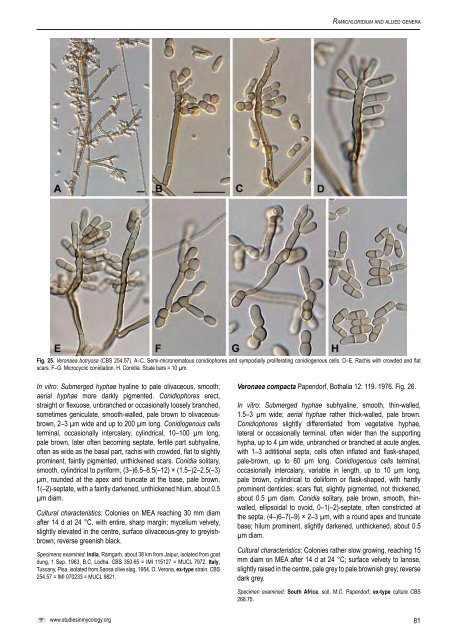
Arzanlou et al. Rhinocladiella mackenziei (C.K. Campb. & Al-Hedaithy) Arzanlou & Crous, comb. nov. MycoBank MB504554. Fig. 22. Basionym: Ramichloridium mackenziei C.K. Campb. & Al-Hedaithy, J. Med. Veterin. Mycol. 31: 330. 1993. In vitro: Submerged hyphae subhyaline, smooth, thin-walled, 2–3 µm wide; aerial hyphae pale brown, slightly narrower. Conidiophores slightly or not differentiated from vegetative hyphae, arising laterally from aerial hyphae, with one or two additional septa, often reduced to a discrete or intercalary conidiogenous cell, pale-brown, 10–25 × 2.5–3.5 µm. Conidiogenous cells terminal or intercalary, variable in length, 5–15 µm long <strong>and</strong> 3–5 µm wide, occasionally slightly wider than the basal part, pale brown, rachis with slightly prominent, unpigmented, non-thickened scars, about 0.5 µm diam. Conidia golden-brown, thin-walled, smooth, ellipsoidal to obovate, subcylindical, (5–)8–9(–12) × (2–)3–3.5(–5) µm, with darkened, inconspicously thickened, protuberant or truncate hilum, less than 1 µm diam. Cultural characteristics: Colonies on MEA reaching 5 mm diam after 14 d at 24 °C, with entire, smooth, sharp margin; mycelium densely lanose <strong>and</strong> elevated in the centre, olivaceous-green to brown; reverse dark olivaceous. Specimens examined: Israel, Haifa, isolated from brain abscess, <strong>CBS</strong> 368.92 = UTMB 3170; human brain abscess, E. Lefler, <strong>CBS</strong> 367.92 = NCPF 2738 = UTMB 3169. Saudi Arabia, from phaeohyphomycosis of the brain, S.S.A. Al-Hedaithy, ex-type strain, <strong>CBS</strong> 650.93 = MUCL 40057 = NCPF 2808; from brain abscess, Pakistani male who travelled to Saudi Arabia, <strong>CBS</strong> 102592 = NCPF 7460. United Arab Emirates, from fatal brain abscess, <strong>CBS</strong> 102590 = NCPF 2853. Notes: Morphologically Rhinocladiella mackenziei is somewhat <strong>similar</strong> to Pleurothecium obovoideum (Matsush.) Arzanlou & Crous, which was originally isolated from dead wood. However, P. obovoideum has distinct conidiophores, <strong>and</strong> the ascending hyphae are thick-walled, <strong>and</strong> the denticles cylindrical, up to 1.5 µm long. In contrast, Rh. mackenziei has only slightly prominent denticles. Rhinocladiella mackenziei is a member of the Chaetothyriales, while P. obovoideum clusters in the Chaetosphaeriales. Thysanorea Arzanlou, W. Gams & Crous, gen. nov. MycoBank MB504555. Etymology: (Greek) thysano = brush, referring to the brush-like branching pattern, suffix derived from Veronaea. Veronaeae similis sed conidiophoris partim Periconiae similibus dense ramosis distinguenda. In vitro: Submerged hyphae subhyaline, smooth, thin-walled; aerial hyphae pale brown, smooth or verrucose. Conidiophores dimorphic; micronematous conidiophores slightly differentiated from vegetative hyphae, branched or simple, multiseptate. Conidiogenous cells terminal, polyblastic, variable in length, smooth, golden- to dark brown at the base, paler towards the apex, later sometimes inconspicuously septate; fertile part often wider than the basal part, clavate to doliiform, with crowded, more or less prominent conidium-bearing denticles, unpigmented, but slightly thickened. Macronematous conidiophores consisting of well-differentiated, thick-walled, dark brown stalks; apically repeatedly densely branched, forming a complex head, each branchlet giving rise to a conidium-bearing denticulate rachis with slightly pigmented, thickened scars. Conidia of both kinds of conidiophore formed singly, smooth, pale brown, obovoidal to pyriform, (0–)1-septate, with a truncate base <strong>and</strong> darkened hilum; conidial secession schizolytic. Type species: Thysanorea papuana (Aptroot) Arzanlou, W. Gams & Crous, comb. nov. Thysanorea papuana (Aptroot) Arzanlou, W. Gams & Crous, comb. nov. MycoBank MB504556. Figs 7C, 23–24. Basionym: Periconiella papuana Aptroot, Nova Hedwigia 67: 491. 