Attention! Your ePaper is waiting for publication!
By publishing your document, the content will be optimally indexed by Google via AI and sorted into the right category for over 500 million ePaper readers on YUMPU.
This will ensure high visibility and many readers!

Your ePaper is now published and live on YUMPU!
You can find your publication here:
Share your interactive ePaper on all platforms and on your website with our embed function

Callister - An introduction - 8th edition
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
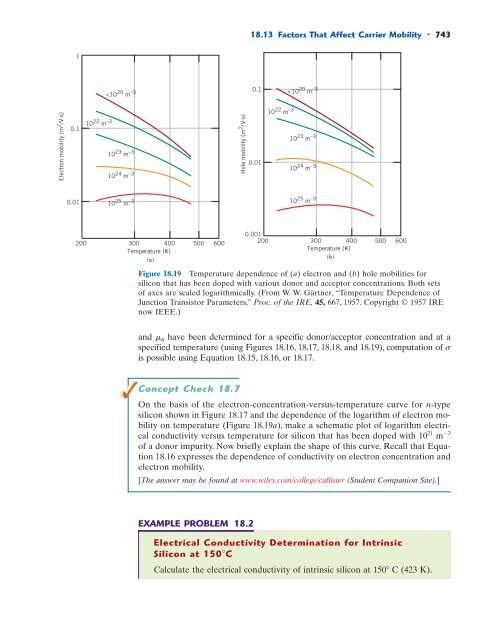
18.13 Factors That Affect Carrier Mobility • 743<br />
1<br />
Electron mobility (m 2 /V.s)<br />
10 22 m –3<br />
742 • Chapter 18 / Electrical Properties 18.13 FACTORS THAT AFFECT CARRIER MOBILITY The conductivity (or resistivity) of a semiconducting material, in addition to being dependent on electron and/or hole concentrations, is also a function of the charge carriers’ mobilities (Equation 18.13)—that is, the ease with which electrons and holes are transported through the crystal. Furthermore, magnitudes of electron and hole mobilities are influenced by the presence of those same crystalline defects that are responsible for the scattering of electrons in metals—thermal vibrations (i.e., temperature) and impurity atoms. We now explore the manner in which dopant impurity content and temperature influence the mobilities of both electrons and holes. Influence of Dopant Content Figure 18.18 represents the dependence of electron and hole mobilities in silicon as a function of the dopant (both acceptor and donor) content, at room temperature— note that both axes on this plot are scaled logarithmically. At dopant concentrations less than about 10 20 m 3 , both carrier mobilities are at their maximum levels and independent of the doping concentration. In addition, both mobilities decrease with increasing impurity content. Also worth noting is that the mobility of electrons is always larger than the mobility of holes. Influence of Temperature The temperature dependences of electron and hole mobilities for silicon are presented in Figures 18.19a and 18.19b, respectively. Curves for several impurity dopant contents are shown for both carrier types; furthermore, both sets of axes are scaled logarithmically. From these plots, note that, for dopant concentrations of 10 24 m 3 and below, both electron and hole mobilities decrease in magnitude with rising temperature; again, this effect is due to enhanced thermal scattering of the carriers. For both electrons and holes, and dopant levels less than 10 20 m 3 , the dependence of mobility on temperature is independent of acceptor/donor concentration (i.e., is represented by a single curve). Also, for concentrations greater than 10 20 m 3 , curves in both plots are shifted to progressively lower mobility values with increasing dopant level. These latter two effects are consistent with the data presented in Figure 18.18. These previous treatments have discussed the influence of temperature and dopant content on both carrier concentration and carrier mobility. Once values of n, p, e , Mobility (m 2 /V.s) 0.1 Electrons 19 10 20 10 21 0.001 10 10 22 10 23 10 24 10 25 Holes 0.01 Impurity concentration (m –3 ) Figure 18.18 For silicon, dependence of roomtemperature electron and hole mobilities (logarithmic scale) on dopant concentration (logarithmic scale). (Adapted from W. W. Gärtner, “Temperature Dependence of Junction Transistor Parameters,” Proc. of the IRE, 45, 667, 1957. Copyright © 1957 IRE now IEEE.)
