Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
A<br />
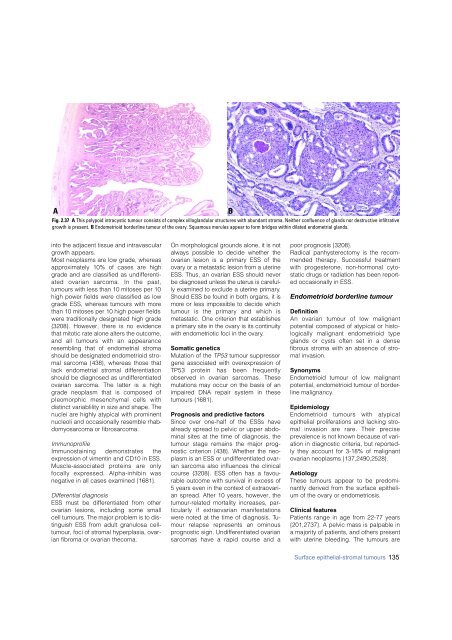
Fig. 2.37 A This polypoid intracystic tumour consists of complex villoglandular structures with abundant stroma. Neither confluence of glands nor destructive infiltrative<br />
growth is present. B Endometrioid borderline tumour of the ovary. Squamous morules appear to form bridges within dilated endometrial glands.<br />
B<br />
into the adjacent tissue and intravascular<br />
growth appears.<br />
Most neoplasms are low grade, whereas<br />
approximately 10% of cases are high<br />
grade and are classified as undifferentiated<br />
ovarian sarcoma. In the past,<br />
tumours with less than 10 mitoses per 10<br />
high power fields were classified as low<br />
grade ESS, whereas tumours with more<br />
than 10 mitoses per 10 high power fields<br />
were traditionally designated high grade<br />
{3208}. However, there is no evidence<br />
that mitotic rate alone alters the outcome,<br />
and all tumours with an appearance<br />
resembling that of endometrial stroma<br />
should be designated endometrioid stromal<br />
sarcoma {438}, whereas those that<br />
lack endometrial stromal differentiation<br />
should be diagnosed as undifferentiated<br />
ovarian sarcoma. The latter is a high<br />
grade neoplasm that is composed of<br />
pleomorphic mesenchymal cells with<br />
distinct variablility in size and shape. The<br />
nuclei are highly atypical with prominent<br />
nucleoli and occasionally resemble rhabdomyosarcoma<br />
or fibrosarcoma.<br />
Immunoprofile<br />
Immunostaining demonstrates the<br />
expression of vimentin and CD10 in ESS.<br />
Muscle-associated proteins are only<br />
focally expressed. Alpha-inhibin was<br />
negative in all cases examined {1681}.<br />
Differential diagnosis<br />
ESS must be differentiated from other<br />
ovarian lesions, including some small<br />
cell tumours. The major problem is to distinguish<br />
ESS from adult granulosa celltumour,<br />
foci of stromal hyperplasia, ovarian<br />
fibroma or ovarian thecoma.<br />
On morphological grounds alone, it is not<br />
always possible to decide whether the<br />
ovarian lesion is a primary ESS of the<br />
o v a ry or a metastatic lesion from a uterine<br />
ESS. Thus, an ovarian ESS should never<br />
be diagnosed unless the uterus is care f u l-<br />
ly examined to exclude a uterine primary.<br />
Should ESS be found in both organs, it is<br />
m o re or less impossible to decide which<br />
tumour is the primary and which is<br />
metastatic. One criterion that establishes<br />
a primary site in the ovary is its continuity<br />
with endometriotic foci in the ovary.<br />
Somatic genetics<br />
Mutation of the TP53 tumour suppressor<br />
gene associated with overexpression of<br />
TP53 protein has been fre q u e n t l y<br />
observed in ovarian sarcomas. These<br />
mutations may occur on the basis of an<br />
impaired DNA repair system in these<br />
tumours {1681}.<br />
Prognosis and predictive factors<br />
Since over one-half of the ESSs have<br />
already spread to pelvic or upper abdominal<br />
sites at the time of diagnosis, the<br />
tumour stage remains the major prognostic<br />
criterion {438}. Whether the neoplasm<br />
is an ESS or undifferentiated ovarian<br />
sarcoma also influences the clinical<br />
course {3208}. ESS often has a favourable<br />
outcome with survival in excess of<br />
5 years even in the context of extraovarian<br />
spread. After 10 years, however, the<br />
tumour-related mortality increases, particularly<br />
if extraovarian manifestations<br />
were noted at the time of diagnosis. Tumour<br />
relapse re p resents an ominous<br />
prognostic sign. Undifferentiated ovarian<br />
sarcomas have a rapid course and a<br />
poor prognosis {3208}.<br />
Radical panhysterectomy is the recommended<br />
therapy. Successful treatment<br />
with progesterone, non-hormonal cytostatic<br />
drugs or radiation has been reported<br />
occasionally in ESS.<br />
Endometrioid borderline tumour<br />
Definition<br />
An ovarian tumour of low malignant<br />
potential composed of atypical or histologically<br />
malignant endometrioid type<br />
glands or cysts often set in a dense<br />
f i b rous stroma with an absence of stromal<br />
invasion.<br />
Synonyms<br />
Endometrioid tumour of low malignant<br />
potential, endometrioid tumour of borderline<br />
malignancy.<br />
Epidemiology<br />
Endometrioid tumours with atypical<br />
epithelial proliferations and lacking stromal<br />
invasion are rare. Their pre c i s e<br />
prevalence is not known because of variation<br />
in diagnostic criteria, but reportedly<br />
they account for 3-18% of malignant<br />
ovarian neoplasms {137,2490,2528}.<br />
Aetiology<br />
These tumours appear to be predominantly<br />
derived from the surface epithelium<br />
of the ovary or endometriosis.<br />
Clinical features<br />
Patients range in age from 22-77 years<br />
{201,2737}. A pelvic mass is palpable in<br />
a majority of patients, and others present<br />
with uterine bleeding. The tumours are<br />
Surface epithelial-stromal tumours 135