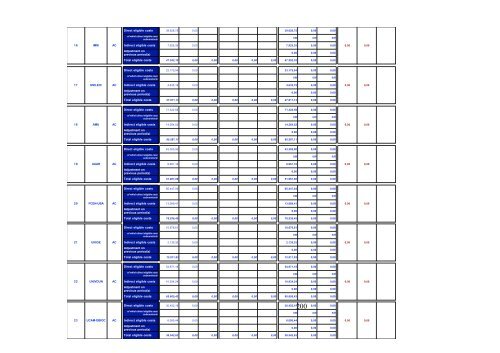
Direct eligible costs 53.240,48 0,00 53.240,48 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,008 MUW AC Indirect eligible costs 10.648,10 0,00 10.648,10 0,00 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 63.888,58 0,00 0,00 0,00 0,00 0,00 63.888,58 0,00 0,00Direct eligible costs 34.349,18 0,00 34.349,18 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,009 UEDINACIndirect eligible costs 6.869,83 0,00 6.869,83 0,00 0,000,000,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 41.219,01 0,00 0,00 0,00 0,00 0,00 41.219,01 0,00 0,00Direct eligible costs 118.241,98 0,00 118.241,98 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0010 CNR FC Indirect eligible costs 74.183,17 0,00 74.183,17 0,00 0,00 0,00Adjustment onprevious period(s)-6.945,39 -6.945,39 0,00 0,00Total eligible costs 185.479,76 0,00 0,00 0,00 0,00 0,00 185.479,76 0,00 0,000,00Direct eligible costs 33.054,83 0,00 33.054,83 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0011 LUGAC Indirect eligible costs 6.610,97 0,00 6.610,97 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 39.665,80 0,00 0,00 0,00 0,00 0,00 39.665,80 0,00 0,000,00Direct eligible costs 298.610,03 26.254,91 298.610,03 0,00 26.254,91of which direct eligible costs ofsubcontracting0,00 0,00 0,0012 CNRS FCF Indirect eligible costs 59.722,01 5.250,98 59.722,01 0,00 5.250,98 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 358.332,04 0,00 31.505,89 0,00 0,00 0,00 358.332,04 0,00 31.505,890,00Direct eligible costs 52.364,00 0,00 52.364,00 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0013 UNIBE AC Indirect eligible costs 10.472,80 0,00 10.472,80 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 62.836,80 0,00 0,00 0,00 0,00 0,00 62.836,80 0,00 0,000,00Direct eligible costs 99.352,86 0,00 39.650,93 99.352,86 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0014 SCRI AC Indirect eligible costs 15.512,60 0,00 7.930,19 15.512,60 0,00 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 114.865,46 0,00 0,00 47.581,12 0,00 0,00 114.865,46 0,00 0,00199Direct eligible costs 27.721,87 0,00 27.721,87 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0015 MRC FC Indirect eligible costs 155,82 155,82 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 27.877,69 0,00 0,00 0,00 0,00 0,00 27.877,69 0,00 0,000,00
Direct eligible costs 39.626,75 0,00 39.626,75 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0016 IMM AC Indirect eligible costs 7.925,35 0,00 7.925,35 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 47.552,10 0,00 0,00 0,00 0,00 0,00 47.552,10 0,00 0,000,00Direct eligible costs 23.175,94 0,00 23.175,94 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0017 UNILEIC AC Indirect eligible costs 4.635,19 0,00 4.635,19 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 27.811,13 0,00 0,00 0,00 0,00 0,00 27.811,13 0,00 0,000,00Direct eligible costs 71.322,59 0,00 71.322,59 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0018 AMU AC Indirect eligible costs 14.264,52 0,00 14.264,52 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 85.587,11 0,00 0,00 0,00 0,00 0,00 85.587,11 0,00 0,000,00Direct eligible costs 43.305,90 0,00 43.305,90 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0019 UAARAC Indirect eligible costs 8.661,18 0,00 8.661,18 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 51.967,08 0,00 0,00 0,00 0,00 0,00 51.967,08 0,00 0,000,00Direct eligible costs 65.447,04 0,00 65.447,04 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0020 FCEN-UBA AC Indirect eligible costs 13.089,41 0,00 13.089,41 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 78.536,45 0,00 0,00 0,00 0,00 0,00 78.536,45 0,00 0,000,00Direct eligible costs 10.676,61 0,00 10.676,61 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0021 UNIGE AC Indirect eligible costs 2.135,32 0,00 2.135,32 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 12.811,93 0,00 0,00 0,00 0,00 0,00 12.811,93 0,00 0,000,00Direct eligible costs 54.671,19 0,00 54.671,19 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0022 UNIVDUN AC Indirect eligible costs 10.934,24 0,00 10.934,24 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 65.605,43 0,00 0,00 0,00 0,00 0,00 65.605,43 0,00 0,00200Direct eligible costs 30.452,19 0,00 30.452,19 0,00 0,00of which direct eligible costs ofsubcontracting0,00 0,00 0,0023 UCAM-DBIOC AC Indirect eligible costs 6.090,44 0,00 6.090,44 0,00 0,00 0,00Adjustment onprevious period(s)0,00 0,00 0,00Total eligible costs 36.542,63 0,00 0,00 0,00 0,00 0,00 36.542,63 0,00 0,000,000,00
- Page 3 and 4:
TABLE OF CONTENTSA. PERIODIC ACTIVI
- Page 5 and 6:
1 PUBLISHABLE EXECUTIVE SUMMARY EUR
- Page 7 and 8:
Dr. Davide Gabellini(28) Swiss Inst
- Page 9 and 10:
WP7 Molecular characterization of s
- Page 11 and 12:
To identify protein-protein interac
- Page 13 and 14:
expressed in a particular tissue. T
- Page 15 and 16:
Duchenne muscular dystrophy (DMD).