18.13 Factors That Affect Carrier Mobility • 743 1 Electron mobility (m 2 /V.s) 10 22 m –3
- Page 2 and 3:
Characteristics of Selected Element
- Page 5 and 6:
With WileyPLUS: This online teachin
- Page 7 and 8:
E IGHTH E DITION Materials Science
- Page 9:
Dedicated to our wives, Nancy and E
- Page 12 and 13:
viii • Preface FEATURES THAT ARE
- Page 14 and 15:
x • Preface 2. Answers to Concept
- Page 16 and 17:
Appreciation is expressed to those
- Page 18 and 19:
xiv • Contents 3. The Structure o
- Page 20 and 21:
xvi • Contents 9.12 Development o
- Page 22 and 23:
xviii • Contents 15.14 Factors Th
- Page 24 and 25:
xx • Contents 21.14 Optical Fiber
- Page 26 and 27:
xxii • List of Symbols HK Knoop
- Page 29 and 30:
Chapter 1 Introduction A familiar i
- Page 31 and 32:
1.2 Materials Science and Engineeri
- Page 33 and 34:
1.4 Classification of Materials •
- Page 35 and 36:
1.4 Classification of Materials •
- Page 37 and 38:
1.4 Classification of Materials •
- Page 39 and 40:
1.5 Advanced Materials • 11 aeros
- Page 41 and 42:
1.6 Modern Materials’ Needs • 1
- Page 43 and 44:
STEELS Processing Diffusion ➣ Rec
- Page 45 and 46:
Question • 17 REFERENCES Ashby, M
- Page 47 and 48:
WHY STUDY Atomic Structure and Inte
- Page 49 and 50:
2.3 Electrons in Atoms • 21 Orbit
- Page 51 and 52:
2.3 Electrons in Atoms • 23 Table
- Page 53 and 54:
2.3 Electrons in Atoms • 25 Table
- Page 55 and 56:
2.4 The Periodic Table • 27 Metal
- Page 57 and 58:
2.5 Bonding Forces and Energies •
- Page 59 and 60:
2.6 Primary Interatomic Bonds • 3
- Page 61 and 62:
2.6 Primary Interatomic Bonds • 3
- Page 63 and 64:
2.7 Secondary Bonding or van der Wa
- Page 65 and 66:
2.7 Secondary Bonding or van der Wa
- Page 67 and 68:
Summary • 39 Primary Interatomic
- Page 69 and 70:
Questions and Problems • 41 QUEST
- Page 71 and 72:
Questions and Problems • 43 Sprea
- Page 73 and 74:
WHY STUDY The Structure of Crystall
- Page 75 and 76:
lattice 3.4 Metallic Crystal Struct
- Page 77 and 78:
3.4 Metallic Crystal Structures •
- Page 79 and 80:
3.5 Density Computations • 51 EXA
- Page 81 and 82:
3.7 Crystal Systems • 53 Another
- Page 83 and 84:
3.8 Point Coordinates • 55 Concep
- Page 85 and 86:
3.9 Crystallographic Directions •
- Page 87 and 88:
3.9 Crystallographic Directions •
- Page 89 and 90:
3.9 Crystallographic Directions •
- Page 91 and 92:
3.10 Crystallographic Planes • 63
- Page 93 and 94:
3.10 Crystallographic Planes • 65
- Page 95 and 96:
3.10 Crystallographic Planes • 67
- Page 97 and 98:
3.12 Close-Packed Crystal Structure
- Page 99 and 100:
3.12 Close-Packed Crystal Structure
- Page 101 and 102:
3.15 Anisotropy • 73 (a) (b) grai
- Page 103 and 104:
3.16 X-Ray Diffraction: Determinati
- Page 105 and 106:
0° 3.16 X-Ray Diffraction: Determi
- Page 107 and 108:
3.17 Noncrystalline Solids • 79 (
- Page 109 and 110:
Summary • 81 Point Coordinates Cr
- Page 111 and 112:
Processing/Structure/Properties/Per
- Page 113 and 114:
Questions and Problems • 85 118.7
- Page 115 and 116:
Questions and Problems • 87 3.40
- Page 117 and 118:
Questions and Problems • 89 Inten
- Page 119 and 120:
WHY STUDY Imperfections in Solids?
- Page 121 and 122:
self-interstitial increases exponen
- Page 123 and 124:
2. Crystal structure. For appreciab
- Page 125 and 126:
4.4 Specification of Composition
- Page 127 and 128:
4.5 Dislocations—Linear Defects
- Page 129 and 130:
4.5 Dislocations—Linear Defects
- Page 131 and 132:
4.6 Interfacial Defects • 103 Ang
- Page 133 and 134:
4.6 Interfacial Defects • 105 MAT
- Page 135 and 136:
the order of 10 13 vibrations per s
- Page 137 and 138:
4.10 Microscopic Techniques • 109
- Page 139 and 140:
4.10 Microscopic Techniques • 111
- Page 141 and 142:
4.11 GRAIN SIZE DETERMINATION grain
- Page 143 and 144:
Summary • 115 Specification of Co
- Page 145 and 146:
Summary • 117 List of Symbols Sym
- Page 147 and 148:
Questions and Problems • 119 (a)
- Page 149 and 150:
Design Problems • 121 (a) at a ma
- Page 151 and 152:
WHY Study Diffusion? Materials of a
- Page 153 and 154:
5.2 Diffusion Mechanisms • 125 se
- Page 155 and 156:
5.3 Steady-State Diffusion • 127
- Page 157 and 158:
5.4 Nonsteady-State Diffusion • 1
- Page 159 and 160:
5.4 Nonsteady-State Diffusion • 1
- Page 161 and 162:
5.5 Factors That Influence Diffusio
- Page 163 and 164:
5.5 Factors That Influence Diffusio
- Page 165 and 166:
5.6 Diffusion in Semiconducting Mat
- Page 167 and 168:
is only an approximation), then the
- Page 169 and 170:
5.6 Diffusion in Semiconducting Mat
- Page 171 and 172:
Summary • 143 Diffusion in Semico
- Page 173 and 174:
Questions and Problems • 145 QUES
- Page 175 and 176:
Diffusion coefficient (m 2 /s) 10 -
- Page 177 and 178:
Design Problems • 149 the process
- Page 179 and 180:
WHY STUDY The Mechanical Properties
- Page 181 and 182:
6.2 Concepts of Stress and Strain
- Page 183 and 184:
6.2 Concepts of Stress and Strain
- Page 185 and 186:
6.3 Stress-Strain Behavior • 157
- Page 187 and 188:
6.4 Anelasticity • 159 Figure 6.8
- Page 189 and 190:
6.5 Elastic Properties Of Materials
- Page 191 and 192:
6.6 Tensile Properties • 163 Figu
- Page 193 and 194:
6.6 Tensile Properties • 165 in F
- Page 195 and 196:
6.6 Tensile Properties • 167 Brit
- Page 197 and 198:
6.6 Tensile Properties • 169 y F
- Page 199 and 200:
6.7 True Stress and Strain • 171
- Page 201 and 202:
6.9 Compressive, Shear, and Torsion
- Page 203 and 204:
Table 6.5 Hardness-Testing Techniqu
- Page 205 and 206:
Brinell Hardness Tests 15 6.10 Hard
- Page 207 and 208:
6.10 Hardness • 179 are contained
- Page 209 and 210:
6.11 Variability Of Material Proper
- Page 211 and 212:
6.12 Design/Safety Factors • 183
- Page 213 and 214:
Summary • 185 • For an isotropi
- Page 215 and 216:
Summary • 187 6.15 s T F True st
- Page 217 and 218:
Questions and Problems • 189 a lo
- Page 219 and 220:
Questions and Problems • 191 tens
- Page 221 and 222:
Questions and Problems • 193 modu
- Page 223 and 224:
Design Problems • 195 (a) Determi
- Page 225 and 226:
Chapter 7 Dislocations and Strength
- Page 227 and 228:
Dislocations and Plastic Deformatio
- Page 229 and 230:
7.3 Characteristics of Dislocations
- Page 231 and 232:
7.4 Slip Systems • 203 D B A (a)
- Page 233 and 234:
7.5 Slip in Single Crystals • 205
- Page 235 and 236:
7.5 Slip in Single Crystals • 207
- Page 237 and 238:
7.6 Plastic Deformation of Polycrys
- Page 239 and 240:
7.7 Deformation by Twinning • 211
- Page 241 and 242:
7.9 Solid-Solution Strengthening
- Page 243 and 244:
7.10 Strain Hardening • 215 (a) (
- Page 245 and 246:
7.10 Strain Hardening • 217 600 2
- Page 247 and 248:
7.11 RECOVERY recovery 7.12 RECRYST
- Page 249 and 250:
7.12 Recrystallization • 221 (e)
- Page 251 and 252:
7.12 Recrystallization • 223 DESI
- Page 253 and 254:
Summary • 225 Figure 7.25 The log
- Page 255 and 256:
Summary • 227 Strain Hardening
- Page 257 and 258:
Questions and Problems • 229 Iron
- Page 259 and 260:
Questions and Problems • 231 and
- Page 261 and 262:
Design Problems • 233 Spreadsheet
- Page 263 and 264:
WHY STUDY Failure? The design of a
- Page 265 and 266:
8.3 Ductile Fracture • 237 Figure
- Page 267 and 268:
8.4 Brittle Fracture • 239 8.4 BR
- Page 269 and 270:
8.4 Brittle Fracture • 241 SEM Mi
- Page 271 and 272:
8.5 Principles of Fracture Mechanic
- Page 273 and 274:
8.5 Principles of Fracture Mechanic
- Page 275 and 276:
8.5 Principles of Fracture Mechanic
- Page 277 and 278:
8.5 Principles of Fracture Mechanic
- Page 279 and 280:
8.6 Fracture Toughness Testing •
- Page 281 and 282:
8.6 Fracture Toughness Testing •
- Page 283 and 284:
8.7 Cyclic Stresses • 255 Most ce
- Page 285 and 286:
8.8 The S-N Curve • 257 fatigue l
- Page 287 and 288:
8.9 Crack Initiation and Propagatio
- Page 289 and 290:
8.9 Crack Initiation and Propagatio
- Page 291 and 292:
8.10 Factors that Affect Fatigue Li
- Page 293 and 294:
8.12 Generalized Creep Behavior •
- Page 295 and 296:
8.13 Stress and Temperature Effects
- Page 297 and 298:
8.15 Alloys For High-Temperature Us
- Page 299 and 300:
Summary • 271 Ductile Fracture
- Page 301 and 302:
Summary • 273 • The presence of
- Page 303 and 304:
fatigue limit fatigue strength frac
- Page 305 and 306:
Questions and Problems • 277 Cycl
- Page 307 and 308:
Design Problems • 279 8.31 A cyli
- Page 309 and 310:
Chapter 9 Phase Diagrams The graph
- Page 311 and 312:
Definitions and Basic Concepts 9.2
- Page 313 and 314:
9.5 PHASE EQUILIBRIA equilibrium fr
- Page 315 and 316:
phases is along curve aO—likewise
- Page 317 and 318:
9.8 Interpretation of Phase Diagram
- Page 319 and 320:
9.8 Interpretation of Phase Diagram
- Page 321 and 322:
9.8 Interpretation of Phase Diagram
- Page 323 and 324:
9.9 Development of Microstructure i
- Page 325 and 326:
9.10 Mechanical Properties of Isomo
- Page 327 and 328:
9.11 Binary Eutectic Systems • 29
- Page 329 and 330:
9.11 Binary Eutectic Systems • 30
- Page 331 and 332:
9.11 Binary Eutectic Systems • 30
- Page 333 and 334:
9.12 Development of Microstructure
- Page 335 and 336:
9.12 Development of Microstructure
- Page 337 and 338:
9.12 Development of Microstructure
- Page 339 and 340:
9.13 Equilibrium Diagrams Having In
- Page 341 and 342:
9.14 Eutectoid and Peritectic React
- Page 343 and 344:
9.15 Congruent Phase Transformation
- Page 345 and 346:
9.17 The Gibbs Phase Rule • 317 F
- Page 347 and 348:
The Iron-Carbon System 9.18 The Iro
- Page 349 and 350:
9.18 The Iron-Iron Carbide (Fe-Fe 3
- Page 351 and 352:
9.19 Development of Microstructure
- Page 353 and 354:
9.19 Development of Microstructure
- Page 355 and 356:
9.19 Development of Microstructure
- Page 357 and 358:
9.19 Development of Microstructure
- Page 359 and 360:
Summary • 331 SUMMARY Introductio
- Page 361 and 362:
Summary • 333 Development of Micr
- Page 363 and 364:
Questions and Problems • 335 Impo
- Page 365 and 366:
Questions and Problems • 337 (b)
- Page 367 and 368:
Questions and Problems • 339 Figu
- Page 369 and 370:
Questions and Problems • 341 9.54
- Page 371 and 372:
WHY STUDY Phase Transformations? Th
- Page 373 and 374:
10.3 The Kinetics of Phase Transfor
- Page 375 and 376:
10.3 The Kinetics of Phase Transfor
- Page 377 and 378:
10.3 The Kinetics of Phase Transfor
- Page 379 and 380:
10.3 The Kinetics of Phase Transfor
- Page 381 and 382:
10.3 The Kinetics of Phase Transfor
- Page 383 and 384:
10.4 Metastable versus Equilibrium
- Page 385 and 386:
10.5 Isothermal Transformation Diag
- Page 387 and 388:
10.5 Isothermal Transformation Diag
- Page 389 and 390:
10.5 Isothermal Transformation Diag
- Page 391 and 392:
10.5 Isothermal Transformation Diag
- Page 393 and 394:
10.5 Isothermal Transformation Diag
- Page 395 and 396:
10.6 Continuous Cooling Transformat
- Page 397 and 398:
10.6 Continuous Cooling Transformat
- Page 399 and 400:
10.7 Mechanical Behavior of Iron-Ca
- Page 401 and 402:
10.7 Mechanical Behavior of Iron-Ca
- Page 403 and 404:
10.8 TEMPERED MARTENSITE In the as-
- Page 405 and 406:
10.8 Tempered Martensite • 377 (b
- Page 407 and 408:
10.9 Review of Phase Transformation
- Page 409 and 410:
Summary • 381 Figure 10.37. Of co
- Page 411 and 412:
Summary • 383 Spheroidite—is co
- Page 413 and 414:
Questions and Problems • 385 Stee
- Page 415 and 416:
Questions and Problems • 387 10.1
- Page 417 and 418:
Questions and Problems • 389 (c)
- Page 419 and 420:
Chapter 11 Applications and Process
- Page 421 and 422:
11.2 Ferrous Alloys • 393 Types o
- Page 423 and 424:
11.2 Ferrous Alloys • 395 Table 1
- Page 425 and 426:
11.2 Ferrous Alloys • 397 Table 1
- Page 427 and 428:
11.2 Ferrous Alloys • 399 is exte
- Page 429 and 430:
11.2 Ferrous Alloys • 401 (c) 20
- Page 431 and 432:
Table 11.5 Designations, Minimum Me
- Page 433 and 434:
11.2 Ferrous Alloys • 405 is pres
- Page 435 and 436:
11.3 Nonferrous Alloys • 407 Tabl
