- Page 1:
United StatesEnvironmental Protecti
- Page 5:
ACKNOWLEDGMENTSThis document was pr
- Page 8 and 9:
2.5.7 Reverse Osmosis .............
- Page 10 and 11:
5.0 POINT-OF-ENTRY/POINT-OF-USE TRE
- Page 12 and 13:
LIST OF FIGURES2-1 Pressure Driven
- Page 14 and 15:
LIST OF ACRONYMSAAAWWAAWWARFBLSBVC/
- Page 16 and 17:
POUpoint-of-useppbparts per billion
- Page 18 and 19:
# Alternative treatment processes s
- Page 20 and 21:
This page was intentionally left bl
- Page 23 and 24:
ends up as ferric hydroxide. In alu
- Page 25 and 26:
Optimization Hierarchy for Coagulat
- Page 27 and 28:
Substantial arsenic removal has bee
- Page 29 and 30:
Vickers et al. (1997) reported that
- Page 31 and 32:
was reduced to 0.05 mg/L when the i
- Page 33 and 34:
Field StudiesSurveys of lime soften
- Page 35 and 36:
Effect of pHpH may have significant
- Page 37 and 38:
RegenerationRegeneration of AA beds
- Page 39 and 40:
een developed provide important inf
- Page 41 and 42:
applicability of IX at a particular
- Page 43 and 44:
TABLE 2-1Typical IX Resins for Arse
- Page 45 and 46:
solution strength. Arsenic elutes r
- Page 47 and 48:
2.4.12 Typical Design ParametersThr
- Page 49 and 50:
2.5.2 Important Factors for Membran
- Page 51 and 52:
arsenic size distribution to correl
- Page 53 and 54:
AWWARF (1998) also performed UF pil
- Page 55 and 56:
presumably due to changes in electr
- Page 57 and 58:
RO performance is adversely affecte
- Page 59 and 60:
TABLE 2-10Arsenic Removal with RO a
- Page 61 and 62:
eversal is the decreased potential
- Page 63 and 64:
2.6 ALTERNATIVE TECHNOLOGIES2.6.1 I
- Page 65 and 66:
20 mg As per gram of iron was remov
- Page 67 and 68:
The most significant weakness of th
- Page 69 and 70:
3.0 TECHNOLOGY COSTS3.1 INTRODUCTIO
- Page 71 and 72:
included in the estimates presented
- Page 73 and 74:
Table 3-3Water Model Capital Cost B
- Page 75 and 76:
3.2.3 Implementing TDP Recommended
- Page 77 and 78:
sources. The September 1998 index v
- Page 79 and 80:
Amortization, or capital recovery,
- Page 81 and 82:
3.3 ADDITIONAL CAPITAL COSTSThe cos
- Page 83 and 84:
PilotingThe Technology Design Panel
- Page 85 and 86:
The 1993 Technology and Cost Docume
- Page 87 and 88:
cause disinfection by-product (DBP)
- Page 89 and 90:
Figure 3-1Pre-oxidation - 1.5 mg/L
- Page 91 and 92:
3.6 PRECIPITATIVE PROCESSES3.6.1 Co
- Page 93 and 94:
enhanced coagulation treatment plan
- Page 95 and 96:
Figure 3-4Enhanced Coagulation/Filt
- Page 97 and 98:
Small Systems (Less than 1 mgd)The
- Page 99 and 100:
Figure 3-6Coagulation Assisted Micr
- Page 101 and 102:
3.6.6 Enhanced Lime SofteningEnhanc
- Page 103 and 104:
Figure 3-8Enhanced Lime SofteningO&
- Page 105 and 106:
3. Empty Bed Contact Time (EBCT) is
- Page 107 and 108:
For design flows greater than 1 mgd
- Page 109 and 110:
Figure 3-9Activated AluminaCapital
- Page 111 and 112:
Figure 3-11Activated Alumina (pH 8
- Page 113 and 114:
Figure 3-13Activated Alumina (pH Ad
- Page 115 and 116:
3.7.2 Granular Ferric HydroxideGran
- Page 117 and 118:
6. The capital costs include a redu
- Page 119 and 120:
Figure 3-15Bed Volumes to Arsenic B
- Page 121 and 122:
Figure 3-17Anion Exchange (< 20 mg/
- Page 123 and 124:
Figure 3-19Anion Exchange (20-50 mg
- Page 125 and 126:
quality feed stream and often requi
- Page 127 and 128:
Figure 3-20Greensand FiltrationCapi
- Page 129 and 130:
3.11 COMPARISON OF COSTSThe April 1
- Page 131 and 132:
4.0 RESIDUALS HANDLING AND DISPOSAL
- Page 133 and 134:
mechanical dewatering processes. Wh
- Page 135 and 136:
Storage lagoons are best suited for
- Page 137 and 138:
the current Industrial Pretreatment
- Page 139 and 140:
4.3.3 Dewatered Sludge Land Applica
- Page 141 and 142:
usually determined by the Paint Fil
- Page 143 and 144:
for arsenic toxicity by a substanti
- Page 145 and 146:
The solids content of the backwash
- Page 147 and 148:
carbonate hardness removal produces
- Page 149 and 150:
Domestic sewage means untreated san
- Page 151 and 152:
4.4.10 Reverse OsmosisReverse osmos
- Page 153 and 154:
Figure 4-1Anion Exchange (< 20 mg/L
- Page 155 and 156:
Figure 4-3Anion Exchange (20-50 mg/
- Page 157 and 158:
Figure 4-5Coagulation Assisted Micr
- Page 159 and 160:
Figure 4-7Coagulation Assisted Micr
- Page 161 and 162: Figure 4-9Activated Alumina (pH 7 -
- Page 163 and 164: Figure 4-11Activated Alumina (pH Ad
- Page 165 and 166: Figure 4-13Greensand FiltrationWast
- Page 167 and 168: 5.0 POINT-OF-ENTRY/POINT-OF-USE TRE
- Page 169 and 170: chromatographic peaking, which occu
- Page 171 and 172: 5.3.1 Case Study 1: Fairbanks, Alas
- Page 173 and 174: Manufacturer and laboratory data su
- Page 175 and 176: Figure 5-2POU Reverse OsmosisO&M Co
- Page 177 and 178: # Installation time - 1 hour unskil
- Page 179 and 180: Figure 5-3POU Activated AluminaTota
- Page 181 and 182: 6.0 REFERENCESAmy, G.L. and P. Bran
- Page 183 and 184: Cooperative Research Centres for Wa
- Page 185 and 186: Fuller, C.C., J.A. Davis, G.W. Zell
- Page 187 and 188: Le, X.C., and M. Ma (1998). “Dete
- Page 189 and 190: Scott, K., J. Green, H.D. Do and S.
- Page 191: Appendix AVery Small Systems Capita
- Page 194 and 195: Table A4 - VSS Document Capital Cos
- Page 197 and 198: Table B1.1 - Base Costs Obtained fr
- Page 199 and 200: Table B3.1 - Base Costs Obtained fr
- Page 201 and 202: Table B5.1 - Base Costs Obtained fr
- Page 203 and 204: Table B7.1 - Base Costs Obtained fr
- Page 205 and 206: Table B9.1 - Base Costs Obtained fr
- Page 207 and 208: Table B11.1 - Base Costs Obtained f
- Page 209 and 210: Table B13.1 - Base Costs Obtained f
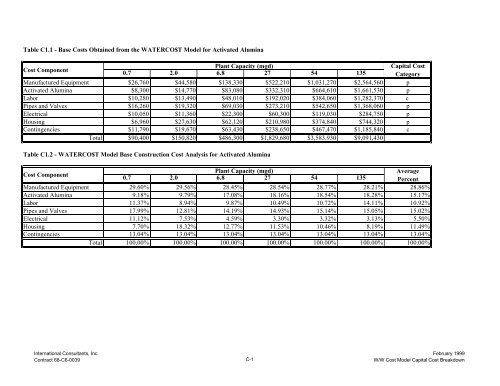
- Page 211: Appendix CW/W Cost Model Capital Co
- Page 215 and 216: Table C3.1 - Base Costs Obtained fr
- Page 217 and 218: Table C5.1 - Base Costs Obtained fr
- Page 219 and 220: Table C7.1 - Base Costs Obtained fr
- Page 221 and 222: Table C9.1 - Base Costs Obtained fr
- Page 223 and 224: Table C11.1 - Base Costs Obtained f
- Page 225 and 226: Table C13.1 - Base Costs Obtained f
- Page 227 and 228: Table C15.1 - Base Costs Obtained f
- Page 229 and 230: Table C17.1 - Base Costs Obtained f
- Page 231 and 232: Table C19.1 - Base Costs Obtained f
- Page 233 and 234: Table C21.1 - Base Costs Obtained f
- Page 235 and 236: Table C23.1 - Base Costs Obtained f
- Page 237 and 238: Table C25.1 - Base Costs Obtained f
- Page 239 and 240: Table C27.1 - Base Costs Obtained f
- Page 241: Appendix DBasis for Revised Activat
- Page 244 and 245: much more oversight than most small
- Page 246 and 247: Basis. An old activated alumina des
- Page 248 and 249: Basis. The Water Model provided con
- Page 250 and 251: systems could underestimate the nee
- Page 252 and 253: For design flows greater than 1 mgd
- Page 254 and 255: The source water at Plant C has the
- Page 256 and 257: length of 10,000 BV for 7 # pH < 8
- Page 258 and 259: The arsenic loaded onto the column
- Page 260 and 261: column. The Phase 3 Report noted th
- Page 262 and 263:
The lower bound run length at pH 6
- Page 264 and 265:
Basis. The labor requirements when
- Page 266 and 267:
23. The disposal cost for the spent
- Page 268 and 269:
8. US EPA. Guide for Implementing P
- Page 271 and 272:
APPENDIX E: BASIS FOR REVISED ANION
- Page 273 and 274:
6. The capital costs include a redu
- Page 275 and 276:
Table E-2Percentage of Ground Water
- Page 277 and 278:
12. The building cost to house the
- Page 279 and 280:
15. The second major component in t
- Page 281 and 282:
Arsenic in the regeneration brine w
- Page 283 and 284:
12. Clifford, D.A. G. Ghurye et al.