Attention! Your ePaper is waiting for publication!
By publishing your document, the content will be optimally indexed by Google via AI and sorted into the right category for over 500 million ePaper readers on YUMPU.
This will ensure high visibility and many readers!

Your ePaper is now published and live on YUMPU!
You can find your publication here:
Share your interactive ePaper on all platforms and on your website with our embed function

Food Lipids: Chemistry, Nutrition, and Biotechnology
Food Lipids: Chemistry, Nutrition, and Biotechnology
Food Lipids: Chemistry, Nutrition, and Biotechnology
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
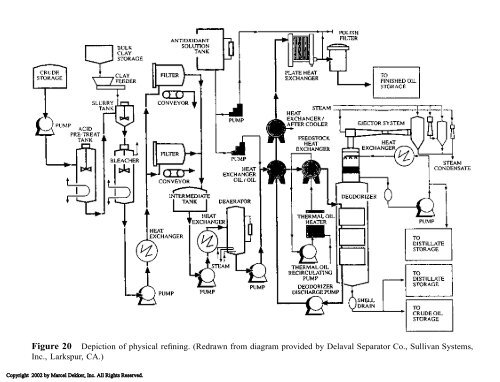
Figure 20 Depiction of physical refining. (Redrawn from diagram provided by Delaval Separator Co., Sullivan Systems,<br />
Inc., Larkspur, CA.)<br />
Copyright 2002 by Marcel Dekker, Inc. All Rights Reserved.
Usually steam alone is not used to heat the oil because very high steam pressures would be required to heat the oil sufficiently. Rather, a eutectic mixture of diphenyl <strong>and</strong> diphenyl oxide, known by the trade name Dowtherm A, is used. This product has a boiling point of 258�C <strong>and</strong> at 304�C generates a pressure of only 110 kPa (16 psi). Deodorization may be conducted in either batch, semibatch, or continuous vessels. The type of process largely depends on the volume <strong>and</strong> number of different products being processed. Batch deodorizers have cycle times of 6–8 hours. Continuous deodorizers are most suitable when a limited number of products are manufactured in very large volume. There are five stages in deodorization: deaeration, heating, deodorization/steam stripping, heat recovery/cooling, <strong>and</strong> final cooling. Stripping steam is provided through sparging rings <strong>and</strong> airlift pumps. After the deodorized oil has cooled, a small amount (0.005–0.01%) of citric acid is added to chelate metal cations so they would not promote oxidation <strong>and</strong> reduce shelf life. Crude oils usually have greater oxidative stability than refined oils. Indeed, many processors do not expose deodorized fats <strong>and</strong> oils to the atmosphere, but discharge oil into tanks blanketed with nitrogen <strong>and</strong> fill bottled oil under nitrogen. Deodorization also removes any residual pesticide <strong>and</strong> hexane. Some pigments, such as �-carotene, are destroyed by the high heat in deodorization <strong>and</strong>, thus, the yellow <strong>and</strong> sometimes red colors are reduced. Deodorized oils have improved flavor, odor, <strong>and</strong> color. The high temperatures used in deodorization cause limited geometric isomerization. Although deodorization removes most peroxides, it cannot reclaim rancid oxidized oils. Deodorizer distillate is condensed, recovered, <strong>and</strong> sold at higher prices per pound than the oil itself. Soybean deodorizer distillate typically contains 12.3% tocopherols <strong>and</strong> 21.9% sterols [49]. Deodorizer distillate may be further processed into valuable fractions rich in tocopherols (vitamin E), which are in high dem<strong>and</strong> by the food <strong>and</strong> pharmaceutical industries. The sterols may also be purified <strong>and</strong> sold into the pharmaceutical industry for manufacturing various synthetic hormones. I. Physical Refining Physical refining (Fig. 20) is also known as steam refining. These terms are applied to the removal of the free fatty acids from the oil rather than reacting them with alkali, as well as to the removal of the compounds normally targeted by deodorization. Physical refining combines both neutralization <strong>and</strong> deodorization into one operation. Physical refining is always preceded by degumming <strong>and</strong> bleaching steps. The major advantage of physical refining is that the yield of oil is improved because there is none of the neutral oil loss that accompanies the production of soapstock. This process also affords the possibility of recovering fatty acids for the oleochemical industry without the need for acidulating soapstock <strong>and</strong> attendant wastewater production. The equipment used for physical refining is similar to deodorization, but with additional steam sparging trays. Not all oils are suitable for physical refining. The oil must be low in phosphorus content (most comes from phosphatides). The exact upper limit for phosphorus in oil suitable for physical refining is a long-st<strong>and</strong>ing controversy, but it may be less than 5 ppm. Higher levels result in dark colored oils. Physical refining is also more appropriate for those higher in free fatty acid contents because the benefit of higher Copyright 2002 by Marcel Dekker, Inc. All Rights Reserved.
Figure 20 Depiction of physical refining. (Redrawn from diagram provided by Delaval Separator Co., Sullivan Systems, Inc., Larkspur, CA.) Copyright 2002 by Marcel Dekker, Inc. All Rights Reserved.