- Page 437 and 438:
11.3 Nonferrous Alloys • 409 Tabl
- Page 439 and 440:
11.3 Nonferrous Alloys • 411 Tabl
- Page 441 and 442:
Table 11.9 Compositions, Mechanical
- Page 443 and 444:
11.3 Nonferrous Alloys • 415 or n
- Page 445 and 446:
11.4 Forming Operations • 417 Met
- Page 447 and 448:
11.5 Casting • 419 that have rath
- Page 449 and 450:
11.6 Miscellaneous Techniques • 4
- Page 451 and 452:
11.7 Annealing Processes • 423 ox
- Page 453 and 454:
modification of mechanical properti
- Page 455 and 456:
11.8 Heat Treatment of Steels • 4
- Page 457 and 458:
11.8 Heat Treatment of Steels • 4
- Page 459 and 460:
11.8 Heat Treatment of Steels • 4
- Page 461 and 462:
11.8 Heat Treatment of Steels • 4
- Page 463 and 464:
11.8 Heat Treatment of Steels • 4
- Page 465 and 466:
11.9 Precipitation Hardening • 43
- Page 467 and 468:
11.9 Precipitation Hardening • 43
- Page 469 and 470:
11.9 Precipitation Hardening • 44
- Page 471 and 472:
Summary • 443 Forming Operations
- Page 473 and 474:
Summary • 445 PROCESSING Recrysta
- Page 475 and 476:
Questions and Problems • 447 REFE
- Page 477 and 478:
Design Problems • 449 and temperi
- Page 479 and 480:
Chapter 12 Structures and Propertie
- Page 481 and 482:
12.2 Crystal Structures • 453 Con
- Page 483 and 484:
12.2 Crystal Structures • 455 Tab
- Page 485 and 486:
12.2 Crystal Structures • 457 Fur
- Page 487 and 488:
12.2 Crystal Structures • 459 VMS
- Page 489 and 490:
12.2 Crystal Structures • 461 Tet
- Page 491 and 492:
12.2 Crystal Structures • 463 EXA
- Page 493 and 494:
12.3 Silicate Ceramics • 465 Si 4
- Page 495 and 496:
12.3 Silicate Ceramics • 467 Figu
- Page 497 and 498:
12.4 Carbon • 469 Figure 12.16 Sc
- Page 499 and 500:
Another molecular form of carbon ha
- Page 501 and 502:
12.5 Imperfections in Ceramics •
- Page 503 and 504:
12.5 Imperfections in Ceramics •
- Page 505 and 506:
12.7 Ceramic Phase Diagrams • 477
- Page 507 and 508:
12.7 Ceramic Phase Diagrams • 479
- Page 509 and 510:
12.8 Brittle Fracture of Ceramics
- Page 511 and 512:
12.8 Brittle Fracture of Ceramics
- Page 513 and 514:
flexural strength Flexural strength
- Page 515 and 516:
12.10 Mechanisms of Plastic Deforma
- Page 517 and 518:
viscosities at ambient temperatures
- Page 519 and 520:
Summary • 491 Table 12.6 Vickers
- Page 521 and 522:
Summary • 493 • Diagrams for Al
- Page 523 and 524:
Questions and Problems • 495 visc
- Page 525 and 526:
Questions and Problems • 497 does
- Page 527 and 528:
Questions and Problems • 499 Stre
- Page 529 and 530:
Chapter 13 Applications and Process
- Page 531 and 532:
13.3 Glass-Ceramics • 503 Ceramic
- Page 533 and 534:
13.5 Refractories • 505 Glass-cer
- Page 535 and 536:
13.6 Abrasives • 507 (2910F). Thu
- Page 537 and 538:
13.8 Advanced Ceramics • 509 Seve
- Page 539 and 540:
13.8 Advanced Ceramics • 511 opti
- Page 541 and 542:
13.9 Fabrication and Processing of
- Page 543 and 544:
13.9 Fabrication and Processing of
- Page 545 and 546:
13.9 Fabrication and Processing of
- Page 547 and 548:
13.10 Fabrication and Processing of
- Page 549 and 550:
13.10 Fabrication and Processing of
- Page 551 and 552:
13.11 Powder Pressing • 523 a sca
- Page 553 and 554:
13.12 Tape Casting • 525 Figure 1
- Page 555 and 556:
Summary • 527 • Requirements fo
- Page 557 and 558:
Summary • 529 This chapter also d
- Page 559 and 560:
13.12 Compare the softening points
- Page 561 and 562:
WHY STUDY Polymer Structures? A rel
- Page 563 and 564:
14.3 Polymer Molecules • 535 Tabl
- Page 565 and 566:
14.4 The Chemistry of Polymer Molec
- Page 567 and 568:
14.4 The Chemistry of Polymer Molec
- Page 569 and 570:
Figure 14.3 Hypothetical polymer mo
- Page 571 and 572:
14.5 Molecular Weight • 543 Table
- Page 573 and 574:
14.7 Molecular Structure • 545 Fi
- Page 575 and 576:
14.8 Molecular Configurations • 5
- Page 577 and 578:
14.8 Molecular Configurations • 5
- Page 579 and 580:
14.10 Copolymers • 551 adjacent c