- Page 17 and 18:
WP21 Reachout to the broader RNA co
- Page 19 and 20:
4.1 TABLE OF MILESTONES JPA MONTH 2
- Page 21 and 22:
Numerous plasmids, constructs, extr
- Page 23 and 24:
collaboration between EURASNET and
- Page 25 and 26:
5.1 WORK PACKAGE REPORTSWork Packag
- Page 27 and 28:
• Ongoing regular addition of new
- Page 29 and 30:
Work Package 4 The Alternative Spli
- Page 31 and 32:
Work Package 6 In silico approaches
- Page 33 and 34:
alternative splicing.162 Compare sp
- Page 35 and 36:
33 Agreement on pre-mRNA substrates
- Page 37 and 38:
its numerous binding sites on both
- Page 39 and 40:
Work Package 8 Genome-wide Analyses
- Page 41 and 42:
173 Further development of data ana
- Page 43 and 44:
Problems and explanations for delay
- Page 45 and 46:
eveal that there is a dramatic chan
- Page 47 and 48:
Work Package 10 Post-translational
- Page 49 and 50:
mutational analysis that led to the
- Page 51 and 52:
Isolation of an active step I splic
- Page 53 and 54:
FBP11 and 21 with these sequences.
- Page 55 and 56:
Similarly to what observed for SF2/
- Page 57 and 58:
prp45-113 mutant, which is compatib
- Page 59 and 60:
cell lines. In contrast in the pres
- Page 61 and 62:
genes with roles in the development
- Page 63 and 64:
Structural analyses of several RRMs
- Page 65:
Work Package 17 Staff Exchange and
- Page 69 and 70:
Barralle's lab, Krämer is acting a
- Page 71 and 72:
alternative splicing) and higher ed
- Page 73 and 74:
Student Symposium “Decision Makin
- Page 75 and 76:
variation and suitability for use w
- Page 77 and 78:
Following feedback from members we
- Page 79 and 80:
Invited seminars: In addition to me
- Page 81 and 82:
Publications 2008Author Title Publi
- Page 83 and 84:
Author Title Publication EURASNET P
- Page 85 and 86:
Author Title Publication EURASNET P
- Page 87 and 88:
Author Title Publication EURASNET P
- Page 89 and 90:
Author Title Publication EURASNET P
- Page 91 and 92:
Author Title Publication EURASNET P
- Page 93 and 94:
Minutes of the General Assembly dec
- Page 96 and 97:
241 Evaluate available careerdevelo
- Page 98 and 99:
156 The EURASNET memberStamm publis
- Page 100 and 101:
y coarse-grained foldingsimulation.
- Page 102 and 103:
overexpressor and mutant lines(mont
- Page 104 and 105:
machinery with particularrespect to
- Page 106 and 107:
alternative splicing, using theEDI
- Page 108 and 109:
contemporary findings withinthe EUR
- Page 110 and 111:
Author Title Publication EURASNET P
- Page 112 and 113:
Author Title Publication EURASNET P
- Page 114 and 115:
Author Title Publication EURASNET P
- Page 116 and 117:
Author Title Publication EURASNET P
- Page 118 and 119:
Licht K, Medenbach J, Lührmann R,K
- Page 120 and 121:
Author Title Publication EURASNET P
- Page 122 and 123:
9 TABULAR OVERVIEW OF MAJOR COST IT
- Page 124 and 125:
total costs * 18.891,12 52778.60 41
- Page 126 and 127:
other (the rest) 48,57 0 56.539,43t
- Page 128 and 129:
travel 6.964,91overhead 3.081,07oth
- Page 130 and 131:
Participant 1A Type of expenditure
- Page 132 and 133:
Participant 1B - Karla Neugebauera)
- Page 134 and 135:
Participant 2 - Juan Valcarcela) Wo
- Page 136 and 137:
Participant 3 - Stefan Stamma) Work
- Page 138 and 139:
Participant 4 - Göran Akusjärvia)
- Page 140 and 141:
Participant 5A/B - Peer Bork/Rolf A
- Page 142 and 143:
Major cost items• ResearchPersonn
- Page 144 and 145:
Participant 7 - Francisco Barallea)
- Page 146 and 147:
Francisco Baralle gave a talk to a
- Page 148 and 149:
alternative splicing on human disea
- Page 150 and 151: J. Beggs has had frequent contact w
- Page 152 and 153: Major cost items• ResearchPersonn
- Page 154 and 155: Major cost items• ResearchPersonn
- Page 156 and 157: Participant 12A - Jamal Tazia) Work
- Page 158 and 159: Participant 12B - Bertrand Séraphi