- Page 1 and 2:
TM Food Lipids Chemistry, Nutrition
- Page 3 and 4:
FOOD SCIENCE AND TECHNOLOGY A Serie
- Page 5 and 6:
37. Omega-3 Fatty Acids in Health a
- Page 7 and 8:
90. Dairy Technology: Principles of
- Page 9 and 10:
Preface to the Second Edition Reade
- Page 11 and 12:
Preface to the First Edition There
- Page 13 and 14:
Contents Preface to the Second Edit
- Page 15 and 16:
20. Dietary Fats and Coronary Heart
- Page 17 and 18:
J. Bruce German Department of Food
- Page 19 and 20:
1 Nomenclature and Classification o
- Page 21 and 22:
described as 2-methyl-3-phytyl-1,4-
- Page 23 and 24:
Table 3 A Summary of Sequence Prior
- Page 25 and 26:
Table 5 Systematic, Common, and Sho
- Page 27 and 28:
saturation of EFA occurs (primarily
- Page 29 and 30:
Figure 5 Prostaglandin metabolites
- Page 31 and 32:
Figure 7 Prostanoic acid and prosta
- Page 33 and 34:
Figure 9 Eicosenoid isomers in part
- Page 35 and 36:
Figure 10 Nomenclature of cyclic fa
- Page 37 and 38:
Figure 13 Hydroxy fatty acid struct
- Page 39 and 40:
Figure 15 Furanoid fatty acid struc
- Page 41 and 42:
Table 7 Short Abbreviations for Som
- Page 43 and 44:
Figure 20 Steroid nomenclature. rep
- Page 45 and 46:
Figure 22 Cholesterol oxidation pro
- Page 47 and 48:
E. Phosphoglycerides (Phospholipids
- Page 49 and 50:
Figure 26 Glyceroglycolipid structu
- Page 51 and 52:
Figure 28 Structures of some vitami
- Page 53 and 54:
Figure 29 Structures of some vitami
- Page 55 and 56:
Figure 32 Structures of some vitami
- Page 57 and 58:
6. IUPAC. Nomenclature of Organic C
- Page 59 and 60:
2 Chemistry and Function of Phospho
- Page 61 and 62:
variant [1]. Phosphonolipids are ma
- Page 63 and 64:
nonelectrolytes, however, the perme
- Page 65 and 66:
pensations in that enthalpy lost by
- Page 67 and 68:
Table 3 Membrane Deterioration in A
- Page 69 and 70:
erols to generate semisolid or plas
- Page 71 and 72:
The interaction of phospholipids wi
- Page 73 and 74:
fatty acid occurs when Fe 3� at t
- Page 75 and 76:
4. M. C. Blok, L. L. M. van Deenen,
- Page 77 and 78:
47. N. Markova, E. Sparr, L. Wadso,
- Page 79 and 80:
87. R. J. Hsieh. Contribution of li
- Page 81 and 82:
3 Lipid-Based Emulsions and Emulsif
- Page 83 and 84:
may be composed of surface-active c
- Page 85 and 86:
2. Cloud Point When a surfactant so
- Page 87 and 88:
HLB=7� � (hydrophilic group num
- Page 89 and 90:
sion is known as the phase inversio
- Page 91 and 92:
strongly with each other rather tha
- Page 93 and 94:
� = � (1 � 2.5�) (3) 0 wher
- Page 95 and 96:
Figure 8 Biopolymer molecules or ag
- Page 97 and 98:
divide homogenization into two cate
- Page 99 and 100:
2� �P 1 = (4) r where � is th
- Page 101 and 102:
flow rate, decreasing the size of t
- Page 103 and 104:
sion be particularly small, it is u
- Page 105 and 106:
2. Electrostatic Interactions Elect
- Page 107 and 108:
profile of interdroplet pair potent
- Page 109 and 110:
Figure 12 Mechanisms of emulsion in
- Page 111 and 112:
[Eq. (9)], but at high droplet conc
- Page 113 and 114:
alteration in the system’s compos
- Page 115 and 116:
Food emulsions always contain dropl
- Page 117 and 118:
creaming and sedimentation in emuls
- Page 119 and 120:
Foams (E. Dickinson and G. Stainsby
- Page 121 and 122:
4 The Chemistry of Waxes and Sterol
- Page 123 and 124:
Shrinkage and flash point are two f
- Page 125 and 126:
lowing discussion on chemical analy
- Page 127 and 128:
violet chromophore. Application of
- Page 129 and 130:
Figure 1 Examples of naturally occu
- Page 131 and 132:
Scheme 1 Synthesis of mevalonic aci
- Page 133 and 134:
Scheme 3 Synthesis of farnesyl pyro
- Page 135 and 136:
Scheme 6 Biosynthesis of cholestero
- Page 137 and 138:
educe the risk of coronary heart di
- Page 139 and 140:
of cholesterol to bile acids (Schem
- Page 141 and 142:
Scheme 10 Metabolic alterations of
- Page 143 and 144:
sample preparation is usually emplo
- Page 145 and 146:
at levels of 1-100 �g per compone
- Page 147 and 148:
30. H. W. Chen, A. A. Kandutsch, an
- Page 149 and 150:
Biologically Significant Steroids (
- Page 151 and 152:
5 Extraction and Analysis of Lipids
- Page 153 and 154:
preparing nutritional labeling mate
- Page 155 and 156:
polar solvents, such as alkanols, f
- Page 157 and 158:
a ternary system consisting of chlo
- Page 159 and 160:
lipids from meat or hydrolytic prod
- Page 161 and 162:
perature under vacuum. Acid hydroly
- Page 163 and 164:
IV. ANALYSIS OF LIPID EXTRACTS Lipi
- Page 165 and 166:
ments, etc.) primarily involves chr
- Page 167 and 168:
2. Gas Chromatography The GC (or GL
- Page 169 and 170:
4. Supercritical Fluid Chromatograp
- Page 171 and 172:
Table 3 Solvent Systems that Could
- Page 173 and 174:
sibility of determining all compone
- Page 175 and 176:
the isolated trans band is another
- Page 177 and 178:
ionized molecule has the highest m/
- Page 179 and 180:
3. W. R. Bloor. Outline of a classi
- Page 181 and 182:
45. Association of Official Analyti
- Page 183 and 184:
90. M. N. Vaghela and A. Kilara. A
- Page 185 and 186:
132. C. G. Walton, W. M. N. Ratnaya
- Page 187 and 188:
6 Methods for trans Fatty Acid Anal
- Page 189 and 190:
where A = abc (1) 1 A = absorbance
- Page 191 and 192:
methyl elaidate weight equivalents
- Page 193 and 194:
method was modified by inclusion of
- Page 195 and 196:
may be packed or bound to a column,
- Page 197 and 198:
Figure 4 The C-18 region of the gas
- Page 199 and 200:
Table 2 Response Factors of Unsatur
- Page 201 and 202:
Figure 5 Separation of the phenacyl
- Page 203 and 204:
B. Gas Chromatography/IR Spectrosco
- Page 205 and 206:
Figure 7 Expanded IR spectral range
- Page 207 and 208:
CFAMs before converting them to the
- Page 209 and 210:
Figure 8 GC-EIMS chromatographic da
- Page 211 and 212:
flame ionization detection. The fat
- Page 213 and 214:
levels in excess of 50% of the tota
- Page 215 and 216:
22. A. Huang and D. Firestone. Dete
- Page 217 and 218:
59. E. G. Perkins and C. Smick. Oct
- Page 219 and 220:
97. Association of Official Analyti
- Page 221 and 222:
136. J. J. Myer and A. Kukis. Elect
- Page 223 and 224:
7 Chemistry of Frying Oils KATHLEEN
- Page 225 and 226: Table 1 Effects of Physical and Che
- Page 227 and 228: Figure 3 Oxidation reactions in fry
- Page 229 and 230: or intermittent frying, oil filtrat
- Page 231 and 232: for the ultimate criteria to evalua
- Page 233 and 234: triacylglycerol polymers, and triac
- Page 235 and 236: 100 compounds identified in hydroge
- Page 237 and 238: 5. J. Pokorny. Flavor chemistry of
- Page 239 and 240: 50. Anonymous. Recommendations of t
- Page 241 and 242: 8 Recovery, Refining, Converting, a
- Page 243 and 244: Figure 1 Depiction of hard screw pr
- Page 245 and 246: Figure 2 Depiction of prepress solv
- Page 247 and 248: Seed containing more than the criti
- Page 249 and 250: Figure 5 Steps in processing soybea
- Page 251 and 252: specification of 50% protein is met
- Page 253 and 254: tent of prepress cake is 15-18%, an
- Page 255 and 256: in successive passes through the be
- Page 257 and 258: Figure 10 Additional commonly used
- Page 259 and 260: that follow the DT. Drying at norma
- Page 261 and 262: B. Extraction of Oil-Bearing Fruits
- Page 263 and 264: 1. Wet Rendering Wet rendering is t
- Page 265 and 266: Figure 15 Process flow sheet for de
- Page 267 and 268: Table 2 Properties of Some Crude an
- Page 269 and 270: Figure 16 Process flow sheet for al
- Page 271 and 272: Figure 17 Process flow sheet for va
- Page 273 and 274: for problematic high wax contents (
- Page 275: Figure 19 Oil processing facilities
- Page 279 and 280: 4-7�C, and then to tanks with slo
- Page 281 and 282: Figure 21 Hydrogenation reaction me
- Page 283 and 284: ond, which may form in its original
- Page 285 and 286: Figure 24 Plasticization of margari
- Page 287 and 288: Figure 25 Equipment used in expande
- Page 289 and 290: selectivity, others claim success i
- Page 291 and 292: 48. E. J. Campbell. Sunflower oil.
