Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
Third Day Poster Session, 17 June 2010 - NanoTR-VI
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
P ions<br />
P<br />
P have<br />
P contact<br />
<strong>Poster</strong> <strong>Session</strong>, Thursday, <strong>June</strong> <strong>17</strong><br />
Theme F686 - N1123<br />
The Creation of Hydrophobic Clay Surfaces with Long Chain Hydrocarbon<br />
1<br />
1<br />
1<br />
UH. Hasan YolcuUP P*, Ahmet GürsesP P, and Metin AçkyldzP P,<br />
1<br />
PAtaturk University, K.K. Education Faculty, Dep. of Chemistry, 25240 Erzurum<br />
Abstract- In this study, the presence of lotus effect for the clay modified by using long chain hydrocarbon was investigated. It was<br />
found the powder sample comparison with pellet form exhibits super hydrophobic character. This may be attributed to the changing of<br />
roughness on the surface of modified clay particles depending on pressing and to the creating of higher energy surface with the adsorption of<br />
long chain hydrocarbon onto interlayer region of clay.<br />
Non wettable surfaces with high water contact angle<br />
(WCA) and facile sliding of drops are called super<br />
hydrophobic surface. Superhydrophobic surfaces with<br />
0<br />
water contact angles larger than 150P received a lot<br />
of research attention, due to important applications ranging<br />
from self cleaning materials to microfluidic devices [1, 2].<br />
Many surfaces in nature are highly hydrophobic and self<br />
cleaning (e.g. lotus leaves). The design synthesis and<br />
application of new kinds of super hydrophobic and self<br />
cleaning organic or inorganic material will be essential and<br />
important task to fulfill [3].<br />
The clay sample was purified by sedimentation, dried at<br />
vacuum oven and sieved to give a 38-85 m (>%92) size<br />
fraction using ASTM Standard sieves. Different amounts<br />
of hydrocarbon (0.05-1.0 g) was mixed with 500 mL<br />
aqueous solutions of CTAB (100, 200, 240, 260, 300, and<br />
320 mg/L). The mixture was shaken at 293 K, for 30 mins<br />
and 1g clay sample was added to this mixture and shaken<br />
for 30 mins in a thermostatic shaker at 200 rpm. The<br />
modified clay samples which produced by above<br />
procedure were filtered through filter paper of Whatman<br />
41 and dried at 383 K in a vacuum oven for 2 h.<br />
and water droplets leads to gaining the hydrophobic<br />
character of samples. But the powder organoclay has<br />
superhydrophobic character, probably due to lotus effect.<br />
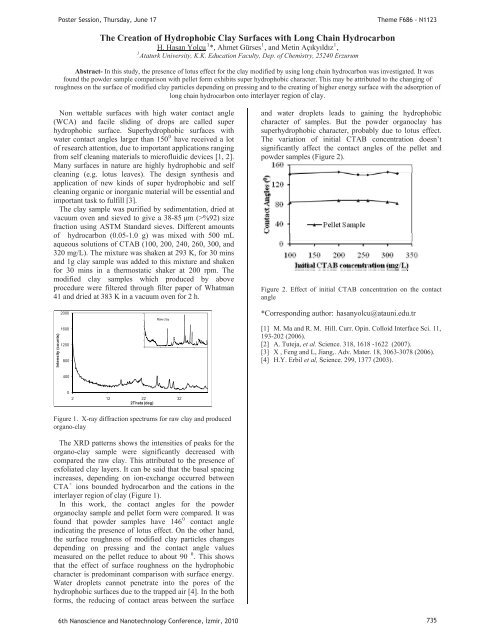
The variation of initial CTAB concentration doesn’t<br />
significantly affect the contact angles of the pellet and<br />
powder samples (Figure 2).<br />
Figure 2. Effect of initial CTAB concentration on the contact<br />
angle<br />
Intensity (counts)<br />
2000<br />
1600<br />
1200<br />
800<br />
Raw clay<br />
*Corresponding author: HThasanyolcu@atauni.edu.trT<br />
[1] M. Ma and R. M. Hill. Curr. Opin. Colloid Interface Sci. 11,<br />
193-202 (2006).<br />
[2] A. Tuteja, et al. Science. 318, 1618 -1622 (2007).<br />
[3] X , Feng and L, Jiang,. Adv. Mater. 18, 3063-3078 (2006).<br />
[4] H.Y. Erbil et al, Science. 299, 1377 (2003).<br />
400<br />
0<br />
2 12 22 32<br />
2Theta (deg)<br />
Figure 1. X-ray diffraction spectrums for raw clay and produced<br />
organo-clay<br />
The XRD patterns shows the intensities of peaks for the<br />
organo-clay sample were significantly decreased with<br />
compared the raw clay. This attributed to the presence of<br />
exfoliated clay layers. It can be said that the basal spacing<br />
increases, depending on ion-exchange occurred between<br />
+<br />
CTAP bounded hydrocarbon and the cations in the<br />
interlayer region of clay (Figure 1).<br />
In this work, the contact angles for the powder<br />
organoclay sample and pellet form were compared. It was<br />
0<br />
found that powder samples have 146P angle<br />
indicating the presence of lotus effect. On the other hand,<br />
the surface roughness of modified clay particles changes<br />
depending on pressing and the contact angle values<br />
0<br />
measured on the pellet reduce to about 90 P P. This shows<br />
that the effect of surface roughness on the hydrophobic<br />
character is predominant comparison with surface energy.<br />
Water droplets cannot penetrate into the pores of the<br />
hydrophobic surfaces due to the trapped air [4]. In the both<br />
forms, the reducing of contact areas between the surface<br />
6th Nanoscience and Nanotechnology Conference, zmir, <strong>2010</strong> 735