Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Poster</strong> <strong>Session</strong>, Thursday, <strong>June</strong> <strong>17</strong><br />
Theme F686 - N1123<br />
PEG assisted synthesis of Mn 3 O 4 Nanoparticles: Structural and Magnetic Study<br />
A.Baykal 1 *, M.Toma 1 , Z.Durmus 1 , H.Kavas 2 and M.S.Toprak 3<br />
1 Department of Chemistry and 2 Physics, Fatih University, B. Cekmece, 34500 Istanbul, Turkey<br />
3 Functional Materials Division, Royal Institute of Technology - KTH, SE16440 Stockholm, Sweden<br />
Abstract- In this work, Mn 3 O 4 nanoparticles have been successfully synthesized by polyethylene glycol (PEG)-assisted<br />
hydrothermal route for the first time. X-ray powder diffraction (XRD), fourier transform infrared spectroscopy (FT-IR), transmission<br />
electron microscopy (TEM) and vibrating scanning magnetometry (VSM), electron spin resonance (ESR) were used for the<br />
structural, morphological and magnetic investigation of the products, respectively.<br />
Among magnetic materials, manganese oxide<br />
(Mn 3 O 4 ) as a magnetic transition metal oxide is an<br />
important material. Nanometer-sized manganese oxide<br />
(Mn 3 O 4 ), with notable increased surface area and greatly<br />
reduced size, is expected to display better performance in<br />
these aspects of application [1]. In this study the crystallite<br />
size from X-ray diffraction pattern and particle size from<br />
transmisson electron micrographs were calculated as 23±1<br />
nm and 24.5±1.5 nm respectively. Transmisson electron<br />
microscopy (TEM) analysis also showed the<br />
polycrystalline nature of the product. Magnetic<br />
characteristics of Mn 3 O 4 NP were evaluated by electron<br />
spin resonance (ESR) measurements in the temperature<br />
range of 24 °K – 294 °K and the Curie temperature was<br />
observed as 43 K. Also the magnetic phases occured in<br />
nano sized Mn 3 O 4 are detected below Tc by this method.<br />
The room temperature paramagnetic characteristic are<br />
verified by vibrating scanning magnetometry (VSM).<br />
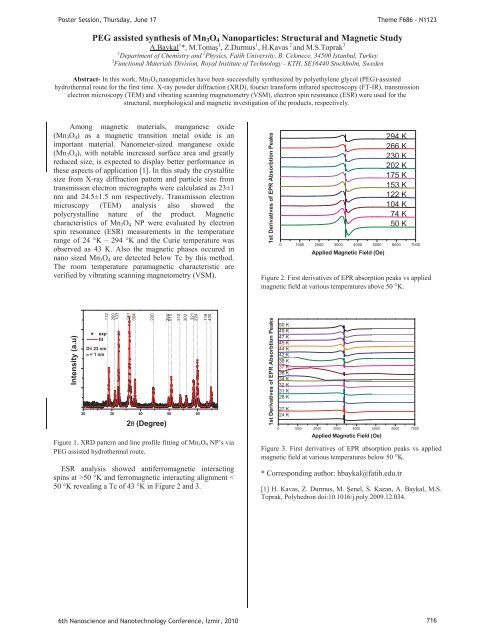
1st Derivatives of EPR Absorbtion Peaks<br />
0 1000 2000 3000 4000 5000 6000 7000<br />
Applied Magnetic Field (Oe)<br />
294 K<br />
266 K<br />
230 K<br />
202 K<br />
<strong>17</strong>5 K<br />
153 K<br />
122 K<br />
104 K<br />
74 K<br />
50 K<br />
Figure 2. First derivatives of EPR absorption peaks vs applied<br />
magnetic field at various temperatures above 50 °K.<br />
Intensity (a.u)<br />
..............................................112<br />
exp<br />
fit<br />
D= 23 nm<br />
= 1 nm<br />
.......................................................................200<br />
................103<br />
211<br />
....................................................004<br />
...........................................................220<br />
20 30 40 50 60<br />
2(Degree)<br />
.....................................................................204<br />
......................................................015<br />
..................................................................312<br />
........................................................................303<br />
.......................................................321<br />
................................................................224<br />
........................................................................116<br />
...........................................................400<br />
Figure 1. XRD pattern and line profile fitting of Mn 3 O 4 NP’s via<br />
PEG assisted hydrothermal route.<br />
ESR analysis showed antiferromagnetic interacting<br />
spins at >50 °K and ferromagnetic interacting alignment <<br />
50 °K revealing a Tc of 43 °K in Figure 2 and 3.<br />
1st Derivatives of EPR Absorbtion Peaks<br />
50 K<br />
49 K<br />
47 K<br />
45 K<br />
44 K<br />
42 K<br />
39 K<br />
37 K<br />
36 K<br />
34 K<br />
32 K<br />
31 K<br />
28 K<br />
27 K<br />
24 K<br />
0 1000 2000 3000 4000 5000 6000 7000<br />
Applied Magnetic Field (Oe)<br />
Figure 3. First derivatives of EPR absorption peaks vs applied<br />
magnetic field at various temperatures below 50 °K.<br />
* Corresponding author: hbaykal@fatih.edu.tr<br />
[1] H. Kavas, Z. Durmus, M. enel, S. Kazan, A. Baykal, M.S.<br />
Toprak, Polyhedron doi:10.1016/j.poly.2009.12.034.<br />
6th Nanoscience and Nanotechnology Conference, zmir, <strong>2010</strong> 716