- Page 293 and 294: 9 Crystallization and Polymorphism
- Page 295 and 296: Generally, it is accepted that both
- Page 297 and 298: Figure 2 A point lattice. (Adapted
- Page 299 and 300: structure.... The term ‘fat’ us
- Page 301 and 302: Figure 6 Schematic representation o
- Page 303 and 304: Figure 8 Polymorphic transitions of
- Page 305 and 306: where A = B = C and all are saturat
- Page 307 and 308: LMF was found to facilitate the tra
- Page 309 and 310: Table 3 Nomenclature and Melting Po
- Page 311 and 312: Other mixed-fat systems have been s
- Page 313 and 314: Figure 12 Proposed intermediate str
- Page 315 and 316: 17. K. Larsson. The crystal structu
- Page 317 and 318: 59. G. G. Jewell. Vegetable fats. I
- Page 319 and 320: 10 Chemical Interesterification of
- Page 321 and 322: however, since monoacylglycerols an
- Page 323 and 324: D. The ‘‘Real’’ Catalyst Th
- Page 325 and 326: Figure 3 Proposed reaction mechanis
- Page 327 and 328:
Figure 5 Kinetics of interesterific
- Page 329 and 330:
Table 1 Theoretical Triacylglycerol
- Page 331 and 332:
Cast et al. [55] demonstrated the r
- Page 333 and 334:
Solid: Liquid: SSS, 33.3% OOO, 8.3%
- Page 335 and 336:
Figure 11 Changes in the fatty acid
- Page 337 and 338:
fication and blending on butterfat-
- Page 339 and 340:
influence of interesterification on
- Page 341 and 342:
B. Margarines In the manufacture of
- Page 343 and 344:
Figure 16 Proportion of soild fat o
- Page 345 and 346:
Tautorus and McCurdy [102] demonstr
- Page 347 and 348:
ACKNOWLEDGMENTS The authors acknowl
- Page 349 and 350:
54. A. Kuksis, M. J. McCarthy, and
- Page 351 and 352:
101. S. Zalewski and A. M. Gaddis.
- Page 353 and 354:
11 Lipid Oxidation of Edible Oil DA
- Page 355 and 356:
Figure 1 Molecular orbital of tripl
- Page 357 and 358:
Figure 3 Singlet oxygen formation b
- Page 359 and 360:
tween substrate and triplet oxygen
- Page 361 and 362:
Figure 8 Conjugated and nonconjugat
- Page 363 and 364:
Figure 11 Conjugated hydroperoxide
- Page 365 and 366:
etween the oxygen and the oxygen of
- Page 367 and 368:
cadienal, trans,trans-2,4-decadiena
- Page 369 and 370:
Figure 14 Mechanism for the formati
- Page 371 and 372:
Copyright 2002 by Marcel Dekker, In
- Page 373 and 374:
Figure 18 Formation and reactions o
- Page 375 and 376:
Figure 19 Effect of 0, 0.25, 0.5, a
- Page 377 and 378:
Figure 21 Singlet oxygen quenching
- Page 379 and 380:
10. E. N. Frankel. Chemistry of aut
- Page 381 and 382:
52. A. L. Callison. Singlet oxygen
- Page 383 and 384:
12 Lipid Oxidation of Muscle Foods
- Page 385 and 386:
A. Initiation The direct reaction o
- Page 387 and 388:
undles), and endomysia (sheaths of
- Page 389 and 390:
sosomes, etc. A comparison of the l
- Page 391 and 392:
dative stability, comparisons betwe
- Page 393 and 394:
5. Hydrolysis of Lipids and Associa
- Page 395 and 396:
of iron from the heme pocket by coo
- Page 397 and 398:
Membrane systems that reduce iron c
- Page 399 and 400:
phases of storage [51,187,188]. In
- Page 401 and 402:
4. Glutathione While the traditiona
- Page 403 and 404:
G. Mathematical Modeling The pathwa
- Page 405 and 406:
with this chemical, treated salmon
- Page 407 and 408:
multicomponent alternatives [304,30
- Page 409 and 410:
and its extent was related to inten
- Page 411 and 412:
esponded similarly to vacuum packag
- Page 413 and 414:
31. M. L. Greaser, R. G. Cassens, W
- Page 415 and 416:
72. J. S. Elmore, D. S. Mottram, M.
- Page 417 and 418:
112. J. Kanner, H. Mendel, and P. B
- Page 419 and 420:
154. M. B. Korycka-Dahl and T. Rich
- Page 421 and 422:
193. P. Akhtar, J. I. Gray, T. H. C
- Page 423 and 424:
230. B. Bjerkeng and G. Johnsen. Fr
- Page 425 and 426:
271. C.-J. Huang, and M.-L. Fwu. De
- Page 427 and 428:
313. M. G. Mast and J. H. MacNeil.
- Page 429 and 430:
353. H. A. Ghanbari, W. B. Wheeler,
- Page 431 and 432:
13 Fatty Acid Oxidation in Plant Ti
- Page 433 and 434:
1. Fatty Acid Activation Prior to d
- Page 435 and 436:
L.) leaf peroxisomes exhibits highe
- Page 437 and 438:
Figure 2 The glyoxylate cycle in gl
- Page 439 and 440:
C. �-Oxidation of Specific Fatty
- Page 441 and 442:
cotyledons and partially purified.