- Page 581 and 582:
14.11 Polymer Crystallinity • 553
- Page 583 and 584:
For copolymers, as a general rule,
- Page 585 and 586:
14.12 Polymer Crystals • 557 ~ 10
- Page 587 and 588:
14.14 Diffusion in Polymeric Materi
- Page 589 and 590:
Summary • 561 PET is permeable to
- Page 591 and 592:
Equation Summary Summary • 563 Po
- Page 593 and 594:
Important Terms and Concepts Questi
- Page 595 and 596:
Questions and Problems • 567 age
- Page 597 and 598:
Chapter 15 Characteristics, Applica
- Page 599 and 600:
15.2 Stress-Strain Behavior • 571
- Page 601 and 602:
15.3 Macroscopic Deformation • 57
- Page 603 and 604:
15.4 Viscoelastic Deformation • 5
- Page 605 and 606:
15.4 Viscoelastic Deformation • 5
- Page 607 and 608:
15.5 Fracture of Polymers • 579 F
- Page 609 and 610:
15.7 Deformation of Semicrystalline
- Page 611 and 612:
t 0 t t 0 t 0 Stage 1 Stage 2 (a) (
- Page 613 and 614:
15.8 Factors That Influence the Mec
- Page 615 and 616:
15.8 Factors That Influence the Mec
- Page 617 and 618:
15.9 Deformation of Elastomers •
- Page 619 and 620:
15.10 Crystallization • 591 Norma
- Page 621 and 622:
15.13 Melting and Glass Transition
- Page 623 and 624:
15.14 Factors That Influence Meltin
- Page 625 and 626:
15.15 Plastics • 597 Table 15.3 (
- Page 627 and 628:
15.16 Elastomers • 599 ing phenol
- Page 629 and 630:
15.18 Miscellaneous Applications
- Page 631 and 632:
15.19 Advanced Polymeric Materials
- Page 633 and 634:
15.19 Advanced Polymeric Materials
- Page 635 and 636:
The tensile modulus of this TPE mat
- Page 637 and 638:
15.20 Polymerization • 609 Additi
- Page 639 and 640:
15.22 Forming Techniques for Plasti
- Page 641 and 642:
15.22 Forming Techniques for Plasti
- Page 643 and 644:
15.24 Fabrication of Fibers and Fil
- Page 645 and 646:
Factors That Influence the Mechanic
- Page 647 and 648:
Summary • 619 Equation Summary Eq
- Page 649 and 650:
Questions and Problems • 621 McCr
- Page 651 and 652:
Questions and Problems • 623 Tens
- Page 653 and 654:
Design Questions • 625 Elastomers
- Page 655 and 656:
3.1 Hardness • 627 WHY STUDY Comp
- Page 657 and 658:
16.1 Introduction • 629 Dispersed
- Page 659 and 660:
16.2 Large-Particle Composites •
- Page 661 and 662:
16.2 Large-Particle Composites •
- Page 663 and 664:
Critical fiber length—dependence
- Page 665 and 666:
16.5 Influence of Fiber Orientation
- Page 667 and 668:
16.5 Influence of Fiber Orientation
- Page 669 and 670:
ut, because s/E, 16.5 Influence o
- Page 671 and 672:
16.5 Influence of Fiber Orientation
- Page 673 and 674:
16.6 The Fiber Phase • 645 Applic
- Page 675 and 676:
eactions with the environment. Such
- Page 677 and 678:
16.8 Polymer-Matrix Composites •
- Page 679 and 680:
16.8 Polymer-Matrix Composites •
- Page 681 and 682:
16.9 Metal-Matrix Composites • 65
- Page 683 and 684:
The high-temperature creep and rupt
- Page 685 and 686:
16.12 HYBRID COMPOSITES hybrid comp
- Page 687 and 688:
16.13 Processing of Fiber-Reinforce
- Page 689 and 690:
16.15 Sandwich Panels • 661 Figur
- Page 691 and 692:
Summary • 663 they remain separat
- Page 693 and 694:
Summary • 665 When l l c , Equat
- Page 695 and 696:
References • 667 List of Symbols
- Page 697 and 698:
Questions and Problems • 669 16.1
- Page 699 and 700:
Design Problems • 671 to be align
- Page 701 and 702:
Chapter 17 Corrosion and Degradatio
- Page 703 and 704:
Corrosion of Metals 17.2 Electroche
- Page 705 and 706:
17.2 Electrochemical Considerations
- Page 707 and 708:
17.2 Electrochemical Considerations
- Page 709 and 710:
17.2 Electrochemical Considerations
- Page 711 and 712:
17.4 Prediction of Corrosion Rates
- Page 713 and 714:
17.4 Prediction of Corrosion Rates
- Page 715 and 716:
17.4 Prediction of Corrosion Rates
- Page 717 and 718:
17.4 Prediction of Corrosion Rates
- Page 719 and 720: 17.5 Passivity • 691 to further c
- Page 721 and 722: 17.7 Forms of Corrosion • 693 sur
- Page 723 and 724: 17.7 Forms of Corrosion • 695 Fig
