Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
P<br />
P<br />
<strong>Poster</strong> <strong>Session</strong>, Thursday, <strong>June</strong> <strong>17</strong><br />
Theme F686 - N1123<br />
Hydrothermal Synthesis of LiMnR2ROR4R Cathode Active Nanoparticles for Li-ion Batteries<br />
1<br />
1<br />
UEmrah BulutUP P* and Mahmut OzacarP<br />
1<br />
PDepartment of Chemistry, Art and Science Faculty, Sakarya University, Sakarya 54187, Turkiye<br />
Abstract- Spinel LiMnR2ROR4R cathode active nanoparticles for li-ion batteries were synthesized by hydrothermal route at low temperatures. The<br />
LiMnR2ROR4R nanoparticles synthesized via hydrothermal technique were investigated by X-ray diffraction (XRD) and scanning electron microscopy<br />
Lithium-ion batteries are nowadays widely used for<br />
portable systems, such as telephones, computers and<br />
telecommunication devices. One of the most studied<br />
materials in this field is spinel lithium manganese oxide<br />
(LiMnR2ROR4R), which is considered as a promising alternative<br />
to LiCoOR2R, currently used in the lithium-ion batteries.<br />
LiMnR2ROR4R is a cubic spine1 with space group symmetry<br />
Fd3m. The lithium ions are located on the 8a tetrahedral sites<br />
of the structure; the manganese ions are positioned on the<br />
16d octahedral sites. The oxygen ions, which are cubicclose-packed<br />
(ccp) occupy the 32e positions.<br />
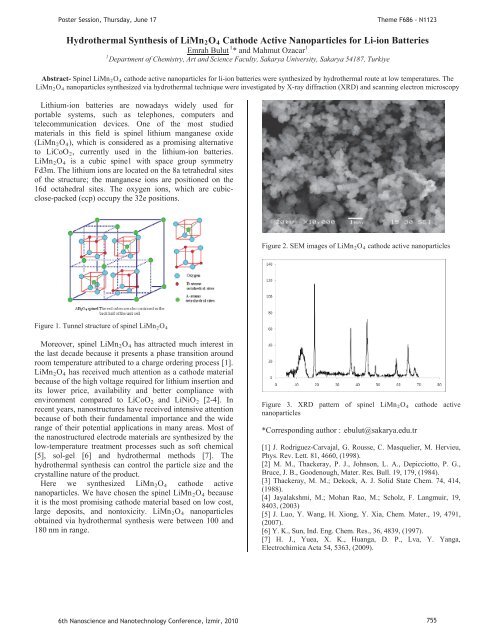
Figure 2. SEM images of LiMnR2ROR4R cathode active nanoparticles<br />
Figure 1. Tunnel structure of spinel LiMnR2ROR4R<br />
Moreover, spinel LiMnR2ROR4R has attracted much interest in<br />
the last decade because it presents a phase transition around<br />
room temperature attributed to a charge ordering process [1].<br />
LiMnR2ROR4R has received much attention as a cathode material<br />
because of the high voltage required for lithium insertion and<br />
its lower price, availability and better compliance with<br />
environment compared to LiCoOR2R and LiNiOR2R [2-4]. In<br />
recent years, nanostructures have received intensive attention<br />
because of both their fundamental importance and the wide<br />
range of their potential applications in many areas. Most of<br />
the nanostructured electrode materials are synthesized by the<br />
low-temperature treatment processes such as soft chemical<br />
[5], sol-gel [6] and hydrothermal methods [7]. The<br />
hydrothermal synthesis can control the particle size and the<br />
crystalline nature of the product.<br />
Here we synthesized LiMnR2ROR4R cathode active<br />
nanoparticles. We have chosen the spinel LiMn R2ROR4R because<br />
it is the most promising cathode material based on low cost,<br />
large deposits, and nontoxicity. LiMnR2ROR4R nanoparticles<br />
obtained via hydrothermal synthesis were between 100 and<br />
180 nm in range.<br />
Figure 3. XRD pattern of spinel LiMnR2ROR4R cathode active<br />
nanoparticles<br />
*Corresponding author : HTebulut@sakarya.edu.trT<br />
[1] J. Rodriguez-Carvajal, G. Rousse, C. Masquelier, M. Hervieu,<br />
Phys. Rev. Lett. 81, 4660, (1998).<br />
[2] M. M., Thackeray, P. J., Johnson, L. A., Depicciotto, P. G.,<br />
Bruce, J. B., Goodenough, Mater. Res. Bull. 19, <strong>17</strong>9, (1984).<br />
[3] Thackeray, M. M.; Dekock, A. J. Solid State Chem. 74, 414,<br />
(1988).<br />
[4] Jayalakshmi, M.; Mohan Rao, M.; Scholz, F. Langmuir, 19,<br />
8403, (2003)<br />
[5] J. Luo, Y. Wang, H. Xiong, Y. Xia, Chem. Mater., 19, 4791,<br />
(2007).<br />
[6] Y. K., Sun, Ind. Eng. Chem. Res., 36, 4839, (1997).<br />
[7] H. J., Yuea, X. K., Huanga, D. P., Lva, Y. Yanga,<br />
Electrochimica Acta 54, 5363, (2009).<br />
6th Nanoscience and Nanotechnology Conference, zmir, <strong>2010</strong> 755