<str<strong>on</strong>g>Proceedings</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> <str<strong>on</strong>g>Third</str<strong>on</strong>g> <str<strong>on</strong>g>Internati<strong>on</strong>al</str<strong>on</strong>g> <str<strong>on</strong>g>C<strong>on</strong>ference</str<strong>on</strong>g> <strong>on</strong> <strong>Invasive</strong> SpartinaChapter 1: Spartina Biologyrespective treatment (drained or flooded) before testing orharvest. All statistical analyses were performed betweenspecies and treatments with a two-factor (species andtreatment) analysis <str<strong>on</strong>g>of</str<strong>on</strong>g> variance (Statview 5; 1998 SASInstitute Inc., Cary, NC; α=0.05).Individual plants were tested for <str<strong>on</strong>g>the</str<strong>on</strong>g>ir ability to transportoxygen internally after <str<strong>on</strong>g>the</str<strong>on</strong>g> method <str<strong>on</strong>g>of</str<strong>on</strong>g> Maricle and Lee(2002). A fiber optic oxygen-sensing probe (FOXY-R probe;Ocean Optics Inc., Dunedin, FL) was used to measuredissolved oxygen c<strong>on</strong>centrati<strong>on</strong>s in sealed flasks c<strong>on</strong>tainingroots <str<strong>on</strong>g>of</str<strong>on</strong>g> intact plants suspended in water. Flask waterc<strong>on</strong>tained penicillin G and streptomycin sulfate at 1 gramper liter (1 g L -1 ) each and 50 mg L -1 chloramphenicol toprevent bacterial respirati<strong>on</strong>. During testing, plants wereplaced under a 250 watt (W) metal halide light (Hydr<str<strong>on</strong>g>of</str<strong>on</strong>g>armgardening products; Petaluma, CA). At plant level, PPFDwas about 150 μmol quanta m -2 s -1 and air temperature was28˚C. Flask dissolved oxygen c<strong>on</strong>centrati<strong>on</strong>s started at 100μM (±10 μM) and were measured over a period <str<strong>on</strong>g>of</str<strong>on</strong>g> 2-3 hoursto observe c<strong>on</strong>sumpti<strong>on</strong> or release <str<strong>on</strong>g>of</str<strong>on</strong>g> oxygen by plants to <str<strong>on</strong>g>the</str<strong>on</strong>g>surrounding medium.Parallel measurements <str<strong>on</strong>g>of</str<strong>on</strong>g> oxygen c<strong>on</strong>sumpti<strong>on</strong> werec<strong>on</strong>ducted <strong>on</strong> plants where aerenchyma transport capabilitieswere blocked. Placing <str<strong>on</strong>g>the</str<strong>on</strong>g> shoot <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> plant in a 100% N 2atmosphere prevents <str<strong>on</strong>g>the</str<strong>on</strong>g> entry <str<strong>on</strong>g>of</str<strong>on</strong>g> oxygen into <str<strong>on</strong>g>the</str<strong>on</strong>g>aerenchyma system (Armstr<strong>on</strong>g 1964, Teal and Kanwisher1966). Plants were maintained in <str<strong>on</strong>g>the</str<strong>on</strong>g> dark during thismeasurement to prevent <str<strong>on</strong>g>the</str<strong>on</strong>g> release <str<strong>on</strong>g>of</str<strong>on</strong>g> photosyn<str<strong>on</strong>g>the</str<strong>on</strong>g>ticoxygen. Rates <str<strong>on</strong>g>of</str<strong>on</strong>g> oxygen c<strong>on</strong>sumpti<strong>on</strong> were comparedbetween plants under a 21% O 2 atmosphere and a 100% N 2atmosphere; <str<strong>on</strong>g>the</str<strong>on</strong>g> difference between <str<strong>on</strong>g>the</str<strong>on</strong>g> two flux rates equals<str<strong>on</strong>g>the</str<strong>on</strong>g> amount <str<strong>on</strong>g>of</str<strong>on</strong>g> oxygen transported internally