1998. In vitro: Submerged hyphae subhyaline, smooth, thin-walled, 1.5–3 µm wide; aerial hyphae pale brown, smooth to verrucose, 1.5–2 µm wide. Conidiophores dimorphic; micronematous conidiophores slightly differentiated from vegetative hyphae, branched or simple, up to 6-septate. Conidiogenous cells terminal or intercalary, variable in length, 5–20 µm long, thin-walled, smooth, golden- to dark brown at the base, paler toward the apex, later sometimes becoming inconspicuously septate, fertile part wider than basal part, often clavate, with crowded, more or less prominent conidium-bearing denticles, about 1 µm diam, unpigmented but slightly thickened. Conidia solitary, subhyaline, thin-walled, smooth, cylindrical to pyriform, rounded at the apex <strong>and</strong> truncate at the base, pale brown, (0–)1-septate, (5–)7–8(–11) × (2–)3(–4) µm, with a truncate base <strong>and</strong> darkened hilum, 1 µm diam. Macronematous conidiophores present in old cultures after 1 mo of incubation, consisting of welldifferentiated, thick-walled, dark brown stalks, up to 220 µm long, (4–)5–6(–7) µm wide, with up to 15 additional septa, often with inflated basal cells; apically densely branched, forming a complex head, with up to five levels of branchlets, 20–50 µm long, each branchlet giving rise to a denticulate conidium-bearing rachis; scars slightly pigmented, thickened, about 1 µm diam. Conidia solitary, thin-walled, smooth, pale brown, obovoidal to pyriform, (0–)1- septate, (4–)5–6(–8) × (2–)3(–4) µm, with a truncate base <strong>and</strong> darkened hilum, 1–2 µm diam. Cultural characteristics: Colonies on MEA reaching 10 mm diam after 14 d at 24 °C, with entire, sharp margin; mycelium velvety, elevated, with colonies up to 2 mm high, surface olivaceous-grey to iron-grey; reverse greenish black. Specimen examined: Papua New Guinea, Madang Province, foothill of Finisterre range, 40.8 km along road Madang-Lae, alt. 200 m, isolated from unknown stipe, 2 Nov. 1995, A. Aptroot, holotype <strong>CBS</strong>-H 6351, culture ex-type <strong>CBS</strong> 212.96. Veronaea Cif. & Montemart., Atti Ist. Bot. Lab. Crittog. Univ. Pavia, sér. 5, 15: 68. 1957. In vitro: Colonies velvety, pale olivaceous-brown, moderately fast-growing. Submerged hyphae hyaline to pale olivaceous, smooth; aerial hyphae, more darkly pigmented. Exophiala-type budding cells absent in culture. Conidiophores erect, straight or flexuose, unbranched or occasionally loosely branched, sometimes geniculate, smooth-walled, pale to medium- or olivaceous-brown. Conidiogenous cells terminally integrated, polyblastic, occasionally intercalary, cylindrical, pale brown, later often becoming septate, fertile part subhyaline, often as wide as the basal part, rachis with crowded, flat to slightly prominent, faintly pigmented, unthickened scars. Conidia solitary, smooth, cylindrical to pyriform, rounded at the apex <strong>and</strong> truncate at the base, pale brown, 1(–2)-septate; conidial secession schizolytic. Type species: Veronaea botryosa Cif. & Montemart., Atti Ist. Bot. Lab. Crittog. Univ. Pavia, sér. 5, 15: 68. 1957. Veronaea botryosa Cif. & Montemart., Atti Ist. Bot. Lab. Crittog. Univ. Pavia, sér. 5, 15: 68. 1957. Fig. 25. 80
