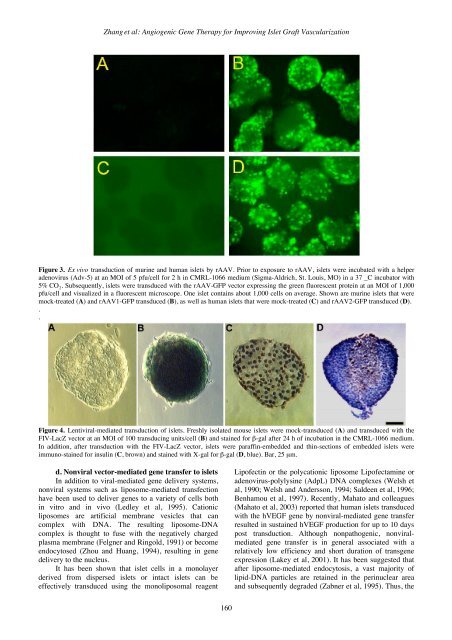
Zhang et al: Angiogenic <strong>Gene</strong> <strong>Therapy</strong> for Improving Islet Graft VascularizationFigure 3. Ex vivo transduction of murine and human islets by rAAV. Prior to exposure to rAAV, islets were incubated with a helperadenovirus (Adv-5) at an MOI of 5 pfu/cell for 2 h in CMRL-1066 medium (Sigma-Aldrich, St. Louis, MO) in a 37 _C incubator with5% CO 2 . Subsequently, islets were transduced with the rAAV-GFP vector expressing the green fluorescent protein at an MOI of 1,000pfu/cell and visualized in a fluorescent microscope. One islet contains about 1,000 cells on average. Shown are murine islets that weremock-treated (A) and rAAV1-GFP transduced (B), as well as human islets that were mock-treated (C) and rAAV2-GFP transduced (D)...Figure 4. Lentiviral-mediated transduction of islets. Freshly isolated mouse islets were mock-transduced (A) and transduced with theFIV-LacZ vector at an MOI of 100 transducing units/cell (B) and stained for β-gal after 24 h of incubation in the CMRL-1066 medium.In addition, after transduction with the FIV-LacZ vector, islets were paraffin-embedded and thin-sections of embedded islets wereimmuno-stained for insulin (C, brown) and stained with X-gal for β-gal (D, blue). Bar, 25 µm.d. Nonviral vector-mediated gene transfer to isletsIn addition to viral-mediated gene delivery systems,nonviral systems such as liposome-mediated transfectionhave been used to deliver genes to a variety of cells bothin vitro and in vivo (Ledley et al, 1995). Cationicliposomes are artificial membrane vesicles that cancomplex with DNA. The resulting liposome-DNAcomplex is thought to fuse with the negatively chargedplasma membrane (Felgner and Ringold, 1991) or becomeendocytosed (Zhou and Huang, 1994), resulting in genedelivery to the nucleus.It has been shown that islet cells in a monolayerderived from dispersed islets or intact islets can beeffectively transduced using the monoliposomal reagentLipofectin or the polycationic liposome Lipofectamine oradenovirus-polylysine (AdpL) DNA complexes (Welsh etal, 1990; Welsh and Andersson, 1994; Saldeen et al, 1996;Benhamou et al, 1997). Recently, Mahato and colleagues(Mahato et al, 2003) reported that human islets transducedwith the hVEGF gene by nonviral-mediated gene transferresulted in sustained hVEGF production for up to 10 dayspost transduction. Although nonpathogenic, nonviralmediatedgene transfer is in general associated with arelatively low efficiency and short duration of transgeneexpression (Lakey et al, 2001). It has been suggested thatafter liposome-mediated endocytosis, a vast majority oflipid-DNA particles are retained in the perinuclear areaand subsequently degraded (Zabner et al, 1995). Thus, the160
<strong>Gene</strong> <strong>Therapy</strong> and <strong>Molecular</strong> <strong>Biology</strong> Vol 7, page 161failure of DNA to leave the endosomal compartmentrepresents a major hurdle to liposome-mediated genetransfer. Nonviral-mediated gene transfer systems are of apreferred choice when persistent transgene expression isnot desirable.Recently, a novel system, known as proteintransduction, is being developed. Unlike gene transfersystems, this protein transduction system allows selectivedelivery of proteins into cells, when linked to a specificprotein transduction domain (PTD). PTD is a smallpeptide domain that can freely cross the cytoplasmicmembrane through a receptor-mediated process, which isindependent of ATP (Hawiger et al, 1999; Schwarze et al,2000). In particular, a PTD designated PTD-5, which isoriginally selected from an M13 phage peptide displaylibrary, has been reported to successfully transduce bothhuman and mouse islets without significant effects on isletfunction (Mi et al, 2000; Rehman et al, 2003). Likewise,Embury et al, (2001) also showed that a small peptide of11 amino acid residues that constitute the PTD of theHIV/TAT protein, when fused to β-galactosidase, is ableto transduce rat islets ex vivo with the fusion protein in adose-dependent manner at a relatively high efficiency.However, such a protein transduction system is normallyassociated with a transient effect, depending on therelative stability of the fusion protein. In addition, fortherapeutic protein delivery, caution should be taken toascertain that the fusion of a PTD does not adverselyaffect the proper folding and compromise the function ofthe therapeutic