through <str<strong>on</strong>g>the</str<strong>on</strong>g>plant’s aerenchyma system (Lee 2003). Rates <str<strong>on</strong>g>of</str<strong>on</strong>g> internaloxygen transport were standardized to g fresh root weight.Oxygen c<strong>on</strong>sumpti<strong>on</strong> in <str<strong>on</strong>g>the</str<strong>on</strong>g> dark under 100% N 2 representstotal oxygen demand by <str<strong>on</strong>g>the</str<strong>on</strong>g> plant and was taken to be <str<strong>on</strong>g>the</str<strong>on</strong>g>dark respirati<strong>on</strong> rate (Maricle and Lee 2007).At harvest, root samples were obtained from each plant,flash-frozen in liquid nitrogen, and stored at –80°C.Cytochrome c oxidase (CytOx) and sulfide oxidase (SOx)activities were determined in extracts from root tissuesamples. Roots were ground in liquid nitrogen and coldextracti<strong>on</strong> buffer was added at 2 milliliters per gram (2 mLg -1 ) (Maxwell and Bateman 1967). This mixture washomogenized with a mortar and pestle, filtered throughMiracloth (Calbiochem; San Diego, CA), and centrifuged at1,000 g for 20 minutes at 4°C. The supernatant was used inCytOx and SOx assays. Alcohol dehydrogenase (ADH) wasextracted from root tissue samples after John and Greenway(1976). Roots were ground in liquid nitrogen, and coldextracti<strong>on</strong> buffer was added at 5 mL g -1 . The resultingmixture was homogenized with a mortar and pestle, filteredthrough Miracloth, and centrifuged at 10,000 g for 10 min at4°C. This supernatant was used in ADH assays. All enzymeassays were performed spectrophotometrically at 25°C(Maricle et al. 2006).CytOx activity was determined as <str<strong>on</strong>g>the</str<strong>on</strong>g> rate <str<strong>on</strong>g>of</str<strong>on</strong>g>cytochrome c oxidati<strong>on</strong>, measured as a decrease inabsorbance at 550 nanometers (nm) (Smith 1955). Rates <str<strong>on</strong>g>of</str<strong>on</strong>g>CytOx activity were corrected for background rates <str<strong>on</strong>g>of</str<strong>on</strong>g>cytochrome c oxidati<strong>on</strong>, <str<strong>on</strong>g>the</str<strong>on</strong>g>n standardized to g fresh rootweight. ADH activity was assayed spectrophotometrically in<str<strong>on</strong>g>the</str<strong>on</strong>g> ethanol-forming directi<strong>on</strong> (John and Greenway 1976).Enzyme activity was determined as <str<strong>on</strong>g>the</str<strong>on</strong>g> rate <str<strong>on</strong>g>of</str<strong>on</strong>g> NADHoxidati<strong>on</strong>, measured as a decrease in absorbance at 340 nm.Rates <str<strong>on</strong>g>of</str<strong>on</strong>g> NADH oxidati<strong>on</strong> in <str<strong>on</strong>g>the</str<strong>on</strong>g> presence <str<strong>on</strong>g>of</str<strong>on</strong>g> acetaldehydewere corrected for background rates <str<strong>on</strong>g>of</str<strong>on</strong>g> ethanol formati<strong>on</strong>,<str<strong>on</strong>g>the</str<strong>on</strong>g>n standardized to g fresh root weight (Maricle et al.2006).A colorimetric method was developed to measure <str<strong>on</strong>g>the</str<strong>on</strong>g>activity <str<strong>on</strong>g>of</str<strong>on</strong>g> sulfide oxidati<strong>on</strong> processes. 