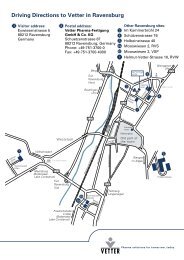
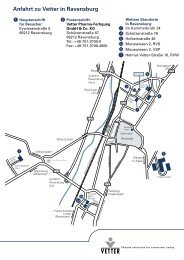
Von der Apotheke zum Weltmarktführer - Vetter im ... - Vetter Pharma
Von der Apotheke zum Weltmarktführer - Vetter im ... - Vetter Pharma
Von der Apotheke zum Weltmarktführer - Vetter im ... - Vetter Pharma
Erfolgreiche ePaper selbst erstellen
Machen Sie aus Ihren PDF Publikationen ein blätterbares Flipbook mit unserer einzigartigen Google optimierten e-Paper Software.
Das Executive Committee (von links nach rechts):<br />
Udo J. <strong>Vetter</strong>, Gunther Strothe und Wolfgang Kerkhoff<br />
The Executive Committee (from left to right):<br />
Udo J. <strong>Vetter</strong>, Gunther Strothe and Wolfgang Kerkhoff<br />
Since January 1, 2008, the company trustees have been<br />
Gunther Strothe and Wolfgang Kerkhoff. The political economist<br />
Gunther Strothe is a financial expert with international<br />
experience. From 1995 to 2006, he set up the German operations<br />
of the international Oliver Wyman global management<br />
consultancy. In addition, he is on the Board of Supervisors of<br />
Germany’s largest independent service provi<strong>der</strong> for private real<br />
estate financing. He joined the company in 1983 together with<br />
Jenik Radon via <strong>Vetter</strong> <strong>Pharma</strong>-Turm Inc. and supported the<br />
sales subsidiary in the USA as an auditor until 1990. Wolfgang<br />
Kerkhoff is a senior partner in the renowned Munich congress<br />
agency EUROKONGRESS. He is active in various international<br />
associations and gives lectures at Munich’s university.<br />
<strong>Vetter</strong> also convened a council of independent experts at<br />
the beginning of the year 2008 to act as an advisory body for<br />
management. It is made up of the trustees, representatives from<br />
business and members of the <strong>Vetter</strong> family.<br />
After some comprehensive structural changes at the beginning<br />
of the new millennium, the company is now being led as<br />
a mo<strong>der</strong>n organization and with clear corporate governance<br />
guidelines while maintaining the testamentary stipulations<br />
of Helmut <strong>Vetter</strong>. For the new trustees Gunther Strothe and<br />
Wolfgang Kerkhoff, maintaining this continuity will represent<br />
an <strong>im</strong>portant task.<br />
Another guarantee for the continuity of the company is the<br />
unified strategy of the three <strong>Vetter</strong> families. Udo J. <strong>Vetter</strong> in particular,<br />
who has been involved in the international pharmaceutical<br />
industry since the mid-1970s and has been working with the<br />
company since 1983, has been actively contributing his over 30<br />
years’ worth of experience in pharmaceuticals.<br />
At the beginning of 2008, <strong>Vetter</strong> received the first approval<br />
for the filling of a drug at the RVS facility. Permission from the<br />
US regulatory agency FDA was a crowning success for the largest<br />
investment in the company’s history. With it, <strong>Vetter</strong> un<strong>der</strong>scored<br />
its broad competencies in the manufacturing of aseptic<br />
prefilled injection systems. It was a success that was rooted not<br />
least in the many years of know-how in the construction of<br />
aseptic facilities and was achieved, too, by close collaboration<br />
with the »Regierungspräsidium« Tübingen and the FDA during<br />
the conception stage.<br />
At this point in t<strong>im</strong>e, the construction of the visual inspection<br />
and secondary packaging building in Ravensburg-Mariatal<br />
was right on schedule. By May 2008, the façade was complete<br />
and work on the interior could begin. From August 2008 till the<br />
beginning of 2009, the packaging lines were gradually built and<br />
put into commission. A total of 20 million euros was invested<br />
in the new construction, which was mostly built with partners<br />
from the region. The project was another cornerstone in assuring<br />
the company’s future and was realized within the context of<br />
growing customer requests for solutions in the rising homecare<br />
sector. Thus, pen systems and autoinjectors can also be<br />
processed in Mariatal and prefilled syringes with safety devices<br />
to prevent needlestick injuries are also an option here in view of<br />
new regulations issued by the authorities.<br />
98 …