- Page 443 and 444:
Catabolism of heptanoyl CoA as well
- Page 445 and 446:
Figure 6 Peroxisome catabolism of m
- Page 447 and 448:
onstrated that �-oxidations requi
- Page 449 and 450:
that the member of the enzyme famil
- Page 451 and 452:
Plant oxylipin pathway, also named
- Page 453 and 454:
Figure 9 Proposed scheme for lipoxy
- Page 455 and 456:
oleic acid. These results suggest t
- Page 457 and 458:
of the seed pod reversed senescnece
- Page 459 and 460:
Figure 11 ‘‘Heterolytic’’-t
- Page 461 and 462:
Since 1971, when this physiologic r
- Page 463 and 464:
Table 1 Physiological Effects of Ja
- Page 465 and 466:
erides, although PUFAs in both form
- Page 467 and 468:
9-hydroperoxides and did not attack
- Page 469 and 470:
aerial parts of plants, constitutes
- Page 471 and 472:
32. J. B. Ohlrogge and V. S. Eccles
- Page 473 and 474:
74. L. J. Morris. The mechanism of
- Page 475 and 476:
115. B. A. Vick. Oxygenated fatty a
- Page 477 and 478:
153. T. K. Peterman and J. N. Siedo
- Page 479 and 480:
193. B. A. Stelmach, A. Müller, P.
- Page 481 and 482:
230. M. Hamberg, C. A. Herman, and
- Page 483 and 484:
14 Methods for Measuring Oxidative
- Page 485 and 486:
ated fatty acids during oxidation (
- Page 487 and 488:
Several other chemical methods have
- Page 489 and 490:
Figure 3 Relationship between perox
- Page 491 and 492:
distillate. In case of the distilla
- Page 493 and 494:
and ketones. This ion is formed fro
- Page 495 and 496:
(Fig. 9), foaming, color, viscosity
- Page 497 and 498:
Yen and Duh [69] and Chen and Ho [7
- Page 499 and 500:
Figure 10 1 H Nuclear magnetic reso
- Page 501 and 502:
to Marquez-Ruiz et al. [93], who us
- Page 503 and 504:
35. F. Shahidi, J. Yun, L.J. Rubin,
- Page 505 and 506:
77. H. Saito and K. Nakamura. Appli
- Page 507 and 508:
15 Antioxidants DAVID W. REISCHE Th
- Page 509 and 510:
Hydroperoxide degradation leads to
- Page 511 and 512:
e cyclical, with regeneration of th
- Page 513 and 514:
A. Synthetic Antioxidants Synthetic
- Page 515 and 516:
Propyl gallate (PG) 212.20 White cr
- Page 517 and 518:
4. 6-Ethoxy-1,2-dihydro-2,2,4-trime
- Page 519 and 520:
Figure 3 Structures of tocopherols
- Page 521 and 522:
Figure 4 Structures of ascorbic aci
- Page 523 and 524:
4. Enzymatic Antioxidants Glucose o
- Page 525 and 526:
would be imprudent to discount any
- Page 527 and 528:
droxycoumarin (scopoletin), and hyd
- Page 529 and 530:
Figure 7 Structures of sesame antio
- Page 531 and 532:
8. G. Minotti. Sources and role of
- Page 533 and 534:
50. K. Shimada, H. Muta, Y. Nakamur
- Page 535 and 536:
16 Antioxidant Mechanisms ERIC A. D
- Page 537 and 538:
For instance, the hydrogen of the h
- Page 539 and 540:
Figure 2 Mechanism by which one phe
- Page 541 and 542:
Figure 4 Formation of �-tocophero
- Page 543 and 544:
Figure 6 Formation of an epoxyquino
- Page 545 and 546:
gallate. The antioxidant mechanism
- Page 547 and 548:
Figure 8 Products formed from the o
- Page 549 and 550:
in food systems, transition metals
- Page 551 and 552:
An intersystem energy transfer occu
- Page 553 and 554:
V. ALTERATIONS IN LIPID OXIDATION B
- Page 555 and 556:
VII. ANTIOXIDANT INTERACTIONS Biolo
- Page 557 and 558:
26. J. Kanner, J. B. German, and J.
- Page 559 and 560:
68. J. Kanner, F. Sofer, S. Harel,
- Page 561 and 562:
17 Fats and Oils in Human Health DA
- Page 563 and 564:
Table 1 Classification of LDL Parti
- Page 565 and 566:
stearic) had been incorporated by i
- Page 567 and 568:
studies contains equal amounts (40-
- Page 569 and 570:
Table 5 Influence of 25% Caloric Re
- Page 571 and 572:
26. L. D. Cowan, D. L. O’Connell,
- Page 573 and 574:
fatty acid margarine on serum lipid
- Page 575 and 576:
nutrition examination survey. I. Ep
- Page 577 and 578:
18 Unsaturated Fatty Acids STEVEN M
- Page 579 and 580:
Figure 1 A generalized scheme for h
- Page 581 and 582:
Plant fatty acids provide a seminal
- Page 583 and 584:
plants [27]. However, the requireme
- Page 585 and 586:
The reciprocal response of the �6
- Page 587 and 588:
fatty acids and a dynamic system fo
- Page 589 and 590:
production of commercially viable o
- Page 591 and 592:
acids synthesized de novo, primaril
- Page 593 and 594:
Figure 4 The cis and trans configur
- Page 595 and 596:
VI. SYNTHESIS AND ABUNDANCE OF PUFA
- Page 597 and 598:
of eicosanoids. It is present in al
- Page 599 and 600:
usual NMIFA structures with potenti
- Page 601 and 602:
10. S. P. Baykousheva, D. L. Luthri
- Page 603 and 604:
47. M. J. T. Alaniz, I. N. T. d. Go
- Page 605 and 606:
86. R. J. Henderson and D. R. Toche
- Page 607 and 608:
19 Dietary Fats, Eicosanoids, and t
- Page 609 and 610:
synthesize EPA from linolenic acid
- Page 611 and 612:
Figure 2 Immune responses as a func
- Page 613 and 614:
done predominantly in rodent specie
- Page 615 and 616:
guinea pigs showed increased immune
- Page 617 and 618:
of energy from fat. Feeding the low
- Page 619 and 620:
14. P. Purasiri, A. Murray, S. Rich
- Page 621 and 622:
20 Dietary Fats and Coronary Heart
- Page 623 and 624:
(HDLs). Each class has its own char
- Page 625 and 626:
70% of the total amount of choleste
- Page 627 and 628:
McGandy and coworkers [8] have care
- Page 629 and 630:
Figure 4 Effects of myristic and pa
- Page 631 and 632:
3. Polyunsaturated Fatty Acids Poly
- Page 633 and 634:
Figure 8 Effects of a mixture of sa
- Page 635 and 636:
chemoattractant protein-1 (MCP-1),
- Page 637 and 638:
Figure 11 In vitro LDL oxidation. F
- Page 639 and 640:
Table 4 Fatty Acid Composition of a
- Page 641 and 642:
Figure 12 Processes involved in thr
- Page 643 and 644:
as compared with a diet rich in but
- Page 645 and 646:
from cardiovascular disease [73,74]
- Page 647 and 648:
Figure 16 Schematic representation
- Page 649 and 650:
aggregation tendency induced by som
- Page 651 and 652:
35. D. R. Janero. Malondialdehyde a
- Page 653 and 654:
68. B. J. Burrl, R. M. Dougherty, D
- Page 655 and 656:
21 Conjugated Linoleic Acids: Nutri
- Page 657 and 658:
method that works optimally in all
- Page 659 and 660:
adipose tissue contained two major
- Page 661 and 662:
providing rats with 0.5% and 1% CLA
- Page 663 and 664:
with the control group. Interesting
- Page 665 and 666:
feeding. These findings suggest tha
- Page 667 and 668:
In contrast to the antioxidative pr
- Page 669 and 670:
esponsible, at least in part, for t
- Page 671 and 672:
Yamasaki et al. studied CLA and ant
- Page 673 and 674:
17. J. K. G. Kramer, P. W. Parodi,
- Page 675 and 676:
55. C. Ip, S. F. Chin, J. A. Scimec
- Page 677 and 678:
90. J. S. Munday, K. G. Thompson, a
- Page 679 and 680:
126. J. Singh, R. Hamid, and B. S.
- Page 681 and 682:
22 Dietary Fats and Obesity DOROTHY
- Page 683 and 684:
For example, some have reported tha
- Page 685 and 686:
the fat component and the other hal
- Page 687 and 688:
shows that there is a negative corr
- Page 689 and 690:
C. Influence of Dietary Fat on Fatt
- Page 691 and 692:
Figure 1 Signal transduction cascad
- Page 693 and 694:
mals indicate that high-fat feeding
- Page 695 and 696:
processes. Numerous studies provide
- Page 697 and 698:
monier (252) suggested that in cert
- Page 699 and 700:
4. National Task Force on Obesity.
- Page 701 and 702:
46. F. Lucas, K. Ackroff, and A. Sc
- Page 703 and 704:
90. D. Mela. Sensory preference for
- Page 705 and 706:
133. D. R. Romsos and G. A. Leveill
- Page 707 and 708:
169. T. Ide, H. Kobayashi, L. Ashak
- Page 709 and 710:
207. A. B. Awad and E. A. Zepp. Alt
- Page 711 and 712:
246. Y. B. Kim, R. Nakajima, T. Mat
- Page 713 and 714:
23 Lipid-Based Synthetic Fat Substi
- Page 715 and 716:
operations, and in theory, can repl
- Page 717 and 718:
Table 3 Types of Lipid-Based Fat Su
- Page 719 and 720:
Figure 1 Structure of sucrose polye
- Page 721 and 722:
Figure 3 Synthetic scheme for olest
- Page 723 and 724:
Figure 5 Structure of sorbitol poly
- Page 725 and 726:
Figure 8 Structure of raffinose pol
- Page 727 and 728:
Figure 11 Structure of methyl galac
- Page 729 and 730:
Table 4 Some Properties of Sucrose
- Page 731 and 732:
Figure 15 Structure of trialkoxytri
- Page 733 and 734:
Figure 19 Structure of polysiloxane
- Page 735 and 736:
inversely related to the degree of
- Page 737 and 738:
Table 8 Some Nutritional Uses of No
- Page 739 and 740:
genetic assay in Chinese hamster ov
- Page 741 and 742:
IX. PERSPECTIVES With the approval
- Page 743 and 744:
30. L. Osipow, F. D. Snell, D. Marr
- Page 745 and 746:
72. K. W. Miller and P. H. Long. A
- Page 747 and 748:
24 Food Applications of Lipids FRAN
- Page 749 and 750:
Table 3 Typical Fatty Acid Composit
- Page 751 and 752:
are significant differences in oil
- Page 753 and 754:
Table 6 Production and Disappearanc
- Page 755 and 756:
and margarine should be pronounced
- Page 757 and 758:
Table 8 Approximate Fatty Acid Comp
- Page 759 and 760:
egions of the world. In addition to
- Page 761 and 762:
50-55% fat. Production figures for
- Page 763 and 764:
utter with up to 5% of another fat
- Page 765 and 766:
market with high overrun and good s
- Page 767 and 768:
11. D. Hettinga. Butter. In: Bailey
- Page 769 and 770:
25 Lipid Biotechnology KUMAR D. MUK
- Page 771 and 772:
Table 1 Lipid Content and Levels of
- Page 773 and 774:
as 80% lipids of which about 90% ar
- Page 775 and 776:
Table 6 Wax Esters Formed by Acinet
- Page 777 and 778:
Some other biosurfactants include e
- Page 779 and 780:
Figure 6 Microbial production of hy
- Page 781 and 782:
Figure 9 Microbial production of ke
- Page 783 and 784:
Figure 12 Microbial production of k
- Page 785 and 786:
Table 8 Specificity of Triacylglyce
- Page 787 and 788:
Figure 16 Lipase-catalyzed transest
- Page 789 and 790:
Figure 20 Preparation of structured
- Page 791 and 792:
Figure 24 Preparation of monoacylgl
- Page 793 and 794:
Fatty acid esters of polyols are us
- Page 795 and 796:
Figure 29 Specificity constants in
- Page 797 and 798:
Figure 31 Preparation of concentrat
- Page 799 and 800:
Figure 35 Enrichment of very long c
- Page 801 and 802:
B. Phospholipases Figure 38 shows t
- Page 803 and 804:
Lysophosphatidic acid has been prep
- Page 805 and 806:
Figure 41 Transesterification of ph
- Page 807 and 808:
Figure 46 Enzymatic production of h
- Page 809 and 810:
Figure 50 Hydration of linoleic aci
- Page 811 and 812:
Figure 51 Principle of enzymatic de
- Page 813 and 814:
13. E. Molina Grima, J. A. Sánchez
- Page 815 and 816:
53. M. Powalla, S. Lang, and V. Wra
- Page 817 and 818:
96. R. Schuch and K. D. Mukherjee.
- Page 819 and 820:
138. K. D. Mukherjee and I. Kiewitt
- Page 821 and 822:
179. U. T. Bornscheuer, H. Stamatis
- Page 823 and 824:
222. H. Stamatis, V. Sereti, and F.
- Page 825 and 826:
265. S. R. Moore and G. P. McNeill.
- Page 827 and 828:
303. C. Virto, I. Svensson, and P.
- Page 829 and 830:
343. E. Blee and F. Schuber. Regio-
- Page 831 and 832:
26 Microbial Lipases JOHN D. WEETE
- Page 833 and 834:
ation. Fungal lipases typically exi
- Page 835 and 836:
of buffer A containing 1 M ammonium
- Page 837 and 838:
without shaking for 30 minutes, whe
- Page 839 and 840:
the C domain of the protein through
- Page 841 and 842:
Figure 2 (Continued) Figure 3 Schem
- Page 843 and 844:
on the substrate and presence or ab
- Page 845 and 846:
Table 3 Selectivities of Multiple E
- Page 847 and 848:
lanuginosa, C. antarctica B, Rhizop
- Page 849 and 850:
10. C. T. Hou and T. M. Johnston. S
- Page 851 and 852:
53. C. C. Akoh. Enzymatic synthesis
- Page 853 and 854:
94. D. M. Lawson, A. M. Brzozowski,
- Page 855 and 856:
135. B. K. Yang and J. P. Chen. Gel
- Page 857 and 858:
27 Enzymatic Interesterification WE
- Page 859 and 860:
esterification of butterfat at 40
- Page 861 and 862:
also found to decrease the crystall
- Page 863 and 864:
T c than animal fats. The T c for v
- Page 865 and 866:
in the oxyanion hole is the amino a
- Page 867 and 868:
of the interface as a measure of su
- Page 869 and 870:
Figure 11 Catalytic mechanism for l
- Page 871 and 872:
Figure 13 Triacylglycerol products
- Page 873 and 874:
imization of interesterification, a
- Page 875 and 876:
supports include high losses of act
- Page 877 and 878:
is a thin layer located directly ne
- Page 879 and 880:
volume per year. The volumetric act
- Page 881 and 882:
and removal of reactants and produc
- Page 883 and 884:
vinyl chloride. In a membrane such
- Page 885 and 886:
of the enzyme. Animal and plant lip
- Page 887 and 888:
esters as surface active agents dur
- Page 889 and 890:
14. P. Kalo, H. Huotari, and M. Ant
- Page 891 and 892:
57. F. Pabai, S. Kermasha, and A. M
- Page 893 and 894:
94. J. Kurashige. Enzymatic convers
- Page 895 and 896:
28 Structured Lipids CASIMIR C. AKO
- Page 897 and 898:
Figure 2 Structure of a physical mi
- Page 899 and 900:
2. Medium Chain Fatty Acids and Tri
- Page 901 and 902:
Figure 4 Pathway for eicosanoid bio
- Page 903 and 904:
leukotrienes (hydroxy fatty acids a
- Page 905 and 906:
Figure 7 Structure of Benefat (bran
- Page 907 and 908:
cause of the huge capital investmen
- Page 909 and 910:
Figure 8 Reaction scheme showing ac
- Page 911 and 912:
alters the native conformation of t
- Page 913 and 914:
Table 6 Advantages of Enzymatic App
- Page 915 and 916:
Figure 13 Stereochemical configurat
- Page 917 and 918:
IV. NUTRITIONAL AND MEDICAL APPLICA
- Page 919 and 920:
Table 9 Factors That Affect Outlook
- Page 921 and 922:
27. G. O. Burr and M. D. Burr. A ne
- Page 923 and 924:
69. M. Reslow, P. Aldercreutz, and
- Page 925 and 926:
113. C. J. Gollaher, E. S. Swenson,
- Page 927 and 928:
29 Biosynthesis of Fatty Acids and
- Page 929 and 930:
olive (Olea europea), and avocado (
- Page 931 and 932:
Table 2 (Continued) Fatty acid a Sp
- Page 933 and 934:
2. Basic Features The functional un
- Page 935 and 936:
Hydroxy-Acyl ACP dehydrase (Crotony
- Page 937 and 938:
Figure 1 Steps of fatty acid biosyn
- Page 939 and 940:
The three KAS isoforms are assigned
- Page 941 and 942:
chain length acyl-ACP residues, and
- Page 943 and 944:
10-carbon and 14- to 16-carbon acyl
- Page 945 and 946:
In developing castor seed, BC and A
- Page 947 and 948:
(viz., seed, fruit) genes, and this
- Page 949 and 950:
Figure 4 Fatty acid modification re
- Page 951 and 952:
12-MO, the cDNA for the enzyme has
- Page 953 and 954:
Table 5 Enzyme Activities Involved
- Page 955 and 956:
of reactivity is also supported by
- Page 957 and 958:
tems, PTAP is a likely candidate fo
- Page 959 and 960:
ifying 18:1 �9 and deacylating 18
- Page 961 and 962:
oils (rich in 18:3 �6,9,12), and
- Page 963 and 964:
eticulum subpopulations also finds
- Page 965 and 966:
CPT (DAG ↔ PC) Extensive involvem
- Page 967 and 968:
available (cf. Sec. V.C.4 and Ref.
- Page 969 and 970:
erol backbone of triacylglycerols c
- Page 971 and 972:
14. E. Heinz. Biosynthesis of polyu
- Page 973 and 974:
56. R. C. Clough, A. L. Matthis, S.
- Page 975 and 976:
91. R. J. Heath and C. O. Rock. Eno
- Page 977 and 978:
124. R. Schuch, F. M. Brück, M. Br
- Page 979 and 980:
Kader and P. Mazliak, eds.). Kluwer
- Page 981 and 982:
197. Y. Cao, K. Oo, and A. H. C. Hu
- Page 983 and 984:
241. M. C. Dobarganes, G. Márquez-
- Page 985 and 986:
30 Genetic Engineering of Crops Tha
- Page 987 and 988:
existing genes in the host plant ge
- Page 989 and 990:
It becomes the plant breeder’s jo
- Page 991 and 992:
previously mentioned low-linolenic
- Page 993 and 994:
develop and apply methods, evaluate
- Page 995 and 996:
enzyme with high activity for placi
- Page 997 and 998:
activity in the oil palm mesocarp s
- Page 999 and 1000:
Figure 4 Commercial applications of
- Page 1001 and 1002:
and mildness. Because the perennial
- Page 1003 and 1004:
In the context of developing increa
- Page 1005 and 1006:
occurs esterified at the sn-2 posit
- Page 1007 and 1008:
as high as coconut or palm kernel o
- Page 1009 and 1010:
systems. Key to assessing the oppor
- Page 1011 and 1012:
property encouraged us to look for
- Page 1013 and 1014:
mercial lauric fats based on both P
Inappropriate
Loading...
Inappropriate
You have already flagged this document.
Thank you, for helping us keep this platform clean.
The editors will have a look at it as soon as possible.
Mail this publication
Loading...
Embed
Loading...
Delete template?
Are you sure you want to delete your template?
DOWNLOAD ePAPER
This ePaper is currently not available for download.
You can find similar magazines on this topic below under ‘Recommendations’.