0.21 0.53 0.79 0.64 0.08 0.08 0.09 0.12 nd 0.12 0.12<br />
MgO 3.23 0.07 1.65 4.25 3.55 3.90 3.19 2.67 1.42 1.84 2.17 2.22 5.15 nd 2.23 2.10<br />
CaO 1.75 1.74 0.89 3.45 1.28 2.13 2.77 1.18 1.43 1.07 1.64 2.79 1.33 nd 2.13 2.17<br />
Na 2 O bd bd bd bd bd bd bd bd bd bd bd bd bd nd 0.39 0.51<br />
K 2 O bd 0.02 bd 0.04 0.03 0.02 0.01 bd 0.01 bd 0.01 0.01 0.09 nd 1.34 1.33<br />
P 2 O 5 0.19 0.23 0.11 0.20 0.24 0.30 0.22 0.21 0.10 0.14 0.21 0.16 0.16 nd 0.27 0.27<br />
S bd bd 0.56 0.24 0.12 0.03 0.11 bd bd 0.57 0.17 0.22 0.54 nd 0.33 0.17<br />
Total 101.04 97.31 101.70 98.84 101.11 99.98 99.73 102.52 100.94 101.78 99.35 104.61 99.84 nd 102.27<br />
mg/kg<br />
Sc 1.81 2.06 b1.30 2.86 3.03 2.83 2.85 b1.30 b1.30 b1.30 b1.30 b1.30 7.20 b1.0 4.94 6<br />
Ti 195 306 101 438 488 384 352 172 47.4 80.7 161 91.9 1284 151 1045 1079<br />
Co 4.11 6.00 1.94 7.73 6.94 6.02 6.24 2.61 1.20 1.58 2.31 1.67 14.3 2.29 6.50 7<br />
Ni 26.2 22.3 12.3 57.6 47.2 33.3 31.0 13.9 5.38 8.94 15.1 11.2 128 15.2 22.0 21<br />
Rb 1.17 1.13 0.703 2.46 2.05 2.00 2.07 1.17 0.368 0.52 0.657 0.498 5.60 0.837 62.6 66<br />
Sr 29.5 28.5 11.0 51.2 15.8 33.8 41.0 24.2 29.8 14.8 25.0 38.4 17.9 21.7 60.8 58<br />
Y 10.3 12.6 4.98 10.5 8.96 15.0 18.3 10.8 5.91 6.01 9.68 7.54 12.7 8.74 12.4 15<br />
Zr 9.88 10.0 4.81 14.4 11.7 16.6 15.6 8.20 1.67 3.87 5.15 2.90 61.4 4.90 36.4 39<br />
Cs 0.366 0.417 0.160 0.633 0.60 0.681 0.661 0.467 0.166 0.144 0.265 0.155 1.45 0.273 4.55 5<br />
Ba 2.13 4.26 1.41 6.13 4.33 3.83 3.03 5.77 1.62 2.07 1.31 1.07 6.26 1.39 223 240<br />
La 4.62 4.96 1.96 4.45 5.28 7.69 9.06 4.47 1.43 2.22 3.42 2.44 8.49 2.60 11.8 14<br />
Ce 8.43 9.14 3.53 8.34 9.56 14.0 18.0 7.87 1.88 3.79 5.56 3.87 17.1 4.38 24.1 25<br />
Pr 1.01 1.12 0.425 1.02 1.11 1.65 2.12 0.942 0.204 0.439 0.633 0.454 2.01 0.505 2.83 3<br />
Nd 4.24 4.89 1.80 4.38 4.68 6.84 8.84 3.91 0.894 1.90 2.76 1.95 8.01 2.20 11.5 12<br />
Sm 0.983 1.16 0.393 0.990 1.08 1.55 2.02 0.890 0.183 0.410 0.592 0.431 1.68 0.504 2.44 2.6<br />
Eu 0.418 0.561 0.190 0.473 0.461 0.688 0.973 0.544 0.121 0.241 0.306 0.276 0.601 0.266 1.20 1.25<br />
Gd 1.27 1.55 0.539 1.31 1.39 1.96 2.55 1.24 0.326 0.594 0.879 0.676 1.87 0.743 2.25 2<br />
Tb 0.192 0.236 0.079 0.211 0.201 0.308 0.417 0.191 0.048 0.090 0.140 0.101 0.294 0.118 0.332 0.32<br />
Dy 1.30 1.59 0.560 1.36 1.25 2.07 2.82 1.31 0.344 0.644 0.960 0.708 1.91 0.809 2.10 2<br />
Ho 0.298 0.357 0.128 0.304 0.272 0.463 0.621 0.300 0.093 0.153 0.237 0.180 0.412 0.195 0.436 0.6<br />
Er 0.910 1.12 0.406 0.957 0.840 1.43 1.92 0.945 0.304 0.489 0.751 0.550 1.30 0.618 1.31 1.5<br />
Tm 0.132 0.158 0.061 0.135 0.118 0.199 0.271 0.136 0.045 0.070 0.107 0.081 0.193 0.089 0.187 0.2<br />
Yb 0.870 1.01 0.368 0.881 0.755 1.26 1.73 0.884 0.276 0.462 0.686 0.515 1.27 0.583 1.23 1.3<br />
Lu 0.137 0.166 0.062 0.143 0.123 0.203 0.268 0.141 0.047 0.076 0.114 0.084 0.200 0.096 0.191 0.2<br />
Hf 0.248 0.258 0.118 0.370 0.291 0.438 0.373 0.220 0.037 0.102 0.135 0.073 1.68 0.114 0.955 1<br />
Ta 0.040 0.044 0.017 0.052 0.046 0.074 0.064 0.036 0.007 0.015 0.022 0.013 0.264 0.020 0.147 0.2<br />
W 50.9 140 21.1 43.4 27.9 61.5 134 31.3 59.9 52.2 74.1 39.2 55.9 86.7 2.36<br />
Pb 1.80 2.09 1.29 2.94 1.94 2.25 2.71 2.34 1.41 1.41 1.37 1.69 2.21 1.27 8.43 11<br />
Th 0.330 0.380 0.155 0.473 0.430 0.523 0.506 0.280 0.066 0.174 0.208 0.124 1.46 0.174 2.44 3<br />
U 0.083 0.102 0.041 0.113 0.108 0.146 0.146 0.079 0.020 0.036 0.053 0.031 0.500 0.050 1.33 1.2<br />
bd=below determination limit.<br />
nd=not determined.<br />
a Ref =values for FeR-2 iron-formation st<strong>and</strong>ard as provided by issuing agency (Canada Centre for Mineral <strong>and</strong> Energy Technology-Mining <strong>and</strong> Mineral <strong>Science</strong>s Laboratories, Abbey<br />
et al., 1983), except for italicized data which are obtained from the GeoReM database (Jochum et al., 2005).