<strong>Cladosporium</strong> herbarum species complex Belgium, isolated from Quercus robur (Fagaceae), <strong>CBS</strong> 157.82; Kampenhout, isolated from Hordeum vulgare (Poaceae), 26 June 2005, J.Z. Groenewald, <strong>CBS</strong>-H 19856, neotype designated here of C. bruhnei, isoneotype HAL 2023 F, cultures ex-type <strong>CBS</strong> 121624 = CPC 12211, CPC 12212. Czech Republic, Lisen, isolated from Polygonatum odoratum (Liliaceae), <strong>CBS</strong> 813.71, albino mutant of <strong>CBS</strong> 812.71. Germany, <strong>CBS</strong> 134.31 = ATCC 11283 = IMI 049632; Nordrhein-Westfalen, Mühlheim an der Ruhr, isolated from industrial water, IWW 727, <strong>CBS</strong> 110024; Sachsen-Anhalt, Halle (Saale), Robert-Franz-Ring, isolated from leaves of Tilia cordata (Tiliaceae), 2004, K. Schubert, CPC 11386. Netherl<strong>and</strong>s, isolated from air, <strong>CBS</strong> 521.68; isolated from Hordeum vulgare, 1 Jan. 2005, P.W. Crous, CPC 12139; isolated from man, skin, <strong>CBS</strong> 159.54 = ATCC 36948; Amsterdam, isolated from Thuja tincture, <strong>CBS</strong> 177.71; Geleen, St. Barbara Ziekenhuis, isolated from man, skin, <strong>CBS</strong> 366.80, <strong>CBS</strong> 399.80; isolated from man, sputum, Aug. 1955, <strong>CBS</strong> 161.55. New Zeal<strong>and</strong>, Otago, Lake Harris, isolated from Ourisia macrophylla (Scrophulariaceae), 30 Jan. 2005, A. Blouin, Hill 1135, CPC 11840. Russia, Moscow region, isolated from Polyporus radiatus (Polyporaceae), Oct. 1978, <strong>CBS</strong> 572.78 = VKM F-405. Slovenia, Ljubljana, isolated from an air conditioning system, 2004, M. Butala, EXF-680 = CPC 12046; Sečovlje, isolated from hypersaline water from salterns (reserve pond), 2005, P. Zalar, EXF-389 = CPC 12042. Spain, Ebro Delta, isolated from hypersaline water from salterns (crystallisation pond), 2004, P. Zalar, EXF-594 = CPC 12045. Sweden, Skåne, on tip blight of living leaves of Allium sp. (Alliaceae), Fr. no. F-09810, UPS- FRIES, holotype of Davidiella allicina. U.S.A., New York, Geneva, isolated from CCA-treated Douglas-fir pole, <strong>CBS</strong> 115683 = ATCC 66670 = CPC 5101. Substrate <strong>and</strong> distribution: Living <strong>and</strong> decaying plant material, man, air, hypersaline <strong>and</strong> industrial water; widespread. Literature: Saccardo (1899: 1076), Linder (1947: 289). Fig. 10. <strong>Cladosporium</strong> bruhnei (CPC 12211). A. Conidiophore with characteristic long secondary ramoconidium <strong>and</strong> complex conidiophore. B. Detail of hila on secondary ramoconidia. C. Details of prominent ornamentation on conidia. Scale bars: A = 10 µm, B = 2 µm, C = 5 µm. Cultural characteristics: Colonies on PDA reaching 22–32 mm diam after 14 d at 25 ºC, olivaceous-grey to iron-grey, sometimes whitish, smoke-grey to pale olivaceous due to abundant aerial mycelium covering almost the whole colony, with age collapsing becoming olivaceous-grey, occasionally zonate, velvety to floccose, margin narrow, entire edge, white, glabrous to somewhat feathery, aerial mycelium sparse to abundant, white, fluffy, growth regular, flat to low convex, sometimes forming few exudates in the colony centre, sporulating. Colonies on MEA reaching 21–32 mm diam after 14 d at 25 ºC, grey-olivaceous, olivaceous-grey to dull green or irongrey, sometimes whitish to pale smoke-grey due to abundant aerial mycelium, olivaceous-grey to iron-grey reverse, velvety, margin narrow, entire edge to slightly undulate, white, radially furrowed, glabrous to slightly feathery, aerial mycelium sparse to abundant, mainly in the centre, white, fluffy, growth convex to raised, radially furrowed, distinctly wrinkled in the colony centre, without prominent exudates, sporulating. Colonies on OA reaching 20–32 mm diam after 14 d at 25 ºC, smoke-grey, grey-olivaceous to olivaceous-grey, greenish black or iron-grey reverse, margin narrow, entire edge, colourless to white, glabrous, aerial mycelium sparse to abundant, dark smoke-grey, diffuse, high, later collapsed, felty, growth flat, without prominent exudates, sporulation profuse. Specimens examined: Sine loco et dato, <strong>CBS</strong> 188.54 = ATCC 11290 = IMI 049638. Australia, N.S.W., Barrington Tops National Park, isolated from leaves of Eucalyptus stellulata (Myrtaceae), 3 Jan. 2006, B. Summerell, CPC 12921. Fig. 11. Davidiella allicina (F-09810, UPS-FRIES, holotype). Ascus <strong>and</strong> ascospores. Scale bar = 10 µm. P.W. Crous del. www.studiesinmycology.org 119
