- Page 1 and 2:
A systematic review and economic mo
- Page 3 and 4:
A systematic review and economic mo
- Page 5 and 6:
Objectives: A systematic review was
- Page 7 and 8:
Glossary and list of abbreviations
- Page 9 and 10:
Glossary Absolute risk reduction Th
- Page 11 and 12:
Glossary continued Expected value o
- Page 13 and 14:
Glossary continued Progression-free
- Page 15:
List of abbreviations ASCO American
- Page 18 and 19:
xvi Executive summary of study desi
- Page 20 and 21:
xviii Executive summary prednisone/
- Page 23 and 24:
Description of underlying health pr
- Page 25 and 26:
● Patients with severe fluid rete
- Page 27 and 28:
Search strategy As stated in Chapte
- Page 29 and 30:
consensus and, if necessary, a thir
- Page 31 and 32:
Quantity of research available A to
- Page 33 and 34:
TABLE 1 Summary of included RCTs St
- Page 35 and 36:
TABLE 2 Treatment comparisons relat
- Page 37 and 38:
tumour response was reported for on
- Page 39 and 40:
TABLE 4 Summary results table for d
- Page 41 and 42:
decrease in PSA level (51 and 39% c
- Page 43 and 44:
Survival (%) 100 80 60 40 20 0 0 co
- Page 45 and 46:
TABLE 8 Summary results table for d
- Page 47 and 48:
TABLE 9 Grade 3 or 4 adverse events
- Page 49 and 50:
Also used was the core questionnair
- Page 51 and 52:
these, 69 patients had measurable t
- Page 53 and 54:
Effectiveness of mitoxantrone plus
- Page 55 and 56:
over to receive additional mitoxant
- Page 57 and 58:
Progression-free survival It is not
- Page 59 and 60:
compared with corticosteroids. The
- Page 61 and 62:
statistically significant improveme
- Page 63 and 64:
Summary of studies included in the
- Page 65 and 66:
TABLE 19 Percentage of costs by res
- Page 67 and 68:
TABLE 21 Summary of submission by S
- Page 69 and 70:
TABLE 24 Other in-trial costs a bod
- Page 71 and 72:
TABLE 27 Cost-effectiveness results
- Page 73 and 74:
Introduction The review of cost-eff
- Page 75 and 76:
TABLE 28 Regression coefficients fr
- Page 77 and 78:
cancer QoL estimation. Studies whic
- Page 79 and 80:
Localised Metastatic mHRPC Bennet (
- Page 81 and 82:
TABLE 34 Unit costs of drugs from t
- Page 83 and 84:
were assigned to the total follow-u
- Page 85 and 86:
Probability cost-effective given da
- Page 87 and 88:
Probability cost-effective given da
- Page 89 and 90:
TABLE 43 Utility values including/e
- Page 91 and 92:
was set to be 1.5 years based on th
- Page 93:
Population value of implementation/
- Page 96 and 97:
76 Discussion (CCI-NOV22) and least
- Page 98 and 99:
78 Discussion subsequent chemothera
- Page 101:
Rodolphe Perard received funding fr
- Page 104 and 105:
84 References Compendium; 2004. URL
- Page 106 and 107:
86 References (D) in patients (pts)
- Page 108 and 109:
88 References 100. Aventis Pharma.
- Page 110 and 111:
90 References low-dose prednisone i
- Page 112 and 113:
92 References 180. Kozloff M, Robin
- Page 114 and 115:
94 References locally advanced pros
- Page 116 and 117:
96 References 251. Scholz MC, Guess
- Page 119 and 120:
Clinical effectiveness Searching fo
- Page 121 and 122:
1. Prostatic Intraepithelial Neopla
- Page 123 and 124:
19. donataxel 20. doxetal 21. doxmi
- Page 125 and 126:
6. Mitozantrone.ti,ab. 7. Mitoxantr
- Page 127 and 128:
Texot or Trazoteva or Trixotene or
- Page 129 and 130:
1. (docetaxel OR asodecel OR doxeta
- Page 131 and 132:
10. (index of wellbeing or quality
- Page 133 and 134:
Appendix 2 Excluded studies Study d
- Page 135 and 136:
Study details Reason for exclusion
- Page 137 and 138:
Meeting: 2004 ASCO Annual Meeting C
- Page 139 and 140:
Studies of clinical effectiveness w
- Page 141 and 142: Using the method outlined by Parmar
- Page 143 and 144: Appendix 6 Data extraction tables
- Page 145 and 146: Study details and Participant detai
- Page 147 and 148: Study details and Participant detai
- Page 149 and 150: Study details and Participant detai
- Page 151 and 152: Study details and Participant detai
- Page 153 and 154: Study details and Participant detai
- Page 155 and 156: Study details and Participant detai
- Page 157 and 158: Study details and Participant detai
- Page 159 and 160: Study details and Participant detai
- Page 161 and 162: Study details and Participant detai
- Page 163 and 164: Study details and Participant detai
- Page 165 and 166: Study details and Participant detai
- Page 167 and 168: Study details and Participant detai
- Page 169 and 170: Study details and Participant detai
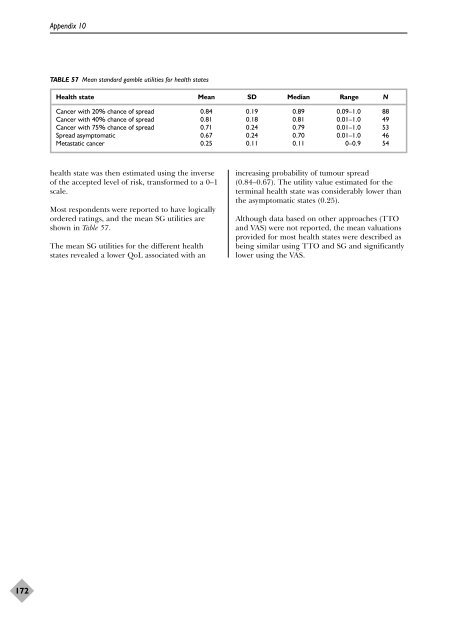
- Page 171 and 172: Study details and Participant detai
- Page 173 and 174: Study details and Participant detai
- Page 175: Study details and Participant detai
- Page 178 and 179: 158 Appendix 7 Ernst et al. 33 Qual
- Page 181 and 182: Appendix 9 © Queen’s Printer and
- Page 183 and 184: Sanofi-Aventis 61 Bloomfield et al.
- Page 185: Sanofi-Aventis 61 Bloomfield et al.
- Page 188 and 189: 168 Appendix 10 TABLE 51 Mean (SD)
- Page 190 and 191: 170 Appendix 10 TABLE 54 Distributi
- Page 194 and 195: 174 Appendix 11 TABLE 63 Drug and a
- Page 196 and 197: 176 Appendix 12 Scenario: moderate
- Page 198 and 199: 178 Appendix 13 ● You occasionall
- Page 202 and 203: Members Chair, Professor Tom Walley
- Page 204 and 205: Members Chair, Professor Bruce Camp
- Page 207: Feedback The HTA Programme and the