50 microliter (μL)aliquots <str<strong>on</strong>g>of</str<strong>on</strong>g> extract were added to a series <str<strong>on</strong>g>of</str<strong>on</strong>g> 1 mL buffersoluti<strong>on</strong>s <str<strong>on</strong>g>of</str<strong>on</strong>g> 100 μM Na 2 S. 40 μL <str<strong>on</strong>g>of</str<strong>on</strong>g> Cline reagent (Cline1969) was added to <str<strong>on</strong>g>the</str<strong>on</strong>g> soluti<strong>on</strong>s after 0, 10, and 20 min todetermine <str<strong>on</strong>g>the</str<strong>on</strong>g> amount <str<strong>on</strong>g>of</str<strong>on</strong>g> sulfide present. Background rates<str<strong>on</strong>g>of</str<strong>on</strong>g> sulfide oxidati<strong>on</strong> were measured by adding 50 μLphosphate buffer instead <str<strong>on</strong>g>of</str<strong>on</strong>g> enzyme extract to a series <str<strong>on</strong>g>of</str<strong>on</strong>g>tubes c<strong>on</strong>taining 100 μM Na 2 S as described above. SOxactivity was measured as a decrease in sulfide c<strong>on</strong>centrati<strong>on</strong>over time. Total rates <str<strong>on</strong>g>of</str<strong>on</strong>g> SOx activity were corrected forbackground rates <str<strong>on</strong>g>of</str<strong>on</strong>g> sp<strong>on</strong>taneous sulfide oxidati<strong>on</strong>, <str<strong>on</strong>g>the</str<strong>on</strong>g>nstandardized to g fresh root weight. Rates <str<strong>on</strong>g>of</str<strong>on</strong>g> n<strong>on</strong>enzymaticsulfide oxidati<strong>on</strong> were determined using 50 μL aliquots <str<strong>on</strong>g>of</str<strong>on</strong>g>boiled enzyme extract. Enzymatic rates <str<strong>on</strong>g>of</str<strong>on</strong>g> sulfide oxidati<strong>on</strong>were calculated from <str<strong>on</strong>g>the</str<strong>on</strong>g> difference between <str<strong>on</strong>g>the</str<strong>on</strong>g> rates <str<strong>on</strong>g>of</str<strong>on</strong>g>total sulfide oxidati<strong>on</strong> and n<strong>on</strong>enzymatic sulfide oxidati<strong>on</strong>(Maricle et al. 2006).RESULTS AND DISCUSSIONSoil waterlogging is <str<strong>on</strong>g>the</str<strong>on</strong>g> most comm<strong>on</strong> cause <str<strong>on</strong>g>of</str<strong>on</strong>g> plantoxygen deficiency (Vartapetian and Jacks<strong>on</strong> 1997). Impacts<str<strong>on</strong>g>of</str<strong>on</strong>g> flooding <strong>on</strong> plant productivity can also have significantimpacts <strong>on</strong> commodity crops. Excessive rains in <str<strong>on</strong>g>the</str<strong>on</strong>g> spring<str<strong>on</strong>g>of</str<strong>on</strong>g> 1993 resulted in a 33% reducti<strong>on</strong> in Midwest cropproducti<strong>on</strong> (Bray et al. 2000). Despite <str<strong>on</strong>g>the</str<strong>on</strong>g>se kinds <str<strong>on</strong>g>of</str<strong>on</strong>g>ec<strong>on</strong>omic losses, many observers may have overlooked <str<strong>on</strong>g>the</str<strong>on</strong>g>cattails and o<str<strong>on</strong>g>the</str<strong>on</strong>g>r wetland plants flourishing in nearbyflooded ditches. Differences in flooding tolerance have l<strong>on</strong>gbeen recognized between plant species, but <str<strong>on</strong>g>the</str<strong>on</strong>g> specificphysiology governing <str<strong>on</strong>g>the</str<strong>on</strong>g> differences has remained largelyunknown.Internal oxygen transport is <str<strong>on</strong>g>of</str<strong>on</strong>g>ten regarded to be animportant factor c<strong>on</strong>ferring plant success in waterloggedareas, and <str<strong>on</strong>g>the</str<strong>on</strong>g>refore is thought to be important in estuarinez<strong>on</strong>ati<strong>on</strong> (e.g., Gleas<strong>on</strong> and Zieman 1981, Bertness 1991).The oxygen transport rates measured in this study exhibited-49-