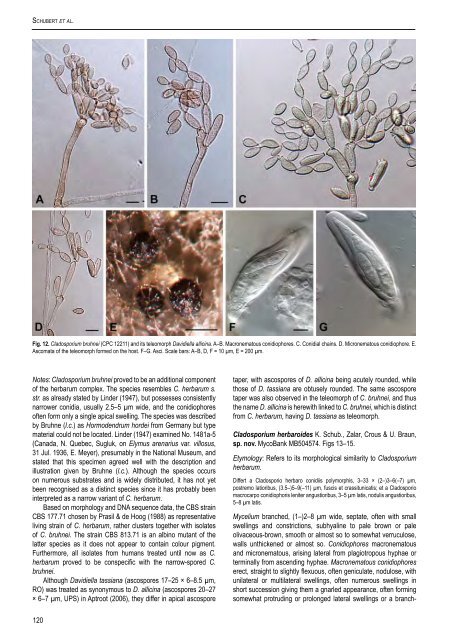
Schubert et al. Fig. 12. <strong>Cladosporium</strong> bruhnei (CPC 12211) <strong>and</strong> its teleomorph Davidiella allicina. A–B. Macronematous conidiophores. C. Conidial chains. D. Micronematous conidiophore. E. Ascomata of the teleomorph formed on the host. F–G. Asci. Scale bars: A–B, D, F = 10 µm, E = 200 µm. Notes: <strong>Cladosporium</strong> bruhnei proved to be an additional component of the herbarum complex. <strong>The</strong> species resembles C. herbarum s. str. as already stated by Linder (1947), but possesses consistently narrower conidia, usually 2.5–5 µm wide, <strong>and</strong> the conidiophores often form only a single apical swelling. <strong>The</strong> species was described by Bruhne (l.c.) as Hormodendrum hordei from Germany but type material could not be located. Linder (1947) examined No. 1481a-5 (Canada, N. Quebec, Sugluk, on Elymus arenarius var. villosus, 31 Jul. 1936, E. Meyer), presumably in the National Museum, <strong>and</strong> stated that this specimen agreed well with the description <strong>and</strong> illustration given by Bruhne (l.c.). Although the species occurs on numerous substrates <strong>and</strong> is widely distributed, it has not yet been recognised as a distinct species since it has probably been interpreted as a narrow variant of C. herbarum. Based on morphology <strong>and</strong> DNA sequence data, the <strong>CBS</strong> strain <strong>CBS</strong> 177.71 chosen by Prasil & de Hoog (1988) as representative living strain of C. herbarum, rather clusters together with isolates of C. bruhnei. <strong>The</strong> strain <strong>CBS</strong> 813.71 is an albino mutant of the latter species as it does not appear to contain colour pigment. Furthermore, all isolates from humans treated until now as C. herbarum proved to be conspecific with the narrow-spored C. bruhnei. Although Davidiella tassiana (ascospores 17–25 × 6–8.5 µm, RO) was treated as synonymous to D. allicina (ascospores 20–27 × 6–7 µm, UPS) in Aptroot (2006), they differ in apical ascospore taper, with ascospores of D. allicina being acutely rounded, while those of D. tassiana are obtusely rounded. <strong>The</strong> same ascospore taper was also observed in the teleomorph of C. bruhnei, <strong>and</strong> thus the name D. allicina is herewith linked to C. bruhnei, which is distinct from C. herbarum, having D. tassiana as teleomorph. <strong>Cladosporium</strong> herbaroides K. Schub., Zalar, Crous & U. Braun, sp. nov. MycoBank MB504574. Figs 13–15. Etymology: Refers to its morphological <strong>similar</strong>ity to <strong>Cladosporium</strong> herbarum. Differt a Cladosporio herbaro conidiis polymorphis, 3–33 × (2–)3–6(–7) µm, postremo latioribus, (3.5–)5–9(–11) µm, fuscis et crassitunicatis; et a Cladosporio macrocarpo conidiophoris leniter angustioribus, 3–5 µm latis, nodulis angustioribus, 5–8 µm latis. Mycelium branched, (1–)2–8 µm wide, septate, often with small swellings <strong>and</strong> constrictions, subhyaline to pale brown or pale olivaceous-brown, smooth or almost so to somewhat verruculose, walls unthickened or almost so. Conidiophores macronematous <strong>and</strong> micronematous, arising lateral from plagiotropous hyphae or terminally from ascending hyphae. Macronematous conidiophores erect, straight to slightly flexuous, often geniculate, nodulose, with unilateral or multilateral swellings, often numerous swellings in short succession giving them a gnarled appearance, often forming somewhat protruding or prolonged lateral swellings or a branch- 120
- Page 1:
Studies in Mycology 58 (2007) The g
- Page 4 and 5:
Studies in Mycology The Studies in
- Page 7 and 8:
CONTENTS P.W. Crous, U. Braun and J
- Page 9 and 10:
lectotype for the genus by Clements
- Page 11:
Schubert K (2005a). Morphotaxonomic
- Page 14 and 15:
Crous et al. Table 1. Isolates for
- Page 16 and 17:
Crous et al. Table 1. (Continued).