Ramichloridium <strong>and</strong> allied genera Fig. 25. Veronaea botryosa (<strong>CBS</strong> 254.57). A–C. Semi-micronematous conidiophores <strong>and</strong> sympodially proliferating conidiogenous cells. D–E. Rachis with crowded <strong>and</strong> flat scars. F–G. Microcyclic conidiation. H. Conidia. Scale bars = 10 µm. In vitro: Submerged hyphae hyaline to pale olivaceous, smooth; aerial hyphae more darkly pigmented. Conidiophores erect, straight or flexuose, unbranched or occasionally loosely branched, sometimes geniculate, smooth-walled, pale brown to olivaceousbrown, 2–3 µm wide <strong>and</strong> up to 200 µm long. Conidiogenous cells terminal, occasionally intercalary, cylindrical, 10–100 µm long, pale brown, later often becoming septate, fertile part subhyaline, often as wide as the basal part, rachis with crowded, flat to slightly prominent, faintly pigmented, unthickened scars. Conidia solitary, smooth, cylindrical to pyriform, (3–)6.5–8.5(–12) × (1.5–)2–2.5(–3) µm, rounded at the apex <strong>and</strong> truncate at the base, pale brown, 1(–2)-septate, with a faintly darkened, unthickened hilum, about 0.5 µm diam. Cultural characteristics: Colonies on MEA reaching 30 mm diam after 14 d at 24 °C, with entire, sharp margin; mycelium velvety, slightly elevated in the centre, surface olivaceous-grey to greyishbrown; reverse greenish black. Specimens examined: India, Ramgarh, about 38 km from Jaipur, isolated from goat dung, 1 Sep. 1963, B.C. Lodha, <strong>CBS</strong> 350.65 = IMI 115127 = MUCL 7972. Italy, Tuscany, Pisa, isolated from Sansa olive slag, 1954, O. Verona, ex-type strain, <strong>CBS</strong> 254.57 = IMI 070233 = MUCL 9821. Veronaea compacta Papendorf, Bothalia 12: 119. 1976. Fig. 26. In vitro: Submerged hyphae subhyaline, smooth, thin-walled, 1.5–3 µm wide; aerial hyphae rather thick-walled, pale brown. Conidiophores slightly differentiated from vegetative hyphae, lateral or occasionally terminal, often wider than the supporting hypha, up to 4 µm wide, unbranched or branched at acute angles, with 1–3 adititional septa, cells often inflated <strong>and</strong> flask-shaped, pale-brown, up to 60 µm long. Conidiogenous cells terminal, occasionally intercalary, variable in length, up to 10 µm long, pale brown, cylindrical to doliiform or flask-shaped, with hardly prominent denticles; scars flat, slightly pigmented, not thickened, about 0.5 µm diam. Conidia solitary, pale brown, smooth, thinwalled, ellipsoidal to ovoid, 0–1(–2)-septate, often constricted at the septa, (4–)6–7(–9) × 2–3 µm, with a round apex <strong>and</strong> truncate base; hilum prominent, slightly darkened, unthickened, about 0.5 µm diam. Cultural characteristics: Colonies rather slow growing, reaching 15 mm diam on MEA after 14 d at 24 °C; surface velvety to lanose, slightly raised in the centre, pale grey to pale brownish grey; reverse dark grey. Specimen examined: South Africa, soil, M.C. Papendorf, ex-type culture <strong>CBS</strong> 268.75. www.studiesinmycology.org 81
- Page 1:
Studies in Mycology 58 (2007) The g
- Page 4 and 5:
Studies in Mycology The Studies in
- Page 7 and 8:
CONTENTS P.W. Crous, U. Braun and J
- Page 9 and 10:
lectotype for the genus by Clements
- Page 11:
Schubert K (2005a). Morphotaxonomic
- Page 14 and 15:
Crous et al. Table 1. Isolates for
- Page 16 and 17:
Crous et al. Table 1. (Continued).
- Page 18 and 19:
Crous et al. 100 10 changes 65 100
- Page 20 and 21:
Crous et al. Treatment of phylogene
- Page 22 and 23:
Crous et al. Teratosphaeria bellula
- Page 24 and 25:
Crous et al. 6. Conidiophores short
- Page 26 and 27:
Crous et al. Habit plant pathogenic
- Page 28 and 29:
Crous et al. Fig. 7. Catenulostroma
- Page 30 and 31:
Crous et al. system, consisting of
- Page 32 and 33:
Crous et al. Fig. 9. Penidiella col
- Page 34 and 35:
Crous et al. Fig. 12. Penidiella ri
- Page 36 and 37:
Crous et al. conidiophores, about 1
- Page 38 and 39:
Crous et al. have conidiomata rangi
- Page 40 and 41:
Crous et al. Schizothyriaceae clade
- Page 42 and 43: Crous et al. Fig. 21. Stigmidium sc
- Page 44 and 45: Crous et al. Gams W, Verkley GJM, C
- Page 46 and 47: Crous et al. Table 1. Isolates for
- Page 48 and 49: Crous et al. 100 61 100 52 100 10 c
- Page 50 and 51: Crous et al. Fig. 3. Rachicladospor
- Page 52 and 53: Crous et al. Fig. 4. Toxicocladospo
- Page 54 and 55: Crous et al. Fig. 5. Verrucocladosp
- Page 56 and 57: Crous et al. Fig. 7. Stenella aragu
- Page 58 and 59: Crous et al. Helotiales, incertae s
- Page 60 and 61: Crous et al. Fig. 10. Ochrocladospo
- Page 62 and 63: Crous et al. Fig. 12. Rhizocladospo
- Page 64 and 65: Crous et al. 7. Conidiophores unbra
- Page 66 and 67: Crous et al. 33. Terminal conidioge
- Page 68 and 69: Crous et al. Crous PW, Kang JC, Bra
- Page 70 and 71: Arzanlou et al. To date 26 species
- Page 72 and 73: Arzanlou et al. Table 1. (Continued
- Page 74 and 75: Arzanlou et al. 10 changes Athelia
- Page 76 and 77: Arzanlou et al. Athelia epiphylla A
- Page 78 and 79: Arzanlou et al. Fig. 3. Periconiell
- Page 80 and 81: Arzanlou et al. Cultural characteri
- Page 82 and 83: Arzanlou et al. Fig. 10. A. Ramichl
- Page 84 and 85: Arzanlou et al. Ramichloridium musa
- Page 86 and 87: Arzanlou et al. Cultural characteri
- Page 88 and 89: Arzanlou et al. Fig. 20. Rhinocladi
- Page 90 and 91: Arzanlou et al. Fig. 22. Rhinocladi
- Page 94 and 95: Arzanlou et al. Fig. 26. Veronaea c
- Page 96 and 97: Arzanlou et al. Cultural characteri
- Page 98 and 99: Arzanlou et al. Fig. 30. Myrmecridi
- Page 100 and 101: Arzanlou et al. Fig. 32. Radulidium
- Page 102 and 103: Arzanlou et al. Fig. 34. Rhodoveron
- Page 104 and 105: Arzanlou et al. even further, thoug
- Page 106 and 107: available online at www.studiesinmy
- Page 108 and 109: Dichocladosporium gen. nov. 10 chan
- Page 110 and 111: Dichocladosporium gen. nov. Fig. 3.
- Page 112 and 113: Dichocladosporium gen. nov. to intr
- Page 114 and 115: Dichocladosporium gen. nov. Czechos
- Page 116 and 117: available online at www.studiesinmy
- Page 118 and 119: Cladosporium herbarum species compl
- Page 120 and 121: Cladosporium herbarum species compl
- Page 122 and 123: Cladosporium herbarum species compl
- Page 124 and 125: Cladosporium herbarum species compl
- Page 126 and 127: Cladosporium herbarum species compl
- Page 128 and 129: Cladosporium herbarum species compl
- Page 130 and 131: Cladosporium herbarum species compl
- Page 132 and 133: Cladosporium herbarum species compl
- Page 134 and 135: Cladosporium herbarum species compl
- Page 136 and 137: Cladosporium herbarum species compl
- Page 138 and 139: Cladosporium herbarum species compl
- Page 140 and 141: Cladosporium herbarum species compl
- Page 142 and 143:
Cladosporium herbarum species compl
- Page 144 and 145:
Cladosporium herbarum species compl
- Page 146 and 147:
Cladosporium herbarum species compl
- Page 148 and 149:
Cladosporium herbarum species compl
- Page 150 and 151:
Cladosporium herbarum species compl
- Page 152 and 153:
Cladosporium herbarum species compl
- Page 154 and 155:
Cladosporium herbarum species compl
- Page 156 and 157:
Cladosporium herbarum species compl
- Page 158 and 159:
Cladosporium herbarum species compl
- Page 160 and 161:
Cladosporium herbarum species compl
- Page 162 and 163:
Cladosporium herbarum species compl
- Page 164 and 165:
Cladosporium herbarum species compl
- Page 166 and 167:
Cladosporium herbarum species compl
- Page 168 and 169:
available online at www.studiesinmy
- Page 170 and 171:
Cladosporium sphaerospermum species
- Page 172 and 173:
Cladosporium sphaerospermum species
- Page 174 and 175:
Cladosporium sphaerospermum species
- Page 176 and 177:
Cladosporium sphaerospermum species
- Page 178 and 179:
Cladosporium sphaerospermum species
- Page 180 and 181:
Cladosporium sphaerospermum species
- Page 182 and 183:
Cladosporium sphaerospermum species
- Page 184 and 185:
Cladosporium sphaerospermum species
- Page 186 and 187:
Cladosporium sphaerospermum species
- Page 188 and 189:
Cladosporium sphaerospermum species
- Page 190 and 191:
Cladosporium sphaerospermum species
- Page 192 and 193:
Cladosporium sphaerospermum species
- Page 194 and 195:
Cladosporium sphaerospermum species
- Page 196 and 197:
Crous et al. The aim of the present
- Page 198 and 199:
Crous et al. 10 changes Athelia epi
- Page 200 and 201:
Crous et al. Table 1. Isolates used
- Page 202 and 203:
Crous et al. Table 1. (Continued).
- Page 204 and 205:
Crous et al. 0.1 expected changes p
- Page 206 and 207:
Crous et al. Fig. 6. Cladophialopho
- Page 208 and 209:
Crous et al. Fig. 11. Cladophialoph
- Page 210 and 211:
Crous et al. Fig. 13. Cladophialoph
- Page 212 and 213:
Crous et al. Fig. 15. Exophiala sp.
- Page 214 and 215:
Crous et al. Fig. 18. Cylindrosympo
- Page 216 and 217:
Crous et al. Fig. 19. Fusicladium a
- Page 218 and 219:
Crous et al. Fig. 22. Fusicladium f
- Page 220 and 221:
Crous et al. Fig. 24. Fusicladium p
- Page 222 and 223:
Crous et al. Fusicladium rhodense C
- Page 224 and 225:
Crous et al. Excluded taxa Polyscyt
- Page 226 and 227:
Crous et al. the conidial tips are
- Page 228 and 229:
available online at www.studiesinmy
- Page 230 and 231:
Cladophialophora carrionii complex
- Page 232 and 233:
Cladophialophora carrionii complex
- Page 234 and 235:
Cladophialophora carrionii complex
- Page 236 and 237:
Cladophialophora carrionii complex
- Page 238 and 239:
Cladophialophora carrionii complex
- Page 240 and 241:
Cladophialophora carrionii complex
- Page 242 and 243:
Cladophialophora carrionii complex
- Page 244 and 245:
available online at www.studiesinmy
- Page 246 and 247:
Hormoconis resinae and morphologica
- Page 248 and 249:
Hormoconis resinae and morphologica
- Page 250 and 251:
Hormoconis resinae and morphologica
- Page 252 and 253:
Fig. 6 Hormoconis resinae and morph
- Page 254 and 255:
Hormoconis resinae and morphologica
- Page 256 and 257:
Cladophialophora, 52 k , 54 k -55,
- Page 258 and 259:
Fusicladium phillyreae, 189 c , 191
- Page 260 and 261:
Ramichloridium epichloës, 60, 89 P
- Page 262:
Trimmatostroma salicis, 3 t , 5, 6