Properties of hemp fibre polymer composites -An optimisation of ...
Properties of hemp fibre polymer composites -An optimisation of ...
Properties of hemp fibre polymer composites -An optimisation of ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
kill the fungus. The <strong>fibre</strong>s were thereafter washed in water and aligned into a planar<br />
sheet followed by vacuum drying. The yield <strong>of</strong> <strong>fibre</strong>s from the original <strong>hemp</strong> stems was<br />
determined by 2-3 repetitions at the small, medium and large scale.<br />
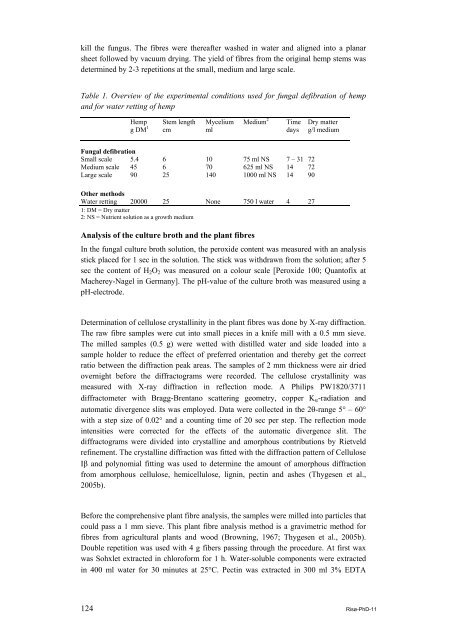
Table 1. Overview <strong>of</strong> the experimental conditions used for fungal defibration <strong>of</strong> <strong>hemp</strong><br />
and for water retting <strong>of</strong> <strong>hemp</strong><br />
Hemp<br />
g DM 1<br />
Stem length<br />
cm<br />
Mycelium<br />
ml<br />
Medium 2<br />
Time<br />
days<br />
Fungal defibration<br />
Small scale 5.4 6 10 75 ml NS 7 – 31 72<br />
Medium scale 45 6 70 625 ml NS 14 72<br />
Large scale 90 25 140 1000 ml NS 14 90<br />
Other methods<br />
Water retting 20000 25 None 750 l water 4 27<br />
1: DM = Dry matter<br />
2: NS = Nutrient solution as a growth medium<br />
<strong>An</strong>alysis <strong>of</strong> the culture broth and the plant <strong>fibre</strong>s<br />
Dry matter<br />
g/l medium<br />
In the fungal culture broth solution, the peroxide content was measured with an analysis<br />
stick placed for 1 sec in the solution. The stick was withdrawn from the solution; after 5<br />
sec the content <strong>of</strong> H2O2 was measured on a colour scale [Peroxide 100; Quant<strong>of</strong>ix at<br />
Macherey-Nagel in Germany]. The pH-value <strong>of</strong> the culture broth was measured using a<br />
pH-electrode.<br />
Determination <strong>of</strong> cellulose crystallinity in the plant <strong>fibre</strong>s was done by X-ray diffraction.<br />
The raw <strong>fibre</strong> samples were cut into small pieces in a knife mill with a 0.5 mm sieve.<br />
The milled samples (0.5 g) were wetted with distilled water and side loaded into a<br />
sample holder to reduce the effect <strong>of</strong> preferred orientation and thereby get the correct<br />
ratio between the diffraction peak areas. The samples <strong>of</strong> 2 mm thickness were air dried<br />
overnight before the diffractograms were recorded. The cellulose crystallinity was<br />
measured with X-ray diffraction in reflection mode. A Philips PW1820/3711<br />
diffractometer with Bragg-Brentano scattering geometry, copper Kα-radiation and<br />
automatic divergence slits was employed. Data were collected in the 2θ-range 5° – 60°<br />
with a step size <strong>of</strong> 0.02° and a counting time <strong>of</strong> 20 sec per step. The reflection mode<br />
intensities were corrected for the effects <strong>of</strong> the automatic divergence slit. The<br />
diffractograms were divided into crystalline and amorphous contributions by Rietveld<br />
refinement. The crystalline diffraction was fitted with the diffraction pattern <strong>of</strong> Cellulose<br />
Iβ and polynomial fitting was used to determine the amount <strong>of</strong> amorphous diffraction<br />
from amorphous cellulose, hemicellulose, lignin, pectin and ashes (Thygesen et al.,<br />
2005b).<br />
Before the comprehensive plant <strong>fibre</strong> analysis, the samples were milled into particles that<br />
could pass a 1 mm sieve. This plant <strong>fibre</strong> analysis method is a gravimetric method for<br />
<strong>fibre</strong>s from agricultural plants and wood (Browning, 1967; Thygesen et al., 2005b).<br />
Double repetition was used with 4 g fibers passing through the procedure. At first wax<br />
was Sohxlet extracted in chlor<strong>of</strong>orm for 1 h. Water-soluble components were extracted<br />
in 400 ml water for 30 minutes at 25°C. Pectin was extracted in 300 ml 3% EDTA<br />
124 Risø-PhD-11