protein..III. ConclusionRapid re-establishment of an appropriatemicrovascular system in newly transplanted islets iscrucial for survival and function of islet grafts.Unfortunately, islets implanted at ectopic sites, such asunder the renal capsule or in the liver and spleen, areinvariably associated with markedly reducedvascularization, in comparison with native islets in thepancreas (Beger et al, 1998; Mattson et al, 2002). Thisimpairment in islet revascularization accounts at least inpart for the demand of sufficiently large quantities of isletmass for restoration of normoglycemia in type 1 diabeticsubjects. In addition, delayed and inadequate islet graftvascularization can deprive islets of oxygen and nutrients,causing islet cells to undergo cellular apoptosis andsubsequent cell death, particularly in the core of largeislets or in the center of aggregated islet clusters posttransplantation. Moreover, a lack of sufficient isletrevascularization may also compromise the optimalperformance of transplanted islets. Indeed, there areclinical data indicating that even after postabsorptiveblood glucose homeostasis is restored to normal post islettransplantation, implanted islets do not seem to function atoptimal levels, as reflected in their significantly impairedglucose tolerance in diabetic recipients in response tointravenous glucose challenge (Ryan et al, 2001, 2002).Thus, it is of great significance to define the molecularmechanism of islet revascularization and developtherapeutic angiogenesis approaches to enhance theprocess of islet revascularization. Such approaches areexpected to ensure adequate microvascular perfusion toislet cells and protect implanted islet cells from hypoxiainducedinflammation and necrosis, which will ultimatelyimprove the outcome of islet transplantation by reducingthe donor/recipient ratio thus increasing the success rate ofislet transplantation.AcknowledgementWe would like to thank Marcia Meseck for criticalreading of this manuscript. This project is supported partlyby the Juvenile Diabetes Research Center at Mount SinaiSchool of Medicine.ReferencesAhmet I, Sawa Y, Yamaguchi T, Matsuda H (2003) <strong>Gene</strong>transfer of hepatocyte growth factor improves angiogenesisand function of chronic ischemic myocardium in canineheart. Ann Thorac Surg 75, 1283-1287.Asplin IR, Wu SM, Mathew S, Bhattacharjee G, Pizzo SV (2001)Differential regulation of the fibroblast growth factor (FGF)family by alpha(2)-macroglobulin: evidence for selectivemodulation of FGF-2-induced angiogenesis. Blood 97, 3450-3457.Bacharach E, Itin A, Keshet E (1992) In vitro patterns ofexpression of urokinase and its inhibitor PA-1 suggest aconcerted role in regulating physiological angiogenesis. ProcNatl Acad Sci U S A. 89, 10686-10690.Beger C, Cirulli V, Vajkoczy P, Halban PA, Menger MD (1998)Vascularization of purified pancreatic islet-like cellaggregates (pseudoislets) after syngerneic transplantation.Diabetes 47, 559-565.Benhamou PY, Moriscot C, Prevost P, Rolland E, Hallmi S,Chroboczek J (1997) Standardization of procedure forefficient ex vivo gene transfer into porcine pancreatic isletswith cationic liposomes. Transplantation 63, 1798-1803, .Bikfalvi A, Klein S, Pintucci G, Rifkin DB (1997) Biologicalroles of fibroblast growth factor-2. Endocrine Reviews 18,26-45.Bloomgarden ZT (1998) The Epidemiology of Complications.Diabetes Care 25, 924-932.Bloomgarden ZT (2002) Treatment Issues in Type 1 Diabetes.Diabetes Care 25, 230-238.Boker A, Rothenberg L, Hernandez C, Kenyon NS, Ricordi C,Alejandro R (2001) Human islet transplantation: update.World J Surg 25, 481-486.Brissova M, Fowler M, Shiota M, Radhika A, Shostak A, WiebeP, Gannon M, Powers A (2003) Intra-islet endothelial cellscontributor to revascularization of transplanted pancreaticislets. Diabetes 52 (suppl. 1), A13.Bussolino F, Di Renzo MF, Ziche M, Bocchietto E, Olivero M,Naldini L, Gaudino G, Tamagnone L, Coffer A, ComoglioPM (1992) Hepatocyte growth factor is a potent angiogenicfactor which stimulates endothelial cell motility and growth.J Cell Biol 119, 629-64.Carlsson P-O, Palm F, Andersson A, Liss P (2000) Chronicallydecreased oxygen tension in rat pancreatic islets transplantedunder the kidney capsule. Transplantation 69, 761-766.Carlsson P-O, Palm F, Andersson A, Liss P (2001) Markedlydecreased oxygen tension in transplanted rat pancreatic isletsirrespective of implantation sites. Diabetes 50, 489-495.161