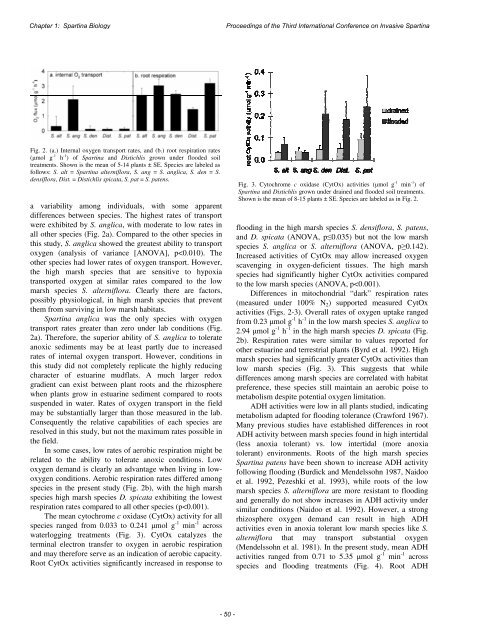
Chapter 1: Spartina Biology<str<strong>on</strong>g>Proceedings</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> <str<strong>on</strong>g>Third</str<strong>on</strong>g> <str<strong>on</strong>g>Internati<strong>on</strong>al</str<strong>on</strong>g> <str<strong>on</strong>g>C<strong>on</strong>ference</str<strong>on</strong>g> <strong>on</strong> <strong>Invasive</strong> SpartinaFig. 2. (a.) Internal oxygen transport rates, and (b.) root respirati<strong>on</strong> rates(μmol g -1 h -1 ) <str<strong>on</strong>g>of</str<strong>on</strong>g> Spartina and Distichlis grown under flooded soiltreatments. Shown is <str<strong>on</strong>g>the</str<strong>on</strong>g> mean <str<strong>on</strong>g>of</str<strong>on</strong>g> 5-14 plants ± SE. Species are labeled asfollows: S. alt = Spartina alterniflora, S. ang = S. anglica, S. den = S.densiflora, Dist. = Distichlis spicata, S. pat = S. patens.a variability am<strong>on</strong>g individuals, with some apparentdifferences between species. The highest rates <str<strong>on</strong>g>of</str<strong>on</strong>g> transportwere exhibited by S. anglica, with moderate to low rates inall o<str<strong>on</strong>g>the</str<strong>on</strong>g>r species (Fig. 2a). Compared to <str<strong>on</strong>g>the</str<strong>on</strong>g> o<str<strong>on</strong>g>the</str<strong>on</strong>g>r species inthis study, S. anglica showed <str<strong>on</strong>g>the</str<strong>on</strong>g> greatest ability to transportoxygen (analysis <str<strong>on</strong>g>of</str<strong>on</strong>g> variance [ANOVA], p
- Page 2 and 3:
Proceedings <stron
- Page 4 and 5:
FORWARD & ACKNOWLEDGEMENTSThe <stro
- Page 6 and 7:
TABLE OF CONTENTSForward & Acknowle
- Page 9 and 10:
Community Spartina Education and St
- Page 11 and 12: included the docum
- Page 14: CHAPTER ONESpartina Biology
- Page 17 and 18: Chapter 1: Spartina Biology
- Page 19 and 20: Chapter 1: Spartina Biology
- Page 21 and 22: Chapter 1: Spartina Biology
- Page 23 and 24: Chapter 1: Spartina Biology
- Page 25 and 26: Chapter 1: Spartina Biology
- Page 28 and 29: Proceedings <stron
- Page 30 and 31: Proceedings <stron
- Page 32 and 33: Proceedings <stron
- Page 34: Proceedings <stron
- Page 37 and 38: Chapter 1: Spartina Biology
- Page 39 and 40: Chapter 1: Spartina Biology
- Page 42 and 43: Proceedings <stron
- Page 44: Proceedings <stron
- Page 47 and 48: Chapter 1: Spartina Biology
- Page 49 and 50: Chapter 1: Spartina Biology
- Page 51 and 52: Chapter 1: Spartina Biology
- Page 53 and 54: Chapter 1: Spartina Biology
- Page 55 and 56: Chapter 1: Spartina Biology
- Page 57 and 58: Chapter 1: Spartina Biology
- Page 60 and 61: Proceedings <stron
- Page 64 and 65: Proceedings <stron
- Page 66: Proceedings <stron
- Page 69 and 70: Chapter 1: Spartina Biology
- Page 71 and 72: Chapter 1: Spartina Biology
- Page 74 and 75: Proceedings <stron
- Page 76: Proceedings <stron
- Page 79 and 80: Chapter 2: Spartina Distribution an
- Page 81 and 82: Chapter 2: Spartina Distribution an
- Page 83 and 84: Chapter 2: Spartina Distribution an
- Page 86 and 87: Proceedings <stron
- Page 88 and 89: Proceedings <stron
- Page 90 and 91: Proceedings <stron
- Page 92 and 93: Proceedings <stron
- Page 94 and 95: Proceedings <stron
- Page 96 and 97: Proceedings <stron
- Page 98: Proceedings <stron
- Page 101 and 102: Chapter 2: Spartina Distribution an
- Page 103 and 104: Chapter 2: Spartina Distribution an
- Page 105 and 106: Chapter 2: Spartina Distribution an
- Page 108 and 109: Proceedings <stron
- Page 110: Proceedings <stron
