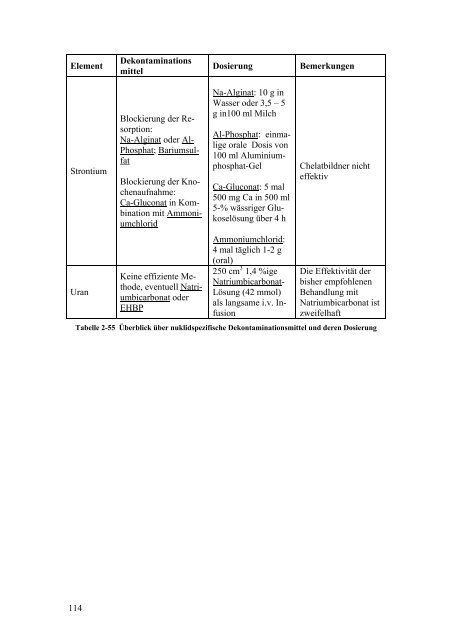
Element Strontium Uran 114 Dekontaminations mittel Blockierung der Resorption: Na-Alginat oder Al- Phosphat; Bariumsulfat Blockierung der Knochenaufnahme: Ca-Gluconat in Kombination mit Ammoniumchlorid Keine effiziente Methode, eventuell Natriumbicarbonat oder EHBP Dosierung Bemerkungen Na-Alginat: 10 g in Wasser oder 3,5 – 5 g in100 ml Milch Al-Phosphat: einmalige orale Dosis <strong>von</strong> 100 ml Aluminiumphosphat-Gel Ca-Gluconat: 5 mal 500 mg Ca in 500 ml 5-% wässriger Glukoselösung über 4 h Ammoniumchlorid: 4 mal täglich 1-2 g (oral) 250 cm 3 1,4 %ige Natriumbicarbonat- Lösung (42 mmol) als langsame i.v. Infusion Chelatbildner nicht effektiv Die Effektivität der bisher empfohlenen Behandlung mit Natriumbicarbonat ist zweifelhaft Tabelle 2-55 Überblick über nuklidspezifische Dekontaminationsmittel und deren Dosierung
3. Literatur 1. Agency for Toxic Substances and Disease Registry (ATSDR), „Toxicological profile for Americium“, Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (2004) 2. Agency for Toxic Substances and Disease Registry (ATSDR), „Toxicological profile for Cobalt“, Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (2004) 3. Agency for Toxic Substances and Disease Registry (ATSDR), „Toxicological profile for Iodine“, Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (2004) 4. Agency for Toxic Substances and Disease Registry (ATSDR). „Toxicological profile for Plutonium“. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (1990) 5. Agency for Toxic Substances and Disease Registry (ATSDR). „Toxicological profile for Strontium“. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (2004) 6. Agency for Toxic Substances and Disease Registry (ATSDR). „Toxicological profile for Uranium“. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service (1999) 7. American Academy of Clinical Toxicology, European Association of Poison Centres and Clinical Toxicologists, “Position Paper: Whole Bowel Irrigation”, Clinical Toxicology, 42:6, pp. 843 — 854 (2004) 8. Andersen O., „Chemical and Biological Considerations in the Treatment of Metal Intoxications by Chelating Agents“, Mini-Reviews in Medicinal Chemistry 4, pp. 11 – 21 (2004) 9. Ansoborlo E. et al., „Review of actinide decorporation with chelating agents“, C. R. Chimie 10 pp. 1010-1019 (2007) 10. Apostoaei A. I. and Miller L. F., “Uncertainties in dose coefficients from ingestion of I, Cs, and Sr”, Health Phys. 86(5), pp. 460–482 (2004) 11. Baker D. H., Czarnecki-Maulden D. L., “Pharmacologic Role of Cysteine in Ameliorating or Exacerbating Mineral Toxicities“, J. Nutr. 117, pp.1003- 1010 (1987) 12. Berger M. E., Jones O. W., Ricks R. C., Garrett S., „Decontaminating the Nasal Passages“, Health Phys. 84 (Supplement 2), pp. S80–S82 (2003) 13. Blanc J. et al., “Intervention medicale en cas d’evenement nucleaire ou radiologique – guide nationale“, Version 2.6, Office de Protection contre les Rayonnements ionisants (2002) 14. Bogdan G. M., Aposhian H. V., “N-(2,3-Dimercaptopropyl)phthalamidic acid (DMPA) increases Polonium-210 excretion“, Biol. Metals 3, pp. 232-236 (1990) 15. Bouvier-Capely C., Ritt J., Baglan N., Cossonnet C., “Potentialities of mass spectrometry (ICP-MS) for actinides determination in urine“, Appl. Radiat. Isot. 60, pp. 629–633 (2004) 16. Brodsky A., Wald N., „Experiences with Early Emergency Response and Rules of Thumb“, in: „Public Protection from Nuclear, Chemical, and Bio- 115