- Page 160 and 161: Participant 12C - Christiane Branla
- Page 162 and 163: Participant 12D - Edouard Bertranda
- Page 164 and 165: Participant 13 - Daniel Schümperli
- Page 166 and 167: Participant 14 - John W. S. Browna)
- Page 168 and 169: Participant 15 - Javier F. Cáceres
- Page 170 and 171: • ResearchPersonnelName WP Person
- Page 172 and 173: MiscellaneousOligonucleotidesUse of
- Page 174 and 175: Major cost items• ResearchConsuma
- Page 176 and 177: Major cost items• ResearchPersonn
- Page 178 and 179: Participant 21 - Angela Krämera) W
- Page 180 and 181: Participant 22 - Angus Lamonda) Wor
- Page 182 and 183: Participant 23 - Chris Smitha) Work
- Page 184 and 185: ChemicalsEnzymesLaboratory supplies
- Page 186 and 187: Participant 26 - Didier Auboeufa) W
- Page 188 and 189: tool for the molecular analysis of
- Page 190 and 191: Participant 29 - Frédéric Allaina
- Page 192 and 193: protein and instead possesses the d
- Page 194 and 195: + overhead 4.730= total eligible co
- Page 196 and 197: Claudia Tamaro (PhD) has begun work
- Page 198 and 199: ETHZIIMCBUPFSOTON-HGDTOTALJoint Pro
- Page 202 and 203: Direct eligible costs 18.720,07 0,0
- Page 204 and 205: 13 REPORT ON THE DISTRIBUTION OF TH
- Page 206 and 207: Report on the Distribution of the C
- Page 208 and 209: For most work packages here follows
- Page 210 and 211: pathways (eg, Reactome ).While PAND
- Page 212 and 213: The possible dependence of the effe
- Page 214 and 215: In some treatments or mutants, simi
- Page 216 and 217: Work Package 12: Mis-splicing and D
- Page 218 and 219: acid. Furthermore, using antibodies
- Page 220 and 221: At the 2007 annual meeting in Ile d
- Page 222 and 223: Partner 13 has extensively studied
- Page 224 and 225: inhibitors/modulators of RNA splici
- Page 226 and 227: 18 MONTH JPA: WORKPACKAGE TIMECOURS
- Page 228 and 229: 15.3 GRAPHICAL PRESENTATION OF WORK
- Page 230 and 231: 15.4 TABLE OF MILESTONES (MONTH 37-
- Page 232 and 233: 156 The EURASNET memberStamm publis
- Page 234 and 235: 194 Bertrand will analyze indetail
- Page 236 and 237: 216 Aubeouf and Neugebauer willcoll
- Page 238 and 239: 238 Establishing joint PhDcommittee
- Page 240 and 241: 283 A unified browser for allknown
- Page 242 and 243: 296 Group 12a (Branlant) willuse th
- Page 244 and 245: 315 Lamond will use a FLIM-FRET bas
- Page 246 and 247: 329 GROUP 7 (F. BARALLE)will contin
- Page 248 and 249: 333 GROUP 27 (GABELLINI)will:1) foc
- Page 250 and 251:
345 Schümperli’s group isplannin
- Page 252 and 253:
366 Quarterly network e-mail atmont
- Page 254 and 255:
2. Sharing Resources, Technology an
- Page 256 and 257:
5. Ensuring DurabilityWorkpackage d
- Page 258 and 259:
7. Molecular Characterization of Sp
- Page 260 and 261:
8. Genome-wide Analyses of Splicing
- Page 262 and 263:
9. Complexity of Spliceosomal Prote
- Page 264 and 265:
202 Srebrow will investigate the ro
- Page 266 and 267:
12. Mis-splicing and DiseaseWorkpac
- Page 268 and 269:
13. Co-transcriptional Mechanisms o
- Page 270 and 271:
14. Chemical Biology and Therapeuti
- Page 272 and 273:
15. Development of Enabling Technol
- Page 274 and 275:
269 IFMs in 20091. “Alternative a
- Page 276 and 277:
18. Career DevelopmentWorkpackage d
- Page 278 and 279:
20. SMEs and Technology TransferWor
- Page 280 and 281:
21. Reachout to the Broader RNA Com
- Page 282 and 283:
15.7 WORK PACKAGE LIST (MONTHS 37-5
- Page 284 and 285:
ETHZIIMCBUPFSOTON-HGDTOTALJoint Pro
- Page 286 and 287:
AMU 5 29 8 42UAAR 5 145 16 1665 29
- Page 288 and 289:
Joint Programme of Activities RESEA
- Page 290 and 291:
60 month period 18 month periodRequ
- Page 292 and 293:
Integration1. Young Investigator Pr
- Page 294 and 295:
UCAM-DBIOC 2350,000x1/3 € 50,000x
- Page 296:
the management team. Audit costs ar