- Page 18 and 19:
Crous et al. 100 10 changes 65 100
- Page 20 and 21:
Crous et al. Treatment of phylogene
- Page 22 and 23:
Crous et al. Teratosphaeria bellula
- Page 24 and 25:
Crous et al. 6. Conidiophores short
- Page 26 and 27:
Crous et al. Habit plant pathogenic
- Page 28 and 29:
Crous et al. Fig. 7. Catenulostroma
- Page 30 and 31:
Crous et al. system, consisting of
- Page 32 and 33:
Crous et al. Fig. 9. Penidiella col
- Page 34 and 35:
Crous et al. Fig. 12. Penidiella ri
- Page 36 and 37:
Crous et al. conidiophores, about 1
- Page 38 and 39:
Crous et al. have conidiomata rangi
- Page 40 and 41:
Crous et al. Schizothyriaceae clade
- Page 42 and 43:
Crous et al. Fig. 21. Stigmidium sc
- Page 44 and 45:
Crous et al. Gams W, Verkley GJM, C
- Page 46 and 47:
Crous et al. Table 1. Isolates for
- Page 48 and 49:
Crous et al. 100 61 100 52 100 10 c
- Page 50 and 51:
Crous et al. Fig. 3. Rachicladospor
- Page 52 and 53:
Crous et al. Fig. 4. Toxicocladospo
- Page 54 and 55:
Crous et al. Fig. 5. Verrucocladosp
- Page 56 and 57:
Crous et al. Fig. 7. Stenella aragu
- Page 58 and 59:
Crous et al. Helotiales, incertae s
- Page 60 and 61:
Crous et al. Fig. 10. Ochrocladospo
- Page 62 and 63:
Crous et al. Fig. 12. Rhizocladospo
- Page 64 and 65:
Crous et al. 7. Conidiophores unbra
- Page 66 and 67:
Crous et al. 33. Terminal conidioge
- Page 68 and 69:
Crous et al. Crous PW, Kang JC, Bra
- Page 70 and 71:
Arzanlou et al. To date 26 species
- Page 72 and 73:
Arzanlou et al. Table 1. (Continued
- Page 74 and 75:
Arzanlou et al. 10 changes Athelia
- Page 76 and 77:
Arzanlou et al. Athelia epiphylla A
- Page 78 and 79:
Arzanlou et al. Fig. 3. Periconiell
- Page 80 and 81: Arzanlou et al. Cultural characteri
- Page 82 and 83: Arzanlou et al. Fig. 10. A. Ramichl
- Page 84 and 85: Arzanlou et al. Ramichloridium musa
- Page 86 and 87: Arzanlou et al. Cultural characteri
- Page 88 and 89: Arzanlou et al. Fig. 20. Rhinocladi
- Page 90 and 91: Arzanlou et al. Fig. 22. Rhinocladi
- Page 92 and 93: Arzanlou et al. Rhinocladiella mack
- Page 94 and 95: Arzanlou et al. Fig. 26. Veronaea c
- Page 96 and 97: Arzanlou et al. Cultural characteri
- Page 98 and 99: Arzanlou et al. Fig. 30. Myrmecridi
- Page 100 and 101: Arzanlou et al. Fig. 32. Radulidium
- Page 102 and 103: Arzanlou et al. Fig. 34. Rhodoveron
- Page 104 and 105: Arzanlou et al. even further, thoug
- Page 106 and 107: available online at www.studiesinmy
- Page 108 and 109: Dichocladosporium gen. nov. 10 chan
- Page 110 and 111: Dichocladosporium gen. nov. Fig. 3.