- Page 113 and 114:
Chapter 2: Spartina Distribution an
- Page 115 and 116:
Chapter 2: Spartina Distribution an
- Page 117 and 118:
Chapter 2: Spartina Distribution an
- Page 119 and 120:
Chapter 2: Spartina Distribution an
- Page 122 and 123:
Proceedings <stron
- Page 124 and 125:
Proceedings <stron
- Page 126 and 127:
Proceedings <stron
- Page 128:
Proceedings <stron
- Page 131 and 132:
Chapter 2: Spartina Distribution an
- Page 134 and 135:
Proceedings <stron
- Page 136 and 137:
Proceedings <stron
- Page 138 and 139:
Proceedings <stron
- Page 140:
CHAPTER THREEEcosystem Effects <str
- Page 143 and 144:
Chapter 3: Ecosystem Effects <stron
- Page 145 and 146:
Chapter 3: Ecosystem Effects <stron
- Page 148 and 149:
Proceedings <stron
- Page 150 and 151:
Proceedings <stron
- Page 152:
Proceedings <stron
- Page 155 and 156:
Chapter 3: Ecosystem Effects <stron
- Page 157 and 158:
Chapter 3: Ecosystem Effects <stron
- Page 160 and 161:
Proceedings <stron
- Page 162 and 163:
Proceedings <stron
- Page 164:
Proceedings <stron
- Page 167 and 168:
Chapter 3: Ecosystem Effects <stron
- Page 169 and 170:
Chapter 3: Ecosystem Effects <stron
- Page 171 and 172:
Chapter 3: Ecosystem Effects <stron
- Page 174 and 175:
Proceedings <stron
- Page 176:
Proceedings <stron
- Page 179 and 180:
Chapter 3: Ecosystem Effects <stron
- Page 181 and 182:
Chapter 3: Ecosystem Effects <stron
- Page 184 and 185:
Proceedings <stron
- Page 186 and 187:
Proceedings <stron
- Page 188 and 189:
Proceedings <stron
- Page 190 and 191:
Proceedings <stron
- Page 192 and 193:
Proceedings <stron
- Page 194 and 195:
Proceedings <stron
- Page 196:
Proceedings <stron
- Page 199 and 200:
Chapter 3: Ecosystem Effects <stron
- Page 201 and 202:
Chapter 3: Ecosystem Effects <stron
- Page 204 and 205:
Proceedings <stron
- Page 206 and 207:
Proceedings <stron
- Page 208 and 209:
Proceedings <stron
- Page 210 and 211:
Proceedings <stron
- Page 212:
Proceedings <stron
- Page 216 and 217:
Proceedings <stron
- Page 218 and 219:
Proceedings <stron
- Page 220 and 221:
Proceedings <stron
- Page 222 and 223:
Proceedings <stron
- Page 224 and 225:
Proceedings <stron
- Page 226 and 227:
Proceedings <stron
- Page 228 and 229:
Proceedings <stron
- Page 230 and 231:
Proceedings <stron
- Page 232 and 233:
Proceedings <stron
- Page 234 and 235:
Proceedings <stron
- Page 236 and 237:
Proceedings <stron
- Page 238 and 239:
Proceedings <stron
- Page 240 and 241:
Proceedings <stron
- Page 242 and 243:
Proceedings <stron
- Page 244 and 245:
Proceedings <stron
- Page 246:
Proceedings <stron
- Page 249 and 250:
Chapter 4: Spartina Control and Man
- Page 251 and 252:
Chapter 4: Spartina Control and Man
- Page 253 and 254:
Chapter 4: Spartina Control and Man
- Page 255 and 256:
Chapter 4: Spartina Control and Man
- Page 257 and 258:
Chapter 4: Spartina Control and Man
- Page 259 and 260:
Chapter 4: Spartina Control and Man
- Page 261 and 262:
Chapter 4: Spartina Control and Man
- Page 263 and 264:
Chapter 4: Spartina Control and Man
- Page 265 and 266:
Chapter 4: Spartina Control and Man
- Page 267 and 268:
Chapter 4: Spartina Control and Man
- Page 269 and 270:
Chapter 4: Spartina Control and Man
- Page 271 and 272:
Chapter 4: Spartina Control and Man
- Page 273 and 274:
Chapter 4: Spartina Control and Man
- Page 276 and 277:
Proceedings <stron
- Page 278 and 279:
Proceedings <stron
- Page 280 and 281:
Proceedings <stron
- Page 282 and 283:
Proceedings <stron
- Page 284 and 285:
Proceedings <stron
- Page 286 and 287:
Proceedings <stron
- Page 288 and 289:
Proceedings <stron
- Page 290:
Proceedings <stron