- Page 725 and 726: 17.7 Forms of Corrosion • 697 Fig
- Page 727 and 728: 17.7 Forms of Corrosion • 699 Fig
- Page 729 and 730: sodium chloride. Dilute sulfuric ac
- Page 731 and 732: 17.10 Oxidation • 703 Zinc coatin
- Page 733 and 734: 17.10 Oxidation • 705 Table 17.3
- Page 735 and 736: Ceramic materials are frequently us
- Page 737 and 738: 17.12 Bond Rupture • 709 17.12 BO
- Page 739 and 740: Summary • 711 marily a result of
- Page 741 and 742: Summary • 713 Corrosion Preventio
- Page 743 and 744: Questions and Problems • 715 REFE
- Page 745 and 746: Questions and Problems • 717 17.1
- Page 747 and 748: Chapter 18 Electrical Properties (b
- Page 749 and 750: 18.3 Electrical Conductivity • 72
- Page 751 and 752: 18.5 Energy Band Structures in Soli
- Page 753 and 754: valence band conduction band energy
- Page 755 and 756: covalent) and relatively weak, whic
- Page 757 and 758: 18.8 Electrical Resistivity of Meta
- Page 759 and 760: 18.9 Electrical Characteristics of
- Page 761 and 762: 18.10 Intrinsic Semiconduction •
- Page 763 and 764: 18.10 Intrinsic Semiconduction •
- Page 765 and 766: 18.11 Extrinsic Semiconduction •
- Page 767 and 768: 18.11 Extrinsic Semiconduction •
- Page 769: 18.12 The Temperature Dependence of
- Page 773 and 774: DESIGN EXAMPLE 18.1 Acceptor Impuri
- Page 775 and 776: 18.14 The Hall Effect • 747 Depen
- Page 777 and 778: 18.15 Semiconductor Devices • 749
- Page 779 and 780: 18.15 Semiconductor Devices • 751
- Page 781 and 782: 18.15 Semiconductor Devices • 753
- Page 783 and 784: 18.16 Conduction in Ionic Materials
- Page 785 and 786: ands that overlap the valence and c
- Page 787 and 788: 18.19 Field Vectors and Polarizatio
- Page 789 and 790: 18.19 Field Vectors and Polarizatio
- Page 791 and 792: ionic polarization Electric dipole
- Page 793 and 794: 18.23 Dielectric Materials • 765
- Page 795 and 796: Summary • 767 + + + + + + + + +
- Page 797 and 798: Summary • 769 • With these mate
- Page 799 and 800: Summary • 771 18.11 r i Ac i 11
- Page 801 and 802: Summary • 773 One common use for
- Page 803 and 804: Questions and Problems • 775 Elec
- Page 805 and 806: Questions and Problems • 777 The
- Page 807 and 808: Design Problems • 779 DESIGN PROB
- Page 809 and 810: Chapter 19 Thermal Properties (a) (
- Page 811 and 812: 19.2 Heat Capacity • 783 Figure 1
- Page 813 and 814: 19.3 Thermal Expansion • 785 Tabl
- Page 815 and 816: 19.3 Thermal Expansion • 787 For
- Page 817 and 818: 19.4 THERMAL CONDUCTIVITY Thermal c
- Page 819 and 820: 19.4 Thermal Conductivity • 791 F
- Page 821 and 822:
19.5 Thermal Stresses • 793 speci
- Page 823 and 824:
Summary • 795 • Coefficient-of-
- Page 825 and 826:
Questions and Problems • 797 19.8
- Page 827 and 828:
Design Problems • 799 rod be fabr
- Page 829 and 830:
WHY STUDY the Magnetic Properties o
- Page 831 and 832:
20.2 Basic Concepts • 803 I B 0 =
- Page 833 and 834:
20.3 Diamagnetism and Paramagnetism
- Page 835 and 836:
20.4 Ferromagnetism • 807 Table 2
- Page 837 and 838:
20.5 Antiferromagnetism and Ferrima
- Page 839 and 840:
20.5 Antiferromagnetism and Ferrima
- Page 841 and 842:
20.6 The Influence of Temperature o
- Page 843 and 844:
20.7 Domains and Hysteresis • 815
- Page 845 and 846:
20.7 Domains and Hysteresis • 817
- Page 847 and 848:
20.9 Soft Magnetic Materials • 81
- Page 849 and 850:
20.9 Soft Magnetic Materials • 82
- Page 851 and 852:
20.10 Hard Magnetic Materials • 8
- Page 853 and 854:
20.11 Magnetic Storage • 825 requ
- Page 855 and 856:
20.11 Magnetic Storage • 827 Figu
- Page 857 and 858:
20.12 Superconductivity • 829 Ele
- Page 859 and 860:
20.12 Superconductivity • 831 Tab
- Page 861 and 862:
Summary • 833 • For cubic ferri
- Page 863 and 864:
Questions and Problems • 835 List
- Page 865 and 866:
Questions and Problems • 837 with
- Page 867 and 868:
Design Problems • 839 H C 1T 2 H
- Page 869 and 870:
WHY STUDY the Optical Properties of