- Page 112 and 113: Dichocladosporium gen. nov. to intr
- Page 114 and 115: Dichocladosporium gen. nov. Czechos
- Page 116 and 117: available online at www.studiesinmy
- Page 118 and 119: Cladosporium herbarum species compl
- Page 120 and 121: Cladosporium herbarum species compl
- Page 122 and 123: Cladosporium herbarum species compl
- Page 124 and 125: Cladosporium herbarum species compl
- Page 126 and 127: Cladosporium herbarum species compl
- Page 128 and 129: Cladosporium herbarum species compl
- Page 132 and 133: Cladosporium herbarum species compl
- Page 134 and 135: Cladosporium herbarum species compl
- Page 136 and 137: Cladosporium herbarum species compl
- Page 138 and 139: Cladosporium herbarum species compl
- Page 140 and 141: Cladosporium herbarum species compl
- Page 142 and 143: Cladosporium herbarum species compl
- Page 144 and 145: Cladosporium herbarum species compl
- Page 146 and 147: Cladosporium herbarum species compl
- Page 148 and 149: Cladosporium herbarum species compl
- Page 150 and 151: Cladosporium herbarum species compl
- Page 152 and 153: Cladosporium herbarum species compl
- Page 154 and 155: Cladosporium herbarum species compl
- Page 156 and 157: Cladosporium herbarum species compl
- Page 158 and 159: Cladosporium herbarum species compl
- Page 160 and 161: Cladosporium herbarum species compl
- Page 162 and 163: Cladosporium herbarum species compl
- Page 164 and 165: Cladosporium herbarum species compl
- Page 166 and 167: Cladosporium herbarum species compl
- Page 168 and 169: available online at www.studiesinmy
- Page 170 and 171: Cladosporium sphaerospermum species
- Page 172 and 173: Cladosporium sphaerospermum species
- Page 174 and 175: Cladosporium sphaerospermum species
- Page 176 and 177: Cladosporium sphaerospermum species
- Page 178 and 179: Cladosporium sphaerospermum species
- Page 180 and 181:
Cladosporium sphaerospermum species
- Page 182 and 183:
Cladosporium sphaerospermum species
- Page 184 and 185:
Cladosporium sphaerospermum species
- Page 186 and 187:
Cladosporium sphaerospermum species
- Page 188 and 189:
Cladosporium sphaerospermum species
- Page 190 and 191:
Cladosporium sphaerospermum species
- Page 192 and 193:
Cladosporium sphaerospermum species
- Page 194 and 195:
Cladosporium sphaerospermum species
- Page 196 and 197:
Crous et al. The aim of the present
- Page 198 and 199:
Crous et al. 10 changes Athelia epi
- Page 200 and 201:
Crous et al. Table 1. Isolates used
- Page 202 and 203:
Crous et al. Table 1. (Continued).
- Page 204 and 205:
Crous et al. 0.1 expected changes p
- Page 206 and 207:
Crous et al. Fig. 6. Cladophialopho
- Page 208 and 209:
Crous et al. Fig. 11. Cladophialoph
- Page 210 and 211:
Crous et al. Fig. 13. Cladophialoph
- Page 212 and 213:
Crous et al. Fig. 15. Exophiala sp.
- Page 214 and 215:
Crous et al. Fig. 18. Cylindrosympo
- Page 216 and 217:
Crous et al. Fig. 19. Fusicladium a
- Page 218 and 219:
Crous et al. Fig. 22. Fusicladium f
- Page 220 and 221:
Crous et al. Fig. 24. Fusicladium p
- Page 222 and 223:
Crous et al. Fusicladium rhodense C
- Page 224 and 225:
Crous et al. Excluded taxa Polyscyt
- Page 226 and 227:
Crous et al. the conidial tips are
- Page 228 and 229:
available online at www.studiesinmy
- Page 230 and 231:
Cladophialophora carrionii complex
- Page 232 and 233:
Cladophialophora carrionii complex
- Page 234 and 235:
Cladophialophora carrionii complex
- Page 236 and 237:
Cladophialophora carrionii complex
- Page 238 and 239:
Cladophialophora carrionii complex
- Page 240 and 241:
Cladophialophora carrionii complex
- Page 242 and 243:
Cladophialophora carrionii complex
- Page 244 and 245:
available online at www.studiesinmy
- Page 246 and 247:
Hormoconis resinae and morphologica
- Page 248 and 249:
Hormoconis resinae and morphologica
- Page 250 and 251:
Hormoconis resinae and morphologica
- Page 252 and 253:
Fig. 6 Hormoconis resinae and morph
- Page 254 and 255:
Hormoconis resinae and morphologica
- Page 256 and 257:
Cladophialophora, 52 k , 54 k -55,
- Page 258 and 259:
Fusicladium phillyreae, 189 c , 191
- Page 260 and 261:
Ramichloridium epichloës, 60, 89 P
- Page 262:
Trimmatostroma salicis, 3 t , 5, 6