- Page 871 and 872:
21.3 Light Interactions with Solids
- Page 873 and 874:
21.4 Atomic and Electronic Interact
- Page 875 and 876:
21.5 Refraction • 847 Velocity of
- Page 877 and 878:
21.7 ABSORPTION Nonmetallic materia
- Page 879 and 880:
21.7 Absorption • 851 Reaction de
- Page 881 and 882:
21.9 Color • 853 21.9 COLOR color
- Page 883 and 884:
21.11 Luminescence • 855 Figure 2
- Page 885 and 886:
21.11 Luminescence • 857 n- and p
- Page 887 and 888:
21.13 Lasers • 859 Ruby Flash lam
- Page 889 and 890:
21.13 Lasers • 861 Partially refl
- Page 891 and 892:
21.4 Optical Fibers in Communicatio
- Page 893 and 894:
Summary • 865 Input impulse Outpu
- Page 895 and 896:
Summary • 867 Transparent nonmeta
- Page 897 and 898:
Questions and Problems • 869 Impo
- Page 899 and 900:
Design Problem • 871 fiber glass
- Page 901 and 902:
WHY STUDY Economic, Environmental,
- Page 903 and 904:
Environmental and Societal Consider
- Page 905 and 906:
Environmental and Societal Consider
- Page 907 and 908:
22.5 Recycling Issues in Materials
- Page 909 and 910:
22.5 Recycling Issues in Materials
- Page 911 and 912:
22.5 Recycling Issues in Materials
- Page 913:
Design Questions • 885 Environmen
- Page 916 and 917:
A2 • Appendix A / The Internation
- Page 918 and 919:
A4 • Appendix B / Properties of S
- Page 920 and 921:
A6 • Appendix B / Properties of S
- Page 922 and 923:
A8 • Appendix B / Properties of S
- Page 924 and 925:
A10 • Appendix B / Properties of
- Page 926 and 927:
A12 • Appendix B / Properties of
- Page 928 and 929:
A14 • Appendix B / Properties of
- Page 930 and 931:
A16 • Appendix B / Properties of
- Page 932 and 933:
A18 • Appendix B / Properties of
- Page 934 and 935:
A20 • Appendix B / Properties of
- Page 936 and 937:
A22 • Appendix B / Properties of
- Page 938 and 939:
A24 • Appendix B / Properties of
- Page 940 and 941:
A26 • Appendix B / Properties of
- Page 942 and 943:
A28 • Appendix B / Properties of
- Page 944 and 945:
A30 • Appendix B / Properties of
- Page 946 and 947:
A32 • Appendix C / Costs and Rela
- Page 948 and 949:
A34 • Appendix C / Costs and Rela
- Page 950 and 951:
Appendix D Repeat Unit Structures f
- Page 952 and 953:
A38 • Appendix D / Repeat Unit St
- Page 954 and 955:
Appendix E Glass Transition and Mel
- Page 956 and 957:
G2 • Glossary atomic mass units (
- Page 958 and 959:
G4 • Glossary according to unit c
- Page 960 and 961:
G6 • Glossary Fick’s first law.
- Page 962 and 963:
G8 • Glossary J Jominy end-quench
- Page 964 and 965:
G10 • Glossary alternating layers
- Page 966 and 967:
G12 • Glossary stock; also, elong
- Page 968 and 969:
G14 • Glossary tempered martensit
- Page 970 and 971:
Answers to Selected Problems Chapte
- Page 972 and 973:
S2 • Answers to Selected Problems
- Page 974:
S4 • Answers to Selected Problems
- Page 977 and 978:
plane strain fracture toughness, 24
- Page 979 and 980:
Clay, characteristics, 518-519 Clay
- Page 981 and 982:
Die casting, 419 Dielectric breakdo
- Page 983 and 984:
Fatigue life, 258, G4 factors that
- Page 985 and 986:
Heat treatable, definition of, 406
- Page 987 and 988:
modulus of elasticity, 486 thermal
- Page 989 and 990:
Nanotubes, carbon, 13, 471 Natural
- Page 991 and 992:
fatigue behavior (PET), 580 magneti
- Page 993 and 994:
single crystal, 451 structure of, 4
- Page 995 and 996:
Stabilized zirconia, 478, 655 Stabi
- Page 997 and 998:
Transparency, 844, G13 Transverse b
- Page 999 and 1000:
Power 1 W 0.239 cal/s 1 cal/s 4.1
Inappropriate
Loading...
Inappropriate
You have already flagged this document.
Thank you, for helping us keep this platform clean.
The editors will have a look at it as soon as possible.
Mail this publication
Loading...
Embed
Loading...
Delete template?
Are you sure you want to delete your template?
DOWNLOAD ePAPER
This ePaper is currently not available for download.
You can find similar magazines on this topic below